Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Alloys, Transition Metal Complexes, Formal Charge, Sigma/Pi Bonds, Hybridization
Learning Objectives
Examine how chemical bonding and structure dictate the physical properties and design of modern materials.
Prior Knowledge Needed
Covalent bonding, metallic bonding, coordination (S2.1, S2.2)
IB Syllabus Reference
S2.3, S2.4
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
2 Lessons Available
Addition Polymers
Condensation polymerization
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
1. How can we We can look at models like: categorise the -> Ionic arrangement of -> Covalent atoms based on -> Metallic electrostatic in- teractions?
2. Chemical bond- So far, we have classified chemical bonding based on the electrostatic interactions ing between charged particles: Ionic - between oppositely charged ions. Covalent - between a shared pair of electrons and positively charged nuclei. Metallic - between cations in a lattice and delocalised electrons.
3. Bonding triangle -> allows classification of a bond between two elements by placing it on a vertex on the triangle, depending on whether the elements are metals or non-metals. -> If the elements making up a chemical bond are both metals, the bonding is classified as metallic. -> If the chemical bond is made up of a metal and non-metal, the bonding is classified as ionic. -> Finally, if the bond is made up of two non-metals, the bonding is classified as covalent -> Notice that the three bonding models are placed together in one triangle instead of separating them entirely.
metallic to covalent bonding. -> This means that bonding is best described as a continuum between the ionic, covalent and metallic models rather than discrete categories.
5. Properties of a material using bonding models
6. Metallic bonding Bonding description: electrostatic attraction between positively charged metal summary cations and a sea of delocalised valence electrons Description of structure: lattice
7. Covalent bond- Bonding description: electrostatic attraction between a shared pair of electrons ing summary and a positively charged nuclei Description of structure: small of large molecules, covalent network structures
8. ionic bonding Bonding description: electrostatic attraction between oppositely charged ions summary Description of structure: lattice
9. What can bond- The three bonding models can be used to explain the properties of a material. ing models be used for?
10. What are the diffi- -> Covalent bonding is usually the most difficult to assign specific physical proper- culties in assign- ties because these properties depend on intermolecular interactions and whether ing physical prop- or not the substance is a small molecule, large molecule or covalent network solid. erties to covalent bonds?
12. Insulating Property of a substance that has a high resistance to thermal or electrical conduc- tivity. -> when a material does not permit thermal energy or an electric current to flow freely -> This is usually due to the absence of freely moving particles.
13. Physical proper- -> electrical conductivity ties of metals -> Melting point -> Insulating -> Flexibility -> Brittleness -> Malleability -> Thermal conductivity
14. What is required To be an insulator, all charged particles in the structure should be fixed and not to be a good insu- permitted to freely move throughout the structure. lator?
15. How does chem- chemical bonding does not exist in discrete categories but rather as a continuum ical bonding ex- between ionic, covalent and metallic models ist?
16. Tendency in met- Metals have a high tendency to lose electrons, low tendency to attract additional als electrons, therefore low electronegativity values.
17. Tendency in non Non-metals have a high tendency to gain electrons, therefore high electronega- metals tivity values.
-> When metals combine with other metals, they are less likely to form compounds, instead they form something called an alloy
19. State an example Steel is an example of an alloy that is a mixture of iron with small amounts of carbon of an alloy
20. Structure of al- The structure of alloys is similar to elemental metals, the key difference being that loys the added element will vary in size from the metal making up the base metal
21. Substitutional al- - where the element added to the base metal simply replaces the metal ions in the loy lattice.
22. Interstitial alloy where the element added occupies vacant space in the metallic lattice of the base metal.
23. What occurs in In both of these types of alloys, bonding is non-directional since the electrostatic both types of al- force between cations and delocalised electrons still occurs in all directions. loys?
-> Alloys are a solid solution held together with metallic bonds -> Alloys can have different chemical properties than their component elements
25. Physical proper- Malleability/hardness ties of alloys Melting point.
26. Malleability/ -> The addition of elements with different sizes prevents the layers in the metallic hardness lattice from sliding past one another easily. -> This causes the alloy to be less malleable and harder than the base metal.
27. Melting point -> The addition of elements into the lattice structure of a base metal will influence the strength of the metallic bond. -> For many alloys, this lattice structure with varying cation size weakens the electrostatic attraction between the cations and the delocalised electrons. -> This causes many alloys to have lower melting temperatures than the base metal due to this weaker metallic bond -> the melting point for alloys is listed as an approximate temperature. -> This is because the melting point will change depending on the amount of alloying element added.
28. Phase diagrams -> Chemists studying alloys regularly make use of a phase diagram which helps to track the melting point of alloys as the percent composition is altered. -> To construct a phase diagram, scientists would study what temperature the alloy of each percentage composition would: show signs of melting complete melting.
