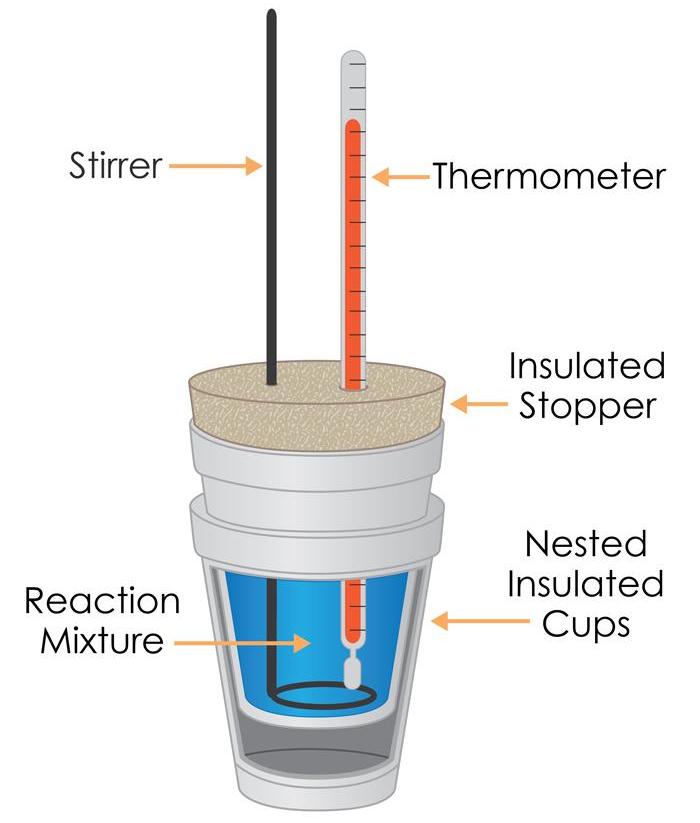
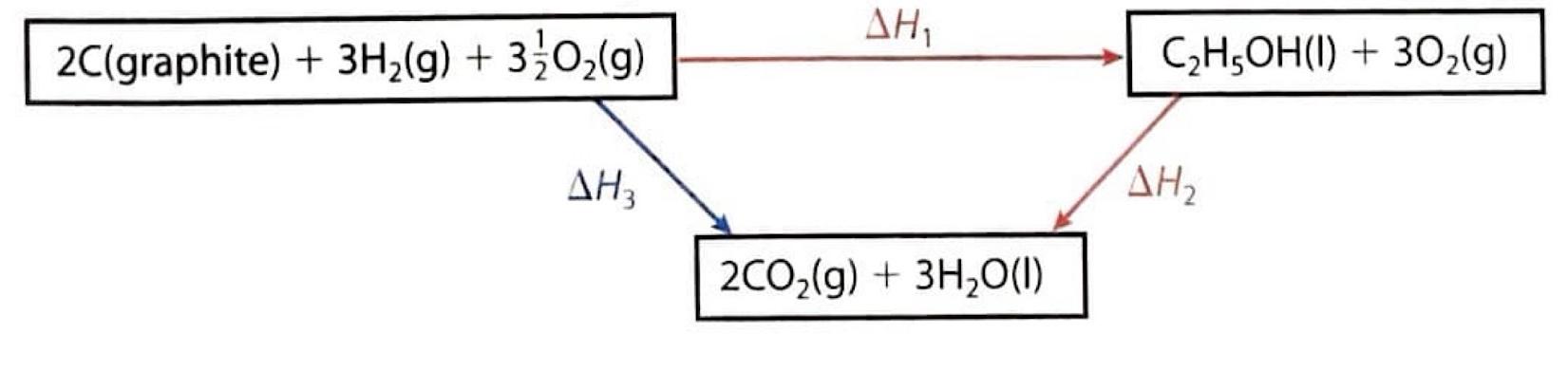
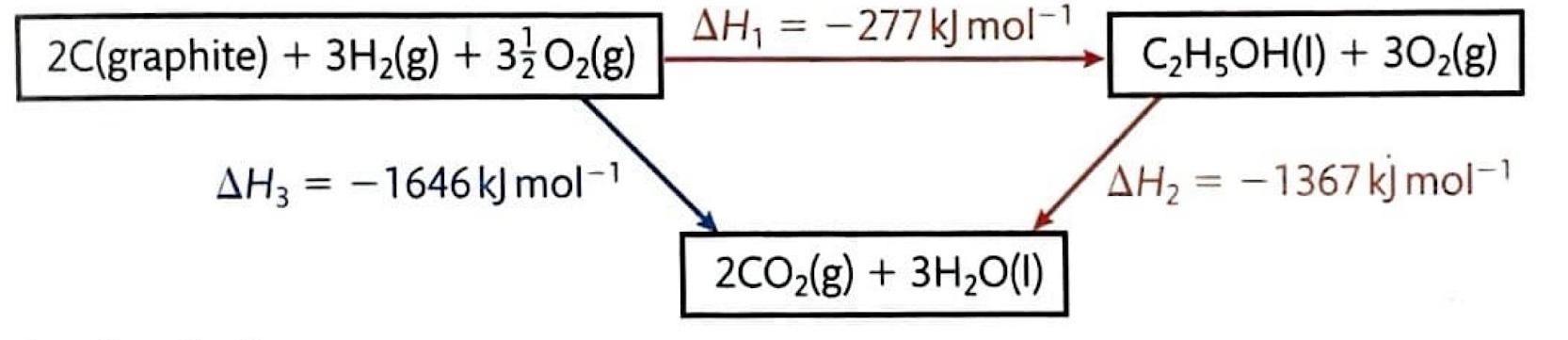
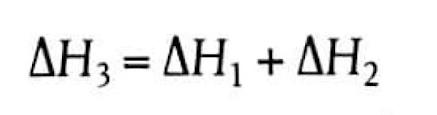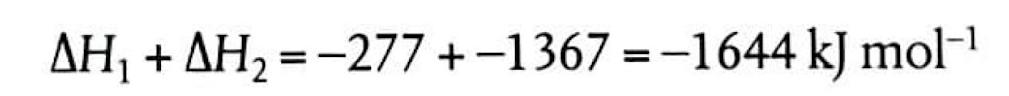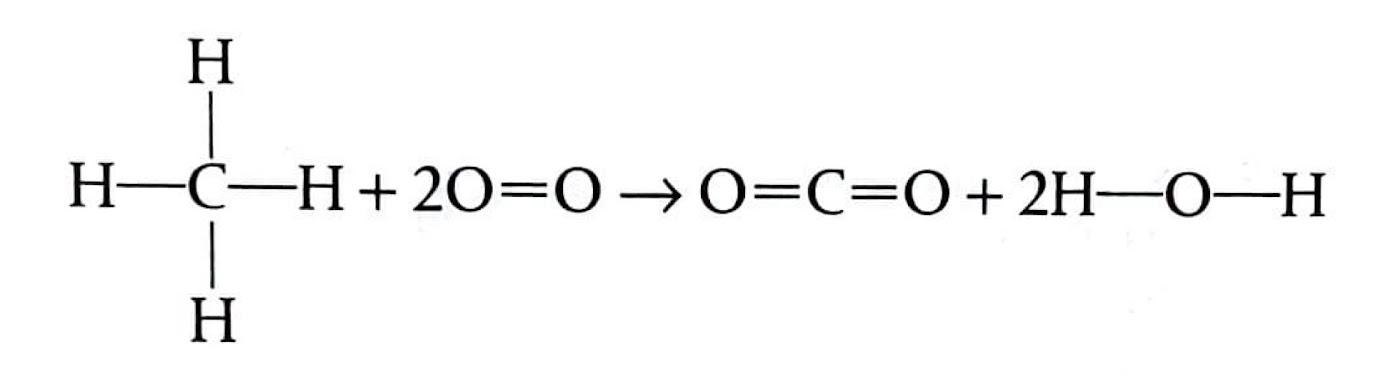Understanding Bond Enthalpies: Breaking and Forming Bonds
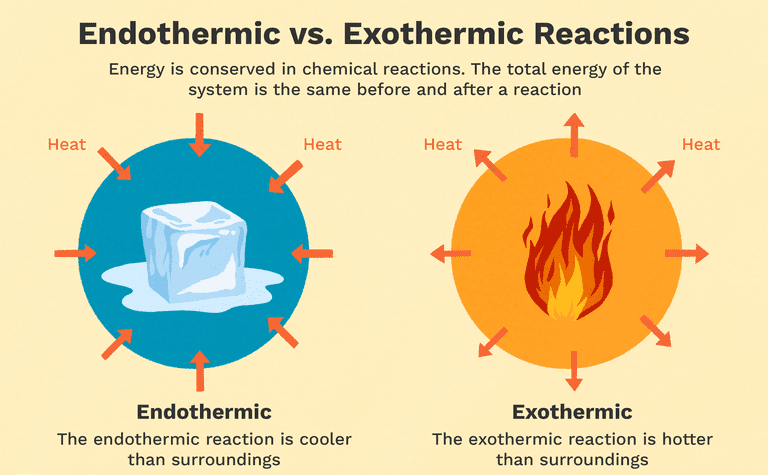
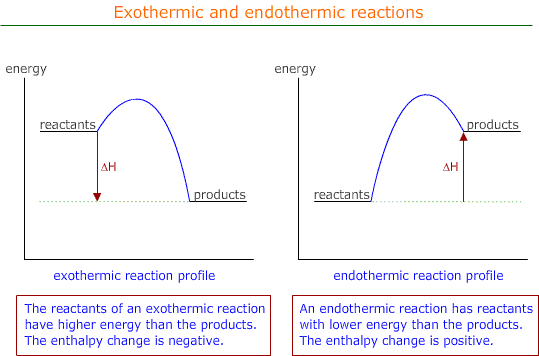
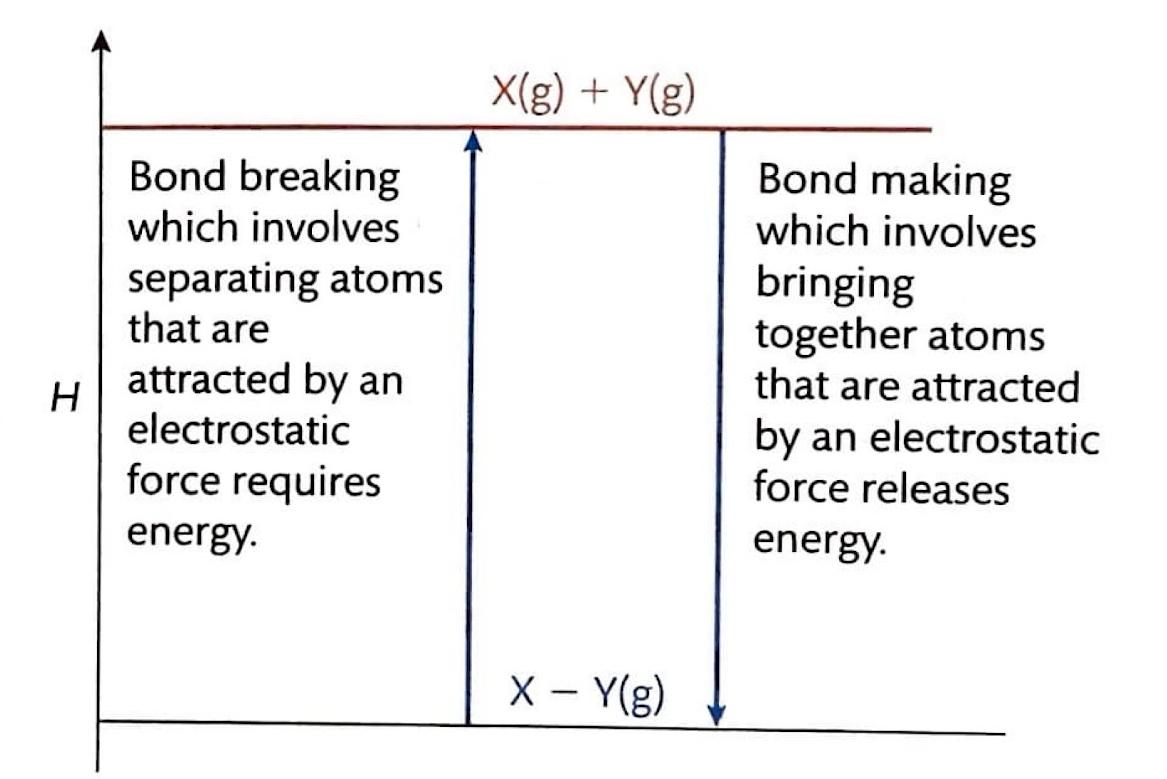
Chemical reactions fundamentally involve the breaking of existing chemical bonds and the formation of new ones. The energy changes associated with these processes are quantified by bond enthalpies. Breaking chemical bonds