Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Brønsted-Lowry Theory, pH Scale, Strong vs Weak Acids, Buffer Solutions, Ka/Kb/Kw
Learning Objectives
Explore Brønsted-Lowry theory, the pH scale, and the properties of strong and weak acid/base systems.
Prior Knowledge Needed
Equilibrium, stoichiometry, hydrogen ion structure
IB Syllabus Reference
R3.1
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
5 Lessons Available
Arrhenius Acids_Bases and Acid Base Reactions
Bronsted Lowry Acids_Bases and Strength of Acids_Bases
Environmental Effects of Acid Rain
Lewis Acids_Ka
Salt Hydrolysis_Buffers
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
acid rain
Rain containing acids that form in the atmosphere when industrial gas emissions (especially sulfur dioxide and nitrogen oxides) combine with water. pH < 5.6

litmus

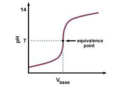
equivalence point
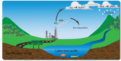
acid deposition
Sulfur oxides and nitrogen oxides, emitted by burning fossil fuels, enter the atmos- phere-where they combine with oxygen and water to form sulfuric acid and nitric acid-and return to Earth's surface
dry deposition
A form of acid deposition in which dry, sulfate-containing particles settle out of the air.

leaching
Process in which various chemicals in upper layers of soil are dissolved and carried to lower layers and, in some cases, to groundwater.

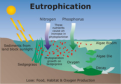
A process by which nutrients, particularly phosphorus and nitrogen, become highly con- centrated in a body of water, leading to increased growth of organisms such as algae or cyanobacteria.
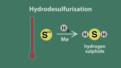
hydrodesulfurization
A process in which sulfur is removed from the molecules in any refinery stream by reacting it with hydrogen in the presence of a catalyst.
flue gas desulfurization
Nonregenrative systems, where the reagent is used to remove the sulfur oxides from the gas stream is used and discarded; regenerative means that the reagent is recovered and reused.
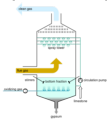
catalytic converter
a platinum, coated device that oxidizes most of the VOCs and some of the CO that would otherwise be emitted in exhaust, converting them to CO2.
amphiprotic
A species that can either accept or donate a proton.
amphoteric
any species that can react as either an acid or a base

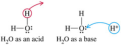
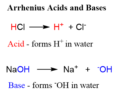
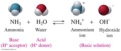
Arrhenius acid
a substance that increases the concentration of hydrogen ions in aqueous solution

Arrhenius base
a substance that increases the concentration of hydroxide ions in aqueous solution
Bronsted-Lowry acid
a molecule or ion that is a proton donor
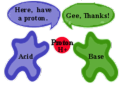
Bronsted-Lowry base
a molecule or ion that is a proton acceptor
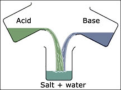
neutralization reaction
a reaction in which an acid and a base react in an aqueous solution to produce a salt and water
metal + acid --> salt + hydrogen gas
acid-carbonate reaction
acid + carbonate -> salt + water + carbon dioxide

pH
power of hydrogen; measures hydrogen ion concentration in solution; -log[H+]

pOH
-log[OH-]

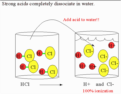
strong acid/base
dissociate/ionize completely in aqueous solution; will be a strong electrolyte
weak acid/base
partially dissociate/ionize in aqueous solution; will be a weak electrolyte
26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments
Paper 1A: MCQ Practice
PDF
Paper 1B: Data & Experiments
PDF
Paper 2: Short Response
PDF
Official Markscheme
PDF
17
Cl
35.45
Assignments & Labs Section 08