Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Equilibrium Constant (Kc), Reaction Quotient (Q), Le Chatelier’s Principle, Haber/Contact Processes
Learning Objectives
Analyze reversible systems and the factors that influence the position of chemical equilibrium.
Prior Knowledge Needed
Kinetics, enthalpy changes, gas volume calculations
IB Syllabus Reference
R2.3
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
6 Lessons Available
3 Equilibrium Exam Version A edited
3 Equilibrium Test Version B Edited
Equilibrium Graphs_Factors that affect Equilibrium
ICE Tables
Relationship between Gibbs free energy and Equilibrium Constant
The Equilibrium Constant and Reaction Quotient
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
equilibrium constant
the ratio of product concentrations to reactant concentrations at equilibrium, with each concentration raised to a power equal to the number of moles of that substance in the balanced chemical equation. Commonly denoted by Kc, Kp, or Ksp
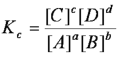
reaction quotient
Ratio of the concentrations of the products to the concentrations of the reactants at any point during the reaction aside from equilibrium, where each reactant and product in the expression is raised to the power of its stoichiometric coefficient. Commonly denoted by Q.
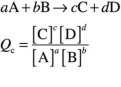
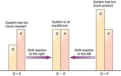
Q = Kc
reaction is at equilibrium
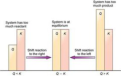
Q>Kc
reaction proceeds left in favor of reactants
Q<Kc
reaction proceeds right in favor of products
heterogeneous equilibrium
a state of equilibrium that occurs when the reactants and products of a reaction are present in more than one physical state

homogeneous equilibrium
all the reactants and products are in the same physical state

Haber process
an industrial process for producing ammonia from nitrogen and hydrogen by combining them under high pressure in the present of an iron catalyst
contact process
industrial method of preparing sulfuric acid by the catalytic oxidation of sulfur dioxide

methanol production
Ì CO (g) + 2H2(g) CH3OH (g Used as starting materials for other chemicals + fuels Lower temp (227C)= favours exo reaction therefore higher yield Higher pressure (10000KPa)= favours forwards reaction as this decreases number of gaseous molecules BUT too high = too expensive catalyst= chromium

dynamic equilibrium
condition of continuous, random movement of particles but no overall change in concentration of materials

Le Chatelier's Principle
when stress is applied to a system at equilibrium, the system changes to relieve the stress

concentration
factor that affects equilibrium by the reaction shifting in the direction to restore the change in concentration of reactant/product
exothermic reaction
for a system at equilibrium, increasing temperature will shift the reaction to the left.
endothermic reaction
for a system at equilibrium, increasing temperature will shift the reaction to the right.
closed system
A system in which no matter is allowed to enter or leave
pressure
for a gaseous system at equilibrium, an increase in pressure will cause the system will shift to the side with less moles of gas.
catalyst
increases the rates of both the forward and the reverse reactions equally
26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments
Paper 1A: MCQ Practice
PDF
Paper 1B: Data & Experiments
PDF
Paper 2: Short Response
PDF
Official Markscheme
PDF
17
Cl
35.45
Assignments & Labs Section 08