Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Ionic Lattice, VSEPR Theory, Resonance, Bond Enthalpy, Polar vs Non-polar Bonds
Learning Objectives
Investigate the forces that hold matter together, from ionic and covalent lattices to metallic bonding models.
Prior Knowledge Needed
Valence electrons, Lewis structures, electronegativity (S3.1)
IB Syllabus Reference
S2.1, S2.2
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
6 Lessons Available
Covalent Bonding and Properties of Covalent Compounds
IB Chem 1 Paper Chromatography
Intermolecular Forces
Ionic Bonding and Properties of Ionic Compounds
Lewis Structures_VSEPR Theory_Metallic Bonding and Alloys
Resonance Structures_Formal Charge_Hybridization
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
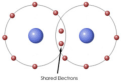
covalent bond
A chemical bond that involves sharing a pair of electrons between atoms in a molecule
bond length
the distance between two bonded atoms at their minimum potential energy, that is, the average distance between two bonded atoms

bond strength
energy required to break a bond (measured using bond enthalpy)
octet rule
atoms react by gaining or losing electrons so as to acquire the stable electron structure of a noble gas, usually eight valence electrons

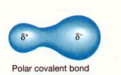
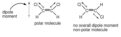
polar covalent bond
A covalent bond between atoms that differ in electronegativity. The shared electrons are pulled closer to the more electronegative atom, making it slightly negative and the other atom slightly positive.
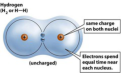
nonpolar covalent bond
A type of covalent bond in which electrons are shared equally between two atoms of similar electronegativity.
a covalent bond in which two atoms share one pair of electrons
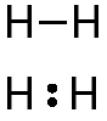
double bond
A covalent bond in which two pairs of electrons are shared between two atoms
triple bond
a covalent bond in which two atoms share three pairs of electrons

dipole
a molecule that has two poles, or regions, with opposite charges
dipole moment
a measure of the separation and magnitude of the positive and negative charges in polar molecules
network covalent (giant molecular covalent) structure
A chemical structure in which the atoms are bonded by a group of covalent bonds in a continuous network.
polyatomic ion
a group of covalently bonded atoms that behaves as a unit and has a positive or negative charge
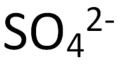
coordinate covalent bond
forms when one atom donates a pair of electrons to be shared with an atom or ion that needs two electrons to become stable

incomplete octet
These elements are stable with fewer than eight electrons in their valence shell and include hydrogen (2), helium (2), lithium (2), beryllium (4), and boron (6).
VSPER theory
Valence-shell electron-pair repulsion theory; because electron pairs repel, molecules adjust their shapes so that valence electron pairs are as far apart as possible

molecular domain geometry
The arrangements of atoms in space of a molecule. Describes the shape of the bonding domains, taking into consideration repulsive effects of lone pairs.
metallic bond
a bond formed by the attraction between positively charged metal ions and the delocalized electrons around them
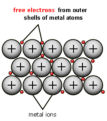
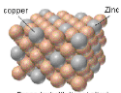
alloy
A mixture of two or more metals; enhances the strength of metals due to the restricted movement of ions over each other and the non-directional movement of delocalized electrons.
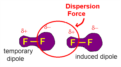
van der Waals forces
the two weakest intermolecular attractions- London dispersion interactions and dipole forces

London dispersion forces
the intermolecular attraction resulting from the uneven distribution of electrons and the creation of temporary dipoles
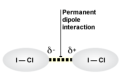
dipole-dipole forces
intermolecular forces that exist between polar molecules. Active only when the molecules are close together. The strengths of intermolecular attractions increase when polarity increases
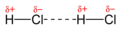
dipole-induced dipole
a weak attraction that results when a polar molecule induces a dipole in an atom or in a nonpolar molecule by disturbing the arrangement of electrons in the nonpolar species
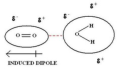
hydrogen bonding
the intermolecular force in which a hydrogen atom that is bonded to a highly electronegative atom (NOF) is attracted to an unshared pair of electrons of an electronegative atom in a nearby molecule
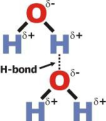
intermolecular force
a weak force of attraction between molecules, ions, or atoms
dipole (IB definition)
permanent separation of electrical charge in a molecule due to unequal distributions of bonding and/or lone pairs of electrons
solubility
A measure of how much solute can dissolve in a given solvent at a given temperature.

boiling point
the temperature at which a substance changes from a liquid to a gas; results from breaking intermolecular bonds (or ionic bonds for ionic compounds).
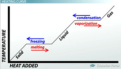
melting point
the temperature at which a substance changes from a solid to a liquid

paper chromatography
method of separating a mixture of different substances. The liquid mobile phase soaks through the stationary paper and carries the mixture with it. Some substances are carried faster than others so the substances are separated along the paper due to differences in intermolecular forces.
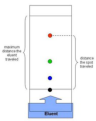
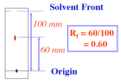
Rf factor
pigment
A colored chemical compound that absorbs light, producing color; produces opaque mixture in water

dye
a usually soluble substance that produces color

26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments
Paper 1A: MCQ Practice
PDF
Paper 1B: Data & Experiments
PDF
Paper 2: Short Response
PDF
Official Markscheme
PDF
17
Cl
35.45
Assignments & Labs Section 08