Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Oxidation Numbers, Winkler Method, Voltaic Cells, Electrolysis, Standard Electrode Potentials
Learning Objectives
Understand electron transfer through oxidation numbers, activity series, and electrochemical cells.
Prior Knowledge Needed
Ionic charge, ionic equations, electronegativity (S2.1)
IB Syllabus Reference
R2.1
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
3 Lessons Available
Electrolytic Cells
Redox Titrations
Voltaic Cells_Calculating Cell Potentials
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
redox reaction
A chemical reaction involving the transfer of one or more electrons from one reactant to another; also called oxidation-reduction reaction.
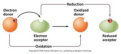
disproportionation reaction
a reaction in which an element in one oxidation state is both oxidized and reduced
oxidation
loss of electrons; results in an increased oxidation state
reduction
gain of electrons; results in a decreased oxidation state
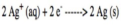
oxidation state
A positive or negative whole number that represents the "charge" an atom in a compound would have if all shared electrons were assigned to the atom with a greater attraction for those electrons.
oxidation number
Positive or negative number that indicates how many electrons an atom has gained, lost, or shared to become stable; used to represent charges of transition metals

oxidizing agent
Accepts electrons and becomes reduced.
Donates electrons and becomes oxidized.
half reaction
an equation showing either the oxidation or the reduction that takes place in a redox reaction
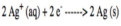
LEO the lion says GER
Loss of electrons is oxidation, gain of electrons is reduction.

Oxidation Rules: Oxygen
Oxygen has an oxidation state of -2 unless it is combined with F (when it is +2), or it is in a peroxide (such as H2O2 or Na2O2), when it is -1.

oxidation rules: groups 1 and 2
In compounds, the elements of groups 1 and 2 as well as aluminum have oxidation states of +1, +2, and +3 respectively.

Oxidation Rules: Hydrogen
The oxidation state of hydrogen in most of its compounds is +1 unless it is combined with a metal, in which case it is -1.

Oxidation Rules: Fluorine
The oxidation state of fluorine in a compound is always -1.
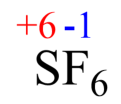
The oxidation state of any uncombined element is 0.

Oxidation Rules: Monoatomic Ions
The oxidation state of a monatomic ion equals the charge on the ion.
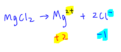
Oxidation Rules: Polyatomic Ions
The sum of the oxidation states of all atoms in a polyatomic ion equals the charge of the ion.

Oxidation Rules: Binary Compounds
The more-electronegative element in a binary compound is assigned the number equal to the charge it would have if it were an ion.

Oxidation Rules: Neutral Compounds
The sum of the oxidation states of all atoms in a neutral compound is 0.
permanganate
MnO4-; strong oxidizing agent
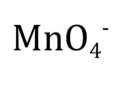
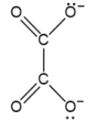
ethanedioate
C₂O₄²⁻, reducing agent
dichromate
Cr2O7^2-; strong oxidizing agent
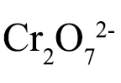
iodine-thiosulfate reaction
reaction in which iodine is titrated with sodium thiosulfate to reduce iodine to iodide (used in many redox titrations).
Winkler method
used to measure Biological Oxygen Demand (a measure of the dissolved oxygen required to decompose organic matter in water over a set time period)

Biological Oxygen Demand (BOD)
amount of dissolved oxygen needed by aerobic decomposers to break down organic mate- rials, usually over a specified period of time

Oxygen fixation
a manganese compound is used to fix dissolved oxygen so it can be measured utilizing an iodine titration.
an electrochemical cell that produces electrical current from a spontaneous chemical reaction
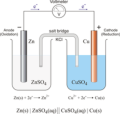
anode (voltaic cell)
the electrode at which oxidation occurs; negative electrode

cathode (voltaic cell)
the electrode at which reduction occurs; positive electrode

half-cell
one part of a voltaic cell in which either oxidation or reduction occurs

electrode
A metal part of an electrochemical cell, which gains or loses electrons.

electromotive force
a difference in potential that tends to give rise to an electric current.
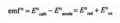
salt bridge
a pathway constructed to allow the passage of ions from one side of a voltaic cell to another
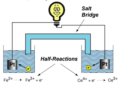
standard reduction potential
The tendency of a species to be reduced, as measured at 25 C when reacting species are of 1 mol dm^-3 concentration and 101 kPa
cell diagram convention
anode left, phase boundary by vertical line, salt bridge by double vertical line. Example: Cu(s)/Cu2+(aq) || H+(aq) / H2(g)
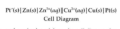
26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments
Paper 1A: MCQ Practice
PDF
Paper 1B: Data & Experiments
PDF
Paper 2: Short Response
PDF
Official Markscheme
PDF
17
Cl
35.45
Assignments & Labs Section 08