is an endothermic process, meaning it requires an input of energy from the surroundings. Conversely, the formation of chemical bonds is an exothermic process, releasing energy into the surroundings. For instance, the dissociation of gaseous chlorine molecules into individual chlorine atoms, represented as Cl
2(g) → 2Cl(g), has a positive enthalpy change (ΔH° = +242 kJ mol
-1), indicating energy absorption. In contrast, the formation of a hydrogen molecule from two gaseous hydrogen atoms, H(g) + H(g) → H
2(g), has a negative enthalpy change (ΔH° = -436 kJ mol
-1), signifying energy release. The overall enthalpy change of a chemical reaction is determined by the balance between the energy absorbed during bond breaking and the energy released during bond formation. If more energy is released than absorbed, the reaction is overall exothermic.
Defining Average Bond Enthalpy
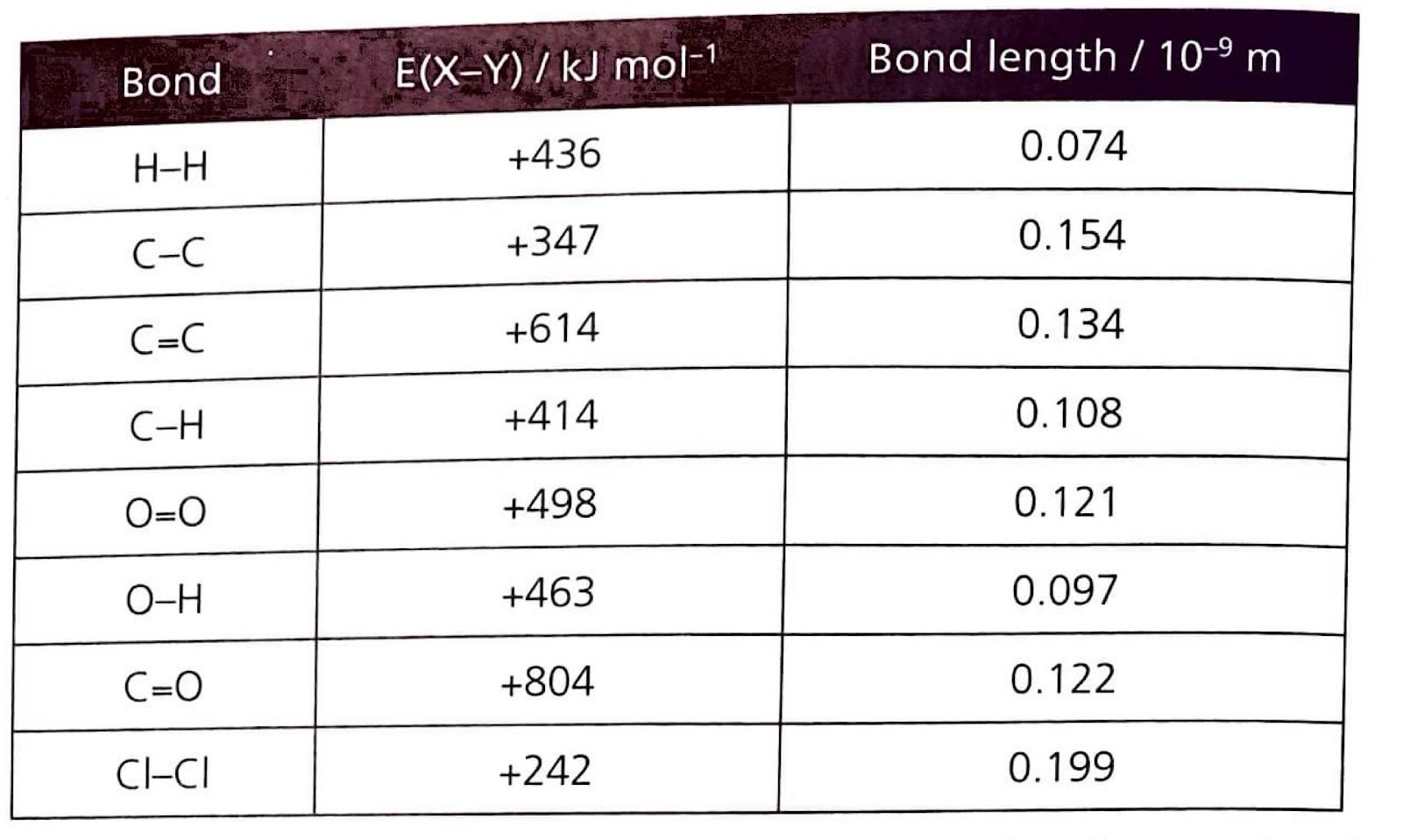
The average bond enthalpy is a crucial concept in thermochemistry. It is defined as the energy required to break one mole of a specific type of bond in gaseous molecules, under standard conditions, averaged over a range of similar compounds. This averaging is necessary because the energy of a particular bond can vary slightly depending on its molecular environment. Generally, multiple