Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Significant Figures, Random vs Systematic Error, Absolute/Percentage Uncertainty, Precision vs Accuracy
Learning Objectives
Master the principles of experimental design, uncertainty propagation, and graphical analysis of chemical data.
Prior Knowledge Needed
Basic algebra, scientific notation, metric system conversions
IB Syllabus Reference
S1.1
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
2 Lessons Available
Errors and Uncertainty
Graphing Data
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
random error
Error that is due to chance and is not standardized. Decreases precision.
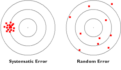
systematic error
Error that shifts all measurements in a standardized way. Decreases accuracy. Can result in bias
accuracy
A description of how close a measurement is to the true value of the quantity measured.
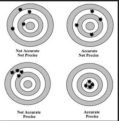
precision
a measure of how close a series of measurements are to one another
percent uncertainty
the ratio of the uncertainty of a measurement to the measured value, expressed as a percent- age

relative uncertainty
Uncertainty of a quantity divided by the value of the quantity. Decimal version of percent uncertainty.

absolute uncertainty
expresses the margin of uncertainty associated with a measurement. For an analogue scale, it is half of the smallest measurement increment. For a digital scale, it is equivalent to the most precise decimal place of the instrument.

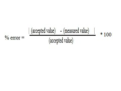
the absolute value of the theoretical value minus the experimental value divided by the theoretical value, multiplied by 100%
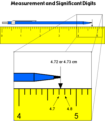
analog scale
A scale with demarcations of the same interval. You can read a measurement based on where it falls on the scale. Read ONE DIGIT past the smallest increment on the scale.

digital scale
An electronic scale with a sensor that measures a specified amount and displays the result electronically.

significant figures
All the digits that can be known precisely in a measurement, plus a last estimated digit
a measure of how close a series of measurements are to one another

meniscus
the curve at a liquid's surface by which one measures the volume of the liquid

reproducible
A measurement is reproducible if the investigation is repeated by another person, or by using different equipment or techniques, and the same results are obtained.

repeatable
A measurement is repeatable if the original experimenter repeats the investigation using same method and equipment and obtains the same results.

26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments
Paper 1A: MCQ Practice
PDF
Paper 1B: Data & Experiments
PDF
Paper 2: Short Response
PDF
Official Markscheme
PDF
17
Cl
35.45
Assignments & Labs Section 08