Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
States of Matter, Kinetic Molecular Theory, Phase Transitions, Pure Substances vs Mixtures
Learning Objectives
Understand the three states of matter and the fundamental particles that compose all chemical substances.
Prior Knowledge Needed
Particle model of matter, basic heating/cooling curves
IB Syllabus Reference
S1.1
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
2 Lessons Available
Atomic Models_Phase Diagrams
Particulate Nature of Matter_Isotopes
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
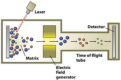
mass spectrometry
technique based of the detection of vaporized molecules and their ionized (charged) frag- ments in an instrument called a mass spectrometer; detection and display of the spectra are based on the mass-to-mass charge ratios of the ions; an experimental method of determining the precise mass and relative abundance of isotopes in a given sample
relative atomic mass
The weighted average mass of an atom of an element compared with one-twelfth of the mass of an atom of carbon-12; denoted by Ar
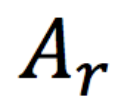
weighted average
Average of data that takes other factors such as the percent abundance into account.
percent abundance
The percentage of atoms of a particular isotope in a natural sample of a pure element

relative isotope intensity
the area under the curve of the peak signal for each isotope in a mass spectrum (relative to the largest signal produced)

isotopic abundance
the percent or fraction of each isotope found in nature

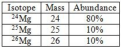
isotope
Atoms of the same element that have different numbers of neutrons
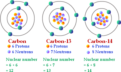
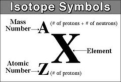
nuclear symbol
the superscript indicates the mass number and the subscript indicates the atomic number
electron orbit
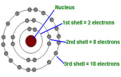
the path of an electron around the nucleus of an atom (Bohr/Rutherford model)
electron orbital
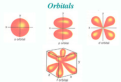
how electrons are spatially distributed surrounding the nucleus; the area where an electron is most likely to be found
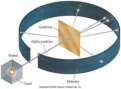
Gold foil experiment
Conducted by Ernest Rutherford in which alpha particles that were shot at gold foil were deflected when they hit the positive center of gold atoms. The nucleus was discovered as a result of this experiment.
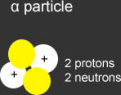
alpha particle
a type of nuclear radiation consisting of two protons and two neutrons
English chemist and physicist who formulated atomic theory and the law of partial pressures

cathode ray tube
evacuated glass tube in which a stream of electrons emitted by a cathode strikes a fluorescent material, causing it to glow
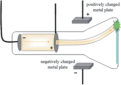
JJ Thomson
used the cathode ray tube to discover electrons; responsible for plum pudding model
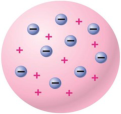
plum pudding model
J.J Thomsons model of an atom, in which he thought electrons were randomly distributed within a positively charged cloud
Bohr/Rutherford model
A model of the atom in which a central positive nucleus is surrounded by the electrons energy levels
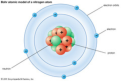
Quantum mechanical model
the modern description, primarily mathematical, of the behavior of electrons in atoms; de- scribes probability of electrons within orbitals
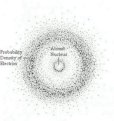
-said that electrons are in energy levels, the further an electron was from the nucleus, the higher its energy -developed the planetary model (built on Rutherford's work)

Ernest Rutherford
gold foil experiment- fired alpha particles at thin sheet of gold foil, discovered the atomic nucleus and proposed a nuclear model of the atom .

Heisenberg uncertainty principle
it is impossible to know exactly both the velocity and the position of a particle at the same time
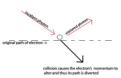
Schrodinger equation
An equation that describes how the electron matter-wave changes in space around the nucleus. Solutions of the equation provide allowable energy levels of the atom.

James Chadwick
Discovered the neutron in 1932

Properties of a Solid
- Particles are arranged in a lattice - Fixed shape - Fixed volume - Cannot be compressed - Have the least amount of energy
Properties of a Liquid
- Takes the shape of its container (not fixed shape) - Can flow - Cannot be compressed - Fixed volume - Middle amount of energy
Properties of a Gas
- Fill its container - Can flow - Particles are far apart - Can be compressed - Most amount of energy
Structure of a Solid
- Particles vibrate in fixed positions - Particles are very close
Structure of a Liquid
- Particles flow over each other - Particles are fairly close
Structure of a Gas
- Particles move in straight lines and collide with each other - Particles are as far apart as they can be
Melting
SOLID TO LIQUID
Boiling
LIQUID TO GAS
Evaporating
LIQUID TO GAS at surface of substance
Sublimation
SOLID TO GAS
Deposition
GAS TO SOLID
Freezing
LIQUID TO SOLID
Condensing
GAS TO LIQUID
Heating / Cooling Curves
Flat lines are increasing/decresing Potential Energy Slanted lines "temp Change" is Kinetic Energy
Kinetic Theory
A theory explaining the states of matter, based on the concept that all matter consists of tiny particles that are in constant motion
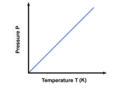
Pressure and Temperature of Gases
- When the temperature of gas increases, the pressure increases (directly proportional) - Heat causes the particles to move faster - Particles hit walls more, ergo, higher pressure
Brownian Motion
- The particles of fluids move randomly - This is because they are bombarded by other particles randomly - Larger particles can be moved because of this
- When particles spread from an area of high concentration to low concentration - Moleules with a higher Mr diffuse slower as they move slower with the same energy
Evidence for Brownian Motion
- Small particles in water - Smoke particles
Atom
Smallest unit of matter that retains the properties of an element
Element
A pure substance made of only one kind of atom
Molecule
a group of atoms bonded together, representing the smallest fundamental unit of a chemical compound that can take part in a chemical reaction.
Compound
A substance made up of atoms of two or more different elements joined by chemical bonds
Mixture
A combination of two or more substances that are not chemically combined
Solution
A mixture that forms when one substance dissolves another.
Solute
A substance that is dissolved in a solution.
Solvent
A liquid substance capable of dissolving other substances
states of matter
solid, liquid, gas, plasma
Plasma
a state of matter that consists of free-moving ions and electrons
boiling point
temperature at which a liquid changes to a gas at a given pressure
melting point
The temperature at which a solid becomes a liquid
expansion
An increase in volume.
contraction
a decrease in volume
26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments
Paper 1A: MCQ Practice
PDF
Paper 1B: Data & Experiments
PDF
Paper 2: Short Response
PDF
Official Markscheme
PDF
17
Cl
35.45
Assignments & Labs Section 08