bonds, such as double or triple bonds, possess higher bond enthalpies and shorter bond lengths compared to their single bond counterparts, reflecting the greater electrostatic attraction between the bonded atoms.
Calculating Enthalpy Change Using Bond Enthalpies
Bond enthalpies can be used to estimate the enthalpy change of a reaction (ΔH°
rxn). The principle is that the total energy required to break all bonds in the reactants minus the total energy released when all bonds in the products are formed gives the overall enthalpy change. This can be expressed by the formula:
ΔH°
rxn = ΣΔH°
(bonds broken) - ΣΔH°
(bonds formed).
Consider the combustion of methane as an example:
CH
4(g) + 2O
2(g) → CO
2(g) + 2H
2O(g)
To calculate the enthalpy change, we sum the bond enthalpies of the bonds broken in the reactants and subtract the sum of the bond enthalpies of the bonds formed in the products.
| Bonds Broken |
ΔH / kJ mol-1 |
Bonds formed |
ΔH / kJ mol-1 |
| 4 C-H |
4(414) |
2 C=O |
2(804) |
| 2 O=O |
2(498) |
4 O-H |
4(463) |
| Total |
=2652 |
|
=3460 |
Using the formula:
ΔH° = ΣΔH°
(bonds broken) - ΣΔH°
(bonds formed)
ΔH° = 2652 kJ mol
-1 - 3460 kJ mol
-1
ΔH° = -808 kJ mol
-1
This negative value indicates that the combustion of methane is an exothermic reaction.
