Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Enthalpy (ΔH), Hess’s Law, Calorimetry, Entropy (ΔS), Gibbs Free Energy (ΔG)
Learning Objectives
Analyze energy changes in chemical systems through enthalpy, Hess's Law, entropy, and spontaneity.
Prior Knowledge Needed
Stoichiometry, bond enthalpy, exo/endothermic reactions
IB Syllabus Reference
R1.1, R1.2
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
3 Lessons Available
Calorimetry
Gibbs Free Energy_Born Haber Cycles
Hess Cycles
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
What is temperature?
The measure of the average kinetic energy of a sample of molecules.
What is a system?
The substances inside a reaction vessel.
What are the types of system?
1. Open. 2. Closed. 3. Isolated.
What does exothermic mean?
When energy is released from a system to the surroundings.
What does endothermic mean?
When energy is absorbed from the surroundings into the system.
What happens when bonds are broken?
Energy is absorbed.
What happens when bonds are made?
Energy is released.
What is the enthalpy change?
A change in the total energy of a substance. Measured in kJmol^-
What are the standard conditions?
100kPa. 298K. Standard states.
Why use standard conditions when measuring enthalpy change?
1. Small changes to temperature, pressure and states will have a significant effect on enthalpy change. 2. Standard conditions are used to limit these variables.
What is the standard enthalpy change?
A change in the total energy of a substance under standard conditions, measured in kilojoules per mole.
What is calorimetry?
The process of measuring the amount of heat released or absorbed during a chemical reaction.
What is the equation for heat energy?
q = m * c * ΔT
What is the molar enthalpy change?
The heat energy change per mole of substance.
What is the equation for molar enthalpy change?
- H = -q / n
What is Hess' Law?
The enthalpy change accompanying a chemical change is independent of the route by which the chemical change occurs.
What is the standard enthalpy of combustion?
The energy required to react one mole of a substance completely with oxygen, under standard conditions, usually measured in kilojoules per mol.
What is the enthalpy of formation?
The energy required to form one mole of compound from its constituent elements, under standard conditions, usually measured in kilojoules per mol.
What is the enthalpy change of neutralisation?
The enthalpy change when one mole of water molecules are formed when an acid reacts with an alkali under standard conditions. Measured in kilojoules per mole. Will always be exothermic.
endothermic
a reaction in which more energy is absorbed than released

exothermic
a reaction in which more energy is released than absorbed

standard enthalpy of a reaction
The change in heat content of a system per mole of reactant under standard conditions: 100kPa 1 mol dm^-3 concentration for all solutions All substances in their standard states
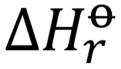
standard enthalpy of formation (ΔHf)
the change in enthalpy when one mole of a substance in the standard state (1 atm of pressure and 298.15 K) is formed from its pure elements under the same conditions
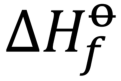
standard enthalpy of combustion (ΔHc)
the enthalpy change when 1 mole of a substance burns (combines vigorously with oxygen) under standard state conditions
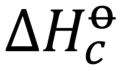
bond enthalpy
the energy needed to break one mole of bonds in gaseous molecules under standard conditions averaged over similar compounds
enthalpy of a reaction equation
H=Σ(n Hf products) - Σ(n Hf reactants)
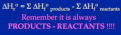
calorimetry
The precise measurement of heat flow into or out of a system for chemical and physical processes

calorimetry equation
q=mcΔT

specific heat capacity
the energy required to raise the temperature of one gram of a substance by one degree Celsius

Specific heat capacity of water
4.18 J / g K

cooling curve (calorimetry)
a graph of the variation of the temperature with time as a substance is allowed to cool; used to determine specific temperature changes in calorimetry experiments

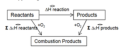
Hess Cycle
A diagram showing a reaction taking place by two or more alternative routes, all of which involve the same enthalpy change, according to Hess's law. Also called an enthalpy cycle.
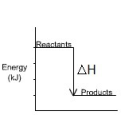
enthalpy level diagram
A diagram that shows the relative enthalpies of the reactants and products in a chemical reaction.
Standard enthalpy change of reaction (ΔHƟ).
Enthalpy change measured under standard conditions
Enthalpy Standard conditions (pressure and temperature)
A pressure of 100 kPa A temperature of 25°C (298 K)
Is product or reactant more stable in exothermic reactions
products ( think of heating and condensing water and the bonds)
Structure of a calorimeter
polystyrene cup (good heat insulator and absorbs very little heat itself)
Errors in calorimetry
Systematic - heat loss to environment Random - always present, do more trials Assumed c is same for substance as water
The enthalpy change of neutralisation (ΔHn)
Enthalpy change when an acid and base react together to form one mole of water.
standard enthalpy of combustion (ΔHƟc)
The enthalpy change when one mole of a substance is burned completely in oxygen under standard conditions
Limitations of finding enthalpy using a bunsen burner
1. heat loss to surroundings 2. Incomplete combustion 3. Assumption of specific heat capacity and density
Hess's law
States that the total enthalpy change in a chemical reaction is independent of the route by which the chemical reaction takes place, as long as the initial and final conditions are the same.
µ Standard enthalpy change of formation (ΔH f )
The enthalpy change when one mole of a compound is formed from the elements in their standard states under standard conditions.
enthalpy change of a reaction can also be calculated using standard enthalpy change of combustion
reactants - products
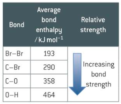
Limitation of bond enthalpy
- only valid for GASES - calculations are inaccurate because the intermolecular forces are not taken into account. - environment of a bond not taken into account Enthalpy changes for reactions calculated from average bond enthalpy data may differ slightly from experimental values. This is because they are average values taken from the same bonds in a range of similar compounds.
Ozone Formation
O2 has double bond (bond energy 498) and UVC light, breaks to form two oxygen radicals, one oxygen radical reacts with O2 gas and forms O3 O3 is has dissociated electrons (364) and UVB then can make an oxygen gas and a radical
26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments
Paper 1A: MCQ Practice
PDF
Paper 1B: Data & Experiments
PDF
Paper 2: Short Response
PDF
Official Markscheme
PDF
17
Cl
35.45
Assignments & Labs Section 08