Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Mass Spectrometry, Subshells (s, p, d, f), Aufbau Principle, Hund's Rule, Pauli Exclusion Principle
Learning Objectives
Explore the internal architecture of the atom, including emission spectra, electron configurations, and ionization energies.
Prior Knowledge Needed
Bohr model, protons/neutrons/electrons, isotopes (Theme 3)
IB Syllabus Reference
S1.3
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
1 Lessons Available
Electron Configuration
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
Planck Equation
E=hf (E=energy, f=frequency, h=Planck's Constant)
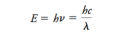
Convergence Limit
the point in a line emission spectrum where the lines merge to form a continuum; may be used to determine the ionization energy.

First Ionization Energy
the energy required to remove the first electron from a neutral atom
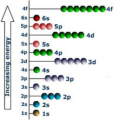
Sublevel
An atomic orbital, or collection of atomic orbitals, that occupy a principal energy level and are called s, p, d, and f. Ionization energy "jumps" for an element give evidence for the existence of sublevels.
Planck's constant
a number used to calculate the radiant energy (E) absorbed or emitted by a body based on the frequency of radiation (6.63 x 10^-34 Js)
Equation for Ionization Energy
X(g) -> X+(g) + e-

26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments
Paper 1A: MCQ Practice
PDF
Paper 1B: Data & Experiments
PDF
Paper 2: Short Response
PDF
Official Markscheme
PDF
17
Cl
35.45
Assignments & Labs Section 08