Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Homologous Series, Functional Groups, IUPAC Nomenclature, Isomerism, SN1/SN2 Mechanisms
Learning Objectives
Master the structure, nomenclature, and electron-sharing mechanisms of complex organic molecules.
Prior Knowledge Needed
Covalent bonding, molecular geometry, hybridization (S2.1, S2.4)
IB Syllabus Reference
S2.5, R3.2
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
5 Lessons Available
Isomers
Organic Chem Nomenclature
Organic Reaction Mechanisms
Organic Reactions
Spectroscopic Identification of Organic Compounds
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
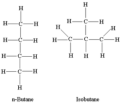
structural isomer
Compounds that have the same molecular formula but differ in the covalent arrangements of their atoms.
primary carbon atom
a carbon atom in an organic molecule that is directly bonded to one other carbon atom
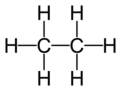
secondary carbon atom
a carbon atom in an organic molecule that is directly bonded to two other carbon atoms
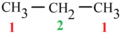
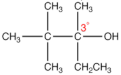
tertiary carbon atom
a carbon atom in an organic molecule that is directly bonded to three other carbon atoms
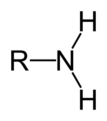
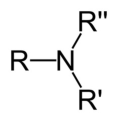
primary amine
An amine where the nitrogen atom is attached to one alkyl chain
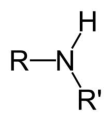
secondary amine
An amine where the nitrogen atom is attached to two alkyl chains
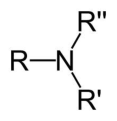
An amine in which the central nitrogen atom is connected to 3 carbon atoms.
aromatic organic compound
An organic compound where some or all of the carbon atoms are found in a delocalised ring structure such as a benzene ring
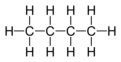
aliphatic organic compound
An organic compound where carbon atoms are joined together in straight or branched chains or non-aromatic rings
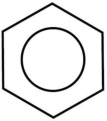
benzene
Consists of a ring of six carbon atoms with delocalized electrons, resulting in an average bond order of 1.5
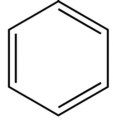
Kekule structure
A classical structural formula for an aromatic compound (first proposed structure of benzene), showing localized double bonds.
a measure of how readily a substance vaporizes; decreases with increasing hydrocarbon chain length and strength of intermolecular forces between organic compounds

inorganic
Not formed from living things or the remains of living things; can contain carbon (e.g. carbon oxides and carbonates).
organic compound
a covalently bonded compound that contains carbon, excluding carbonates and oxides

catenation
the covalent bonding of carbon to itself to form chains or rings
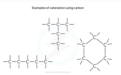
hydrocarbon
An organic molecule consisting only of carbon and hydrogen.
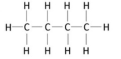
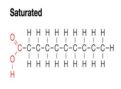
saturated
hydrocarbons containing only single bonds
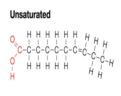
unsaturated
hydrocarbons containing double or triple carbon-carbon bonds
functional group
group of atoms within a molecule that interacts in predictable ways with other molecules
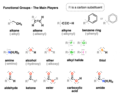
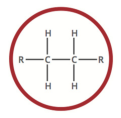
alkane
a hydrocarbon containing only single covalent bonds
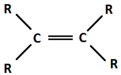
alkene
C=C
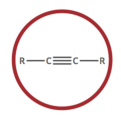
alkyne
carbon-carbon triple bond
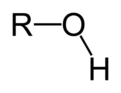
alcohol
R-OH; contains hydroxyl functional group
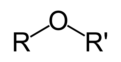
R-O-R
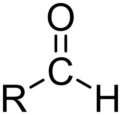
aldehyde
An organic molecule with a carbonyl group covalently bonded to a hydrogen
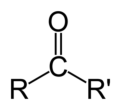
ketone
R-C=O-R
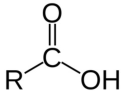
carboxylic acid
R-COOH
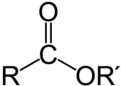
ester
RCOOR
amide
amine covalently bonded to a carbonyl group; functional group is called carboxyamide
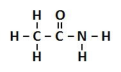
R-NH2
nitrile
carbon triple bonded to nitrogen
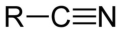
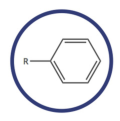
arene
aromatic ring; contains phenyl functional group
structural formula
a chemical formula that shows the arrangement of atoms in a molecule or a polyatomic ion; each dash between a pair of atoms indicates a pair of shared electrons

condensed structural formula
structural molecular formula showing the general arrangement of atoms but without showing all the covalent bonds
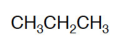
molecular formula
A chemical formula that shows the number and kinds of atoms in a molecule, but not the arrangement of the atoms.
stereochemical formula
a chemical formula that represents the three-dimensional shape of the molecule
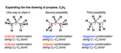
nomenclature
naming system
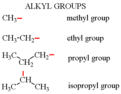
substituent
side chain on an organic compound; determines the prefix in organic compound nomencla- ture
stem
determined by longest carbon chain in organic compound nomenclature
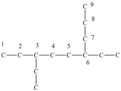
suffix
determined by the functional group in organic compound nomenclature

26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments
Paper 1A: MCQ Practice
PDF
Paper 1B: Data & Experiments
PDF
Paper 2: Short Response
PDF
Official Markscheme
PDF
17
Cl
35.45
Assignments & Labs Section 08