Physical properties: High strength, very hard material Uses: Construction materials
30. Alloy: Stainless Alloy: Stainless steel steel Constituent elements: Steel + Cr, Ni Physical properties Resistant to corrosion Uses: Metal does not rust as easily, Kitchenware and surfaces that needs to be cleaned readily
31. Alloy: Bronze Alloy: Bronze Constituent elements: Cu + Sn Physical properties: Hard material, low melting point, Uses: Weapons and tools
32. Alloy: Brass Alloy: Brass Constituent elements: Cu + Zn Physical properties Resistant to corrosion Uses: Musical instruments, gears, hinges and valves
33. Alloy: Aluminium Alloy: Aluminium alloy alloy Constituent elements: Al + Cu, Mn, Ni, Zn Physical properties: High strength, resistant to corrosion, Low density (due to low density of Al) Uses: Aerospace manufacturing and gas cylinders for scuba diving
34. Alloy: Solder Alloy: Solder Constituent elements: Sn + Pb Physical properties: Low melting point. More conductive than Pb but less conduc- tive than Sn Uses: Connecting electrical wires and plumbing pieces
Physical properties: Hardens quickly upon formation of alloy, Resistant to corro- sion Uses: Dental fillings
36. Alloy: Sterling sil- Alloy: Sterling silver ver Constituent elements: Ag + Cu Physical properties: Strong and hard, Resistant to tarnish Uses: Victorian era cutlery sets and jewellery
37. Alloy: White gold Alloy: White gold Constituent elements: Au + Pd, Ni, Zn Physical properties: Hard and strong, Light grey colour Uses: Jewellery
38. Alloy: rose gold Alloy: Rose gold Constituent properties: Au + Cu Physical properties: Hard and strong. Red hue in colour Uses: Jewellery
39. Corrosion -> many alloys have resistance to corrosion as a physical property. -> Corrosion is the deterioration of a material on its surface as it interacts with substances in the surroundings. -> For metals, corrosion is typically the formation of brittle metal oxides on the surface due to the process of oxidation.
40. Melting points of Alloys tend to have lower melting points than their base metals, not higher melting alloys points.
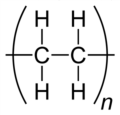
polymer Carbon is also the backbone of most polymer structures.
42. What is the back- Carbon bone of most polymer struc- tures?
43. Structural formu- Representation of a molecule that shows the order in which atoms are bonded. las for polymers -> makes it much easier to identify the repeating unit of the polymer
44. Plastic -> Plastic is a category of polymer that can be moulded or extruded into a desired shape. -> Plastics are a wide range of materials used for consumer products, medical products, packaging, textiles, electronics, transportation, construction and more
45. Main properties Flexible of plastics Durable Mould easily into desired shapes Lightweight Good electrical and thermal insulators
46. Polymer struc- -> They involve very long chains of molecules, but the individual polymer chains are tures held together by intermolecular forces. -> The intermolecular forces holding separate polymer chains together is similar to how intermolecular forces hold separate sheets together within graphite.
the intermolecular forces between polymer chains, the more flexible a plastic is. -> Conversely, the stronger the intermolecular forces between polymer chains, the more rigid a plastic is.
48. Durability of poly- -> Durability is the ability of a material to be functional over extended periods of mers time. -> Polymer chains are made up of a very large number of covalent bonds that do not break apart easily. -> This results in a long-lasting material that is durable. -> Since the large number of covalent bonds do not break apart easily, they are difficult to break down. -> Once a plastic is made it is very difficult to get rid of. -> This is a global concern as plastics accumulate in landfills all over the world.
49. Polymers ability -> Since polymers are formed from smaller molecules that make up the repeating to mould into de- subunits called monomers, they can be moulded into shape during the formation sired shapes of the polymer as monomers react together. -> In addition, since polymer chains are held together by weak intermolecular forces, many polymer plastics can be melted and reshaped to form new materials. -> A plastic container for example will melt and reshape when placed too close to the stove.
-> Carbon has a relatively low atomic mass, further contributing to the low density, lightweight nature of plastics.
51. Polymers and in- -> In all of the plastic polymer examples, there are no examples where electrons sulation can delocalise throughout the polymer chains. -> This makes plastic polymers poor electrical and thermal conductors, which therefore make them good electrical and thermal insulators.
