Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Enthalpy (ΔH), Hess’s Law, Calorimetry, Entropy (ΔS), Gibbs Free Energy (ΔG)
Learning Objectives
Analyze energy changes in chemical systems through enthalpy, Hess's Law, entropy, and spontaneity.
Prior Knowledge Needed
Stoichiometry, bond enthalpy, exo/endothermic reactions
IB Syllabus Reference
R1.1, R1.2
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
3 Lessons Available
Calorimetry
Gibbs Free Energy_Born Haber Cycles
Hess Cycles
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
What is the enthalpy change of solution?
The enthalpy change when one mole of solute is dissolved in excess solvent to form a solution of "infinite dilution" under standard conditions. Measured in kilojoules per mole.
What is infinite dilution?
The solute particles are assumed not to interact with each-other, such that any further dilution produces no further enthalpy change.
What is average bond enthalpy?
The energy required to break one mole of a bond in a gaseous molecule, averaged over similar compounds.
What sign is H in oxygen radical formation from oxygen molecules?
positive: More bonds are broken than made.
What sign is H in ozone formation?
negative: More bonds are made than broken.
What sign is H in ozone depletion from just ozone?
positive: More bonds are broken than made.
What sign is H in ozone depletion from ozone reacting with an oxygen radical?
negative: More bonds are made than broken.
What is standard lattice enthalpy?
The enthalpy change when 1 mole an ionic solid separated into its gaseous ions under standard conditions. Measured in kilojoules per mole.
What is the standard atomization enthalpy?
The enthalpy change that occurs when one mole of a substance in its standard state is atomized completely into its individual gas-phase atoms, each in its ground state, under standard conditions. Measured in kilojoules per mole.
What is the enthalpy change of hydration?
The enthalpy change when one mole of gaseous ions dissolve in water.
What does a more negative enthalpy change of solution mean?
The more soluble a lattice is.
What factors affect hydration enthalpy?
1. Ionic radius 2. Ionic charge
What is the standard enthalpy change of reactants?
The enthalpy change when molar amounts of reactants react together under STP.
What equation links the enthalpy of solution with that of lattice and hydration?
E(sol) = E(lat) + E(hyd)
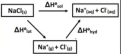
Enthalpy of formation
The enthalpy change when one mole of a substance is formed from its elements, with all substances in their standard states under standard conditions

Atomization enthalpy
The energy change to make 1 mole of gaseous atoms from its standard-state elements
Ionization Enthalpy
the energy required to remove an electron from one mole of atoms in their gaseous state
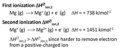
Lattice enthalpy of formation
Enthalpy change when one mole of a solid ionic compound is formed from its constituent ions in the gas phase
Lattice enthalpy of dissociation
Enthalpy change when one mole of a solid ionic compound is broken up into its constituent ions in the gas phase
electron affinity enthalpy
The enthalpy change when one mole of electrons is added to a mole of gaseous atoms under standard conditions
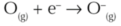
Bond enthalpy
The energy required to break one mole of a molecular bond in a gaseous state, under standard conditions.
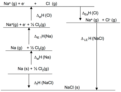
Born-Haber cycle
thermochemical cycle relating the various energetic steps involved in the formation of an ionic solid from the relevant elements
entropy
a measure of the disorder of a system

standard entropy change (of system)
the difference in absolute entropy between the reactants and products under standard conditions entropy of products - entropy of reactants
entropy change of surroundings
ΔSsurr = -ΔH/T
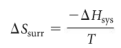
the energy of a system that is available to do work at a constant temperature and pressure
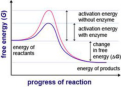
Gibbs free energy equation
ΔG = ΔH - TΔS

Change in Gibbs free energy of system
delta G= G (products)-G(reactants)

total entropy change
entropy change of system + entropy change of surroundings
enthalpy formation equation (from Born-Haber cycle)
delta Hf = sum of all other steps in Born-Haber cycle
Ozone Formation
O2 has double bond (bond energy 498) and UVC light, breaks to form two oxygen radicals, one oxygen radical reacts with O2 gas and forms O3 O3 is has dissociated electrons (364) and UVB then can make an oxygen gas and a radical
µ lattice enthalpy (ΔH lat)
Enthalpy change when one mole of a solid ionic compound breaks down to form gaseous ions under standard conditions.
The two factors that affect the lattice enthalpy are ionic charge and ionic radii.
As ionic charge increases or ionic radii decrease, the lattice enthalpy increases. Conversely, as ionic charge decreases or ionic radii increase, the lattice enthalpy decreases.
Enthalpy of atomisation
the enthalpy change when one mole of gaseous atoms is formed from an element in its standard state.
Bond dissociation energy
the energy required to break one mole of bonds in its gaseous state.
Ionisation energy
the energy required to remove one mole of electrons from one mole of gaseous atoms.
Electron affinity
the energy released when one mole of electrons are added to one mole of gaseous atoms.
The enthalpy of hydration
Enthalpy change when one mole of gaseous ions dissolves in water to form a solution of infinite dilution.
The enthalpy change of solution
Enthalpy change when one mole of solute dissolves to form a solution of infinite dilution.
INsoluble
Insoluble ionic compounds generally have a relatively high positive value for the enthalpy of solution
Soluble
Ionic substances that are soluble usually have a negative exothermic value for the enthalpy of solution
Entropy (S)
Refers to the distribution of energy among the particles in a system.
spontaneous
A spontaneous process occurs without the addition of energy, other than that required to overcome the initial energy barrier. A spontaneous process can be compared to a ball rolling down a hill; once the ball is given the initial push, it will continue to roll down the hill until it reaches the bottom.
The Gibbs free energy
Energy associated with a chemical reaction that can be used to do work.
ΔS
change in entropy of a system; J K−1 mol−1.
The Gibbs free energy of formation
Change in free energy when one mole of a compound is formed from its elements in their standard states under standard conditions.
The standard Gibbs free energy change
Change in Gibbs free energy for a reaction at a temperature of 298 K.
What are the signs of H and S for a reaction that is non-spontaneous at low temperatures but spontaneous at high temperatures?
positive ΔH and positive ΔS
26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments (HL)
Paper 1A: MCQ Practice (HL)
PDF
Paper 1B: Data & Experiments (HL)
PDF
Paper 2: Short Response (HL)
PDF
Official Markscheme (HL)
PDF
17
Cl
35.45
Assignments & Labs Section 08