Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Rate Equations, Activation Energy, Maxwell-Boltzmann Distribution, Rate-Determining Step, Arrhenius Equation
Learning Objectives
Investigate dynamic chemical kinetics, including rate constant calculations and Arrhenius theory models.
Prior Knowledge Needed
Collision theory, energy profiles, stoichiometry (R1.1)
IB Syllabus Reference
R2.2
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
4 Lessons Available
Instantaneous Rates
Maxwell Boltzmann Distribution Curves
Rate Laws
Reaction Mechanisms_Arrhenius Plots
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
kinetics
the study of reaction rates
rate of reaction
the change in concentration of a reactant or product per unit time
Units of rate
mol dm-3 s-1
Maxwell Boltzmann Distribution Curve
The distribution of the molecular speeds of gas particles at a given temperature; as temper- ature increases, average kinetic energy increases and the distribution becomes wider and flatter.
activation energy (Ea)
minimum kinetic energy required for a reaction to occur
kinetic energy
energy of motion
collision theory
For a reaction to occur, the particles must collide, they must collide with the appropriate orientation, and they must collide with sufficient energy.
Colorimetry
Using an instrument or device to determine chemical concentration by comparison of a liquid's color with standard colors; used to measure reaction rates by examining changes in color concentration over time

Spectrophotometry
An analytical method for identifying a substance by its selective absorption of different wavelengths of light; used to measure the rate of reaction by examining changes in ab- sorbance/transmittance over time

conductivity
method of measuring reaction rates through change in concentration of ions; usually done using a calibration curve

instantaneous rate
The reaction rate at a particular time as opposed to the average rate over an interval of time.

titration
A solution of known concentration is used to determine the concentration of another solution; used to determine reaction rate by taking reaction samples at various time intervals

clock reaction
a reaction in which some visible change takes place in the reaction mixture when a certain point in the reaction is reached; more convenient way of measuring the rate of a reaction by taking single measurement

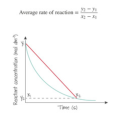
a reaction rate calculated from a change in concentration divided by a change in time
26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments
Paper 1A: MCQ Practice
PDF
Paper 1B: Data & Experiments
PDF
Paper 2: Short Response
PDF
Official Markscheme
PDF
17
Cl
35.45
Assignments & Labs Section 08