Bond Order and Ozone Depletion
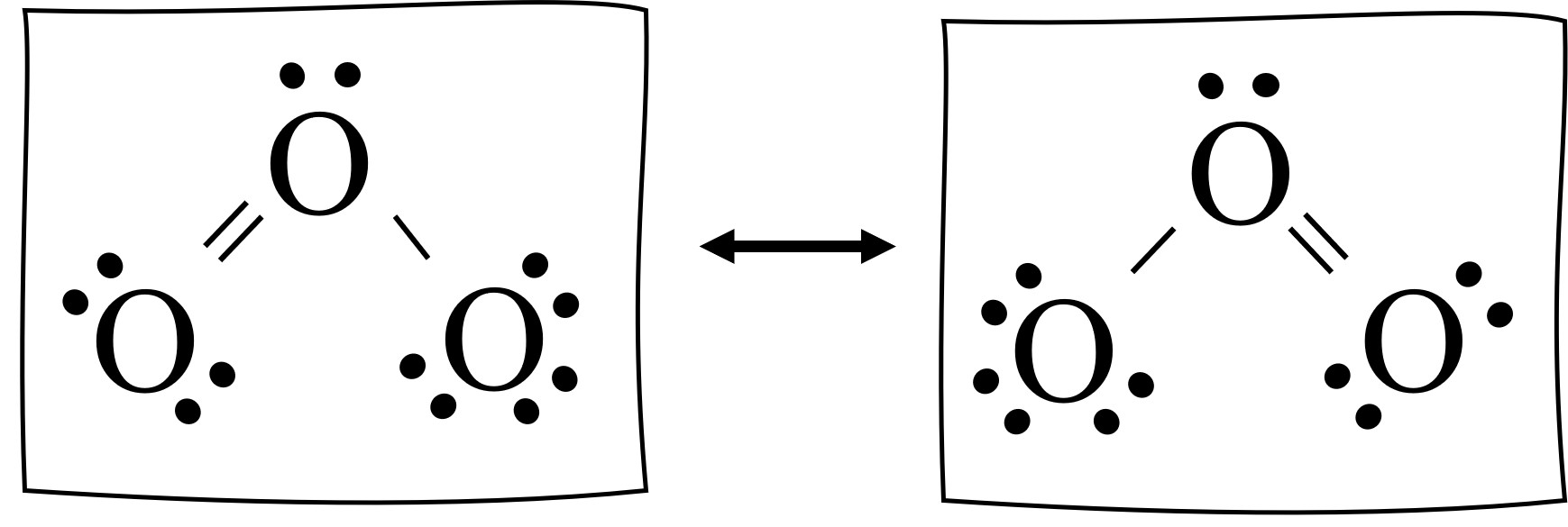
The stability of molecules and their susceptibility to bond breaking by radiation can be related to their bond order. For ozone (O
3), the bond order is 1.5, indicating a resonance hybrid between single and double bonds. In contrast, diatomic oxygen (O
2) has a bond order of 2, representing a stronger double bond. Consequently, the bonds in oxygen molecules are stronger than those in ozone molecules. This difference in bond strength means that ozone bonds require less energy to break and can be dissociated by lower energy, longer wavelength ultraviolet (UV) light compared to the bonds in diatomic oxygen. This characteristic is fundamental to understanding ozone depletion in the stratosphere.
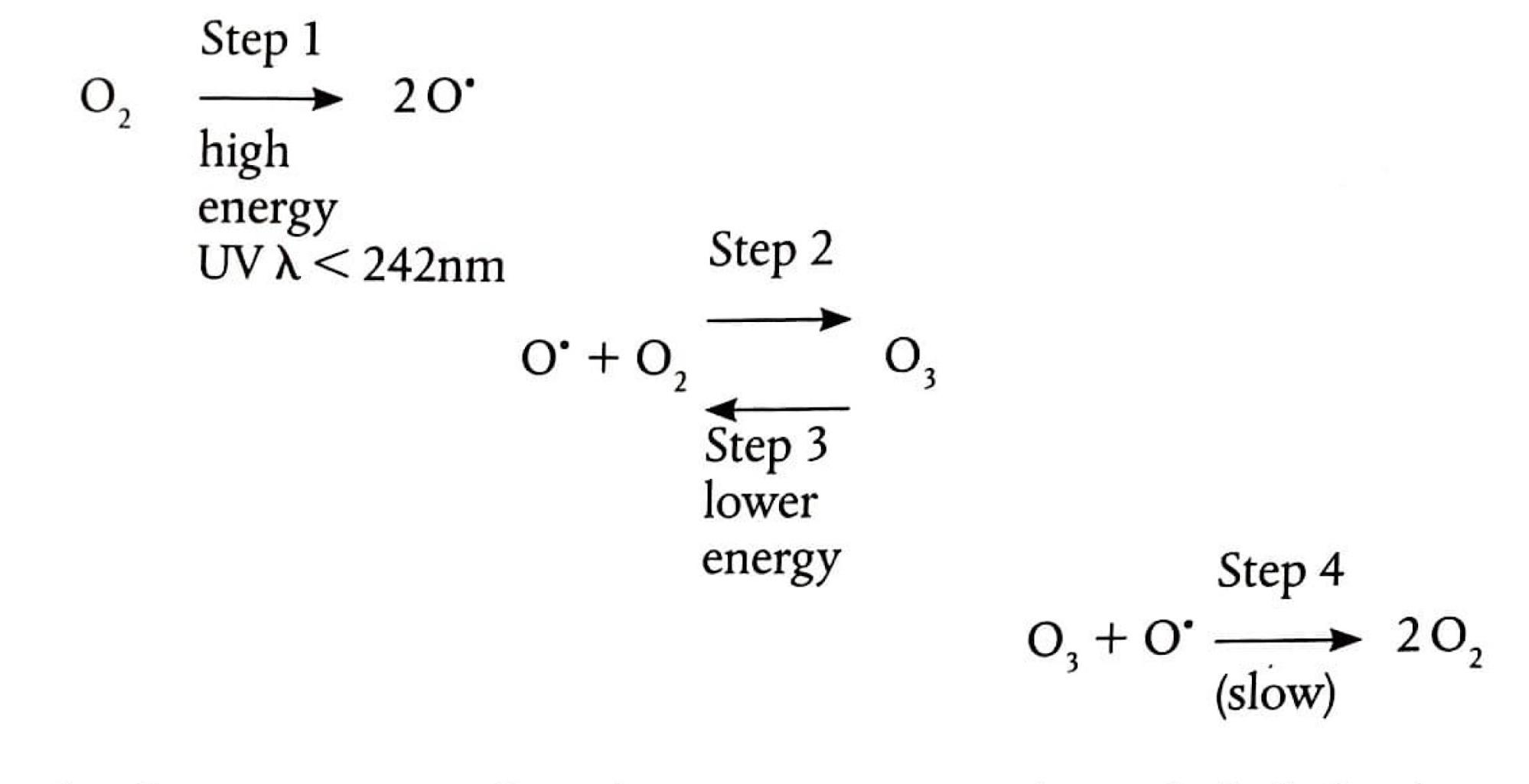
The Chapman Cycle: Ozone Formation and Removal
The concentration of ozone in the stratosphere is maintained through a dynamic equilibrium known as the Chapman Cycle. This cycle involves both the formation and removal of ozone, ideally at balanced rates, leading to a steady state. The process begins with the photodissociation of diatomic oxygen (O
2) by high-energy UV radiation, forming highly reactive oxygen free radicals (O•). This bond breaking is endothermic. These free radicals then react with intact oxygen molecules to form ozone (O
3). The formation of ozone is an exothermic process because new bonds are formed. Ozone can then be removed through two primary mechanisms: photodissociation by UV light, breaking it back into O
