Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Periodic Law, Atomic/Ionic Radii, Ionization Energy, Electronegativity, Electron Affinity
Learning Objectives
Predict patterns and trends in the physical and chemical properties of elements using the periodic table.
Prior Knowledge Needed
Atomic structure, valence electrons, shells/subshells (S1.3)
IB Syllabus Reference
S3.1
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
2 Lessons Available
Breaks in Ionization Energy Trends
Periodic Trends
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
Alkali Metals (group)
group 1 on the periodic table.
Alkaline Earth Metals (group)
group 2 on the periodic table.
Transition Metals (group)
groups 3-12.
Halogens (group)
group 17.
valence electrons
are found in the outermost energy level.
Noble Gases (group)
group 18.
Electronegativity
A measure of the ability of an atom in a chemical compound to attract electrons

Ionization Energy
the energy required to remove one mole of electrons from one mole of atoms in its gaseous state

Atomic Radius
Distance from the center of an atom's nucleus to its outermost electron

Metals
lose electrons/form cations.
Non Metals
gain electrons/form anions. upper right corner of periodic table.
Orbital
region of space where electrons are most likely to be found.
Halogens (picture)

Alkali Metals (picture)

Alkaline Earth Metals (picture)
Metalloids (picture)
Non Metals (picture)
Transition Metals (picture)
Noble Gases (Picture)

Periods/Rows.
From left to right across the periodic table.
Alkaline Earth Metals (v.e.)
two valence electrons.
Halogens (v.e.)
seven valence electrons.
Noble Gases (v.e)
8 valence electrons/ least reactive.
Noble Gases
high ionization energy/ NO electronegativity.
Electronegativity Trend Across A Period goes (up/down)
goes up left to right across period/ row
Electronegativity Trend Down a Group goes (up/down)
goes down while going down the group/column/family
Ionization energy Trend Across A Period (increase/decrease)
increases left to right across period/row.
Ionization Energy Trend Down A Group (increase/decrease)
decreases going down the group/column/family.
Atomic Radius Trend Across A Period
decrease right to left across the period/row..
Atomic Radius Trend Down A Group
increases going down the group/column/family
Periodic Law
The principle that states that chemical properties of the elements are periodic functions of their atomic numbers
Nuclear Charge
number of protons in the nucleus

Shielding Effect
Refers to the protection of valence electrons by core electrons in inner energy levels (distance)
Which of the following has the largest atomic radius: Ga Al Si
Ga
Which of the following has the smallest atomic radius Ca Mg Sr
Mg
Which of the following has the largest atomic radius: Cl S P
P
Which of the following has the largest ionization energy: P, S, Cl,
Cl
Which of the following has the largest ionization energy: N K He
He
Which of the following has the smallest ionization energy: N K He Na Ca
K
Which of the following has the smallest electronegativity Li, Be, Mg, Na
Na
Which of the following has the smallest electronegativity: Br As K
As
Which of the following has the highest electronegativity: He F I Cl
F
Which of the following has the largest atomic radius: C N O
C
An increase in distance between a proton and electron (increase/ decrease) the force.
decrease (force of attraction)
As orbitals are added down a group how does the attractive force between valence electrons and the nucleus charge change? (Increase or Decrease)
Decrease (force of attraction)
The attractive force (increase/ decrease) as number of protons in the nucleus increases.
Increase (force of attraction)
Periodicity
A regular periodic variation of properties of elements with atomic number and position in the Periodic Table.

effective nuclear charge
the charge that a valence electron experiences from the nucleus, equal to the nuclear charge but reduced by any shielding or screening from any intervening electron distribution

shielding
the effect on an electron of repulsion by electrons in lower-energy orbitals that screen it from the full effects of nuclear charge
ionic radius
Distance from the center of an ion's nucleus to its outermost electron

electron affinity
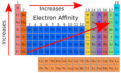
metallic character
a measure of how easily an element loses a valence electron; increases going down metal groups

26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments
Paper 1A: MCQ Practice
PDF
Paper 1B: Data & Experiments
PDF
Paper 2: Short Response
PDF
Official Markscheme
PDF
17
Cl
35.45
Assignments & Labs Section 08