52. Properties of The intermolecular interactions between polymer chains are correlated with the polymers and in- observed physical properties. termolecular in- teractions
53. Three common- Polyethylene (also known as polyethene) ly encountered Polyvinylchloride (also known as PVC or polychloroethene) plastic polymers Kevlar are:
54. Polyethelyne Intermolecular forces between polymer chains: London dispersion forces and dipole-dipole forces Properties: rigid Example of use: plumbing
55. Kevlar Intermolecular forces between polymer chains: London dispersion forces and hydrogen bonding Properties: Flexible Example of use: plastic bags
56. Polyvinylchloride Intermolecular forces between polymer chains: London dispersion forces and hydrogen bonding Properties: Very strong and tough Example of use: Bulletproof vests
57. Synthetic poly- -> Polymers intentionally produced that are not found in nature. mers -> Synthetic materials are those made by humans that cannot be found in nature. -> Monomers for synthetic polymers are usually derived from fossil fuels.
58. Natural polymers -> Polymers found in nature.
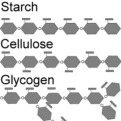
59. examples of nat- -> Sugars such as cellulose found in wood and plants, and starch found in foods ural polymers: such as rice, potatoes and wheat. -> Proteins making up your skin, hair, muscles and nails are polymers made out of amino acids as the repeating units. -> DNA making up your genetic code is made up of two polymer strands that interact with one another through hydrogen bonding forming a double helix. -> Spider silk made from a polymer that has structural similarities to Kevlar.
60. Teflon -> Teflon™ is a material commonly used in non-stick cookware. -> You might have a frying pan in your kitchen that has a teflon coating on the cooking surface. -> Teflon is made of the polymer tetrafluoropolyethene (PTFE). -> This polymer has a relatively high melting point (327 °C), it does not interact with water and is very unreactive with most substances. -> These properties make it ideal for use in cookware.
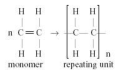
-> When the polymer is formed, one of the bonds in the double bond is broken in each monomer allowing a single bond to form with other monomers.
62. How are all how the polymer formed is named after the monomer with the prefix poly- added in all addition poly- front mers named?
63. What is required I. The monomer requires a double bond. for addition poly- II. The monomers undergo an addition reaction merisation?
64. What is required For addition polymerisation to occur we need a monomer with a double bond or to undergo addi- triple bond between carbon atoms in its structure. tion polymerisa- tion?
65. % ionic character can be determined by assessing the difference in electronegativity between the atoms (section 9 of the data booklet for the EN values)
66. the properties of - non directional bonding metals can be ex- - delocalised electrons plained in terms - ions of different size of having
67. using a bond- - physical properties ing triangle, what - type of bonding present can be deduced - whether it is an element or a compound about an un- known triangle?
monomer
small chemical unit that makes up a polymer
polymer
large compound formed from combinations of many monomers
addition polymer
a polymer formed by chain addition reactions between monomers that contain a double bond
hydrocarbon
Compounds composed of only carbon and hydrogen
homologous series
A series of organic compounds with the same functional group but with each successive member differing by CH2
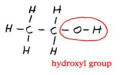
alcohol
R-OH
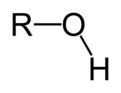
A functional group consisting of a hydrogen atom joined to an oxygen atom by a polar covalent bond. Molecules possessing this group are soluble in water and are called alcohols.
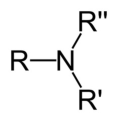
amine
R-NH2
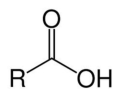
carboxylic acid
R-COOH
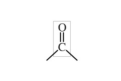
carbonyl group
C=O
amide
a carbonyl group with one R group being an NH2
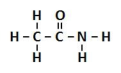
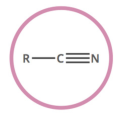
nitrile
carbon triple bonded to nitrogen
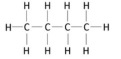
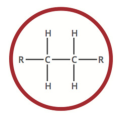
a hydrocarbon containing only single covalent bonds
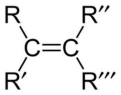
alkene
carbon-carbon double bond
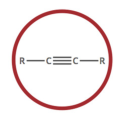
alkyne
carbon-carbon triple bond
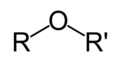
ether
R-O-R
aldehyde
An organic molecule with a carbonyl group located at the end of the carbon skeleton; one of the R groups is a hydrogen atom
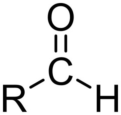
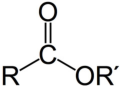
ester
RCOOR
structural formula
a formula that shows the arrangement of atoms in the molecule of a compound.
functional group
A specific configuration of atoms commonly attached to the carbon skeletons of organic molecules and involved in chemical reactions.

halogenoalkane
An alkane with at least one halogen atom in place of a hydrogen atom
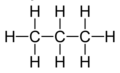
repeating unit
the shortest repeating section of a polymer
26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments
Paper 1A: MCQ Practice
PDF
Paper 1B: Data & Experiments
PDF
Paper 2: Short Response
PDF
Official Markscheme
PDF
17
Cl
35.45
Assignments & Labs Section 08