2 and O•, or by reacting with an oxygen free radical to form two molecules of O
2. Both of these removal processes are also exothermic. The delicate balance of these reactions ensures the protective ozone layer remains stable.
Calculating Wavelength for Bond Breaking
The energy required to break a chemical bond can be directly related to the wavelength of electromagnetic radiation needed to cause that dissociation. This relationship is described by Planck's equation, E = hf, where E is energy, h is Planck's constant, and f is frequency. Since the speed of light (c) is related to frequency and wavelength (λ) by c = λf, we can substitute f = c/λ into Planck's equation to get E = hc/λ, or rearranged to find wavelength: λ = hc/E.
Let's calculate the wavelength of UV radiation needed to break the bond in ozone, given that its bond energy is 363 kJ mol
-1.
First, convert the molar bond energy to the energy required to break a single bond:
Energy per photon (E
photon) = (363,000 J mol
-1) / (6.02 × 10
23 mol
-1) = 6.03 × 10
-19 J
Now, use the rearranged Planck's equation:
λ = (h × c) / E
photon
λ = (6.63 × 10
-34 J s × 3.00 × 10
8 m s
-1) / (6.03 × 10
-19 J)
λ = 3.30 × 10
-7 m
Converting this to nanometers:
λ = 3.30 × 10
-7 m × (10
9 nm / 1 m) = 330 nm
This calculation demonstrates that UV radiation with a wavelength of 330 nm is sufficient to break the bonds in ozone. Remember to use Avogadro's Number to convert between molar energy and the energy of a single photon.