Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
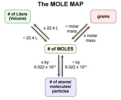
The Mole, Limiting Reactants, Molar Volume, Theoretical/Percentage Yield, Titration Calculations
Learning Objectives
Perform quantitative calculations involving the mole, ideal gas laws, and stoichiometric relationships in reactions.
Prior Knowledge Needed
Balancing equations, relative atomic mass (S1.1)
IB Syllabus Reference
S1.1, R1.1
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
4 Lessons Available
Amount of Chemical Change
Atom Economy and Empirical Formula of Hydrate
Gas Laws
Stoichiometry
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
Avogadro's Law
equal volumes of gases at the same temperature and pressure contain equal numbers of molecules; V1/n1 = V2/n2

Molar volume of a gas
The volume occupied by one mole of the gas. It is equal to 22.7 litres at STP.
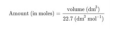
Molar gas constant
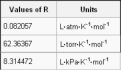
the constant in the equation of state of an ideal gas - symbol R, 8.31 m^3 Pa K^-1 mol^-1
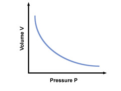
Boyle's Law
A principle that describes the relationship between the pressure and volume of a gas at constant temperature; P1V1 = P2V2
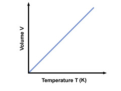
Charles' Law
the law that states that for a fixed amount of gas at a constant pressure, the volume of the gas increases as the temperature of the gas increases and the volume of the gas decreases as the temperature of the gas decreases; V1/T1=V2/T2
Gay-Lussac's Law
the pressure of a gas is directly proportional to the Kelvin temperature if the volume is constant; P1/T1=P2/T2
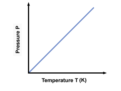
Combined gas law
shows the relationship between the pressure, volume, and temperature of a fixed amount of gas; P1V1/T1=P2V2/T2
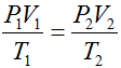
Ideal Gas Law
the relationship PV=nRT, which describes the behavior of an ideal gas

STP
standard temperature and pressure (273 K and 100 kPa/1 atm)
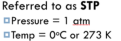
SATP
standard ambient temperature and pressure; exactly 25 degrees Celsius and 100 kPa/1 atm

Kelvin scale
the temperature scale in which the freezing point of water is 273 K and the boiling point is 373 K; 0 K is absolute zero

Celsius scale
a temperature scale that defines the freezing point of water as 0 degrees and the boiling point of water as 100 degrees

stoichiometry
the relationship between the relative quantities of substances taking part in a reaction or forming a compound, typically a ratio of whole integers.
limiting reactant
the substance that controls the quantity of product that can form in a chemical reaction
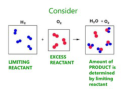
excess reactant
the substance that is not used up completely in a reaction

ionic equations
used to show details of reactions that involve ions in an aqueous solution

relative atomic mass
The weighted mean mass of an atom of an element compared with one-twelfth of the mass of an atom of carbon-12.
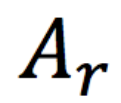
theoretical yield
the maximum amount of product that can be produced from a given amount of reactant

actual yield
the measured amount of a product obtained from a reaction

percent yield
the ratio of the actual yield to the theoretical yield expressed as a percent

atom economy
(Molecular mass of desired products/ Molecular mass of all products) x 100

percent composition
the percent by mass of each element in a compound

empirical formula
a formula with the lowest whole-number ratio of elements in a compound
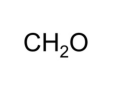
molecular formula
shows the types and numbers of atoms combined in a single molecule of a molecular compound
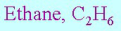
anhydrate

hydrate
compound that has water chemically attached to its ions in a specific ratio and written into its chemical formula
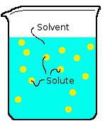
solute
A substance that is dissolved in a solution.
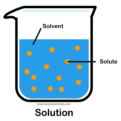
solvent
In a solution, the substance in which the solute dissolves.
concentration
A measurement of how much solute exists within a certain volume of solution, usually given in mol per cubic decimeter

M1V1=M2V2
Formula used to determine how to create a solution of desired concentration from a stock solution of known molarity and volume; 1 indicates the initial concentration and 2 indicates the final concentration

primary standard solution
A solution prepared from a solid that: - Is water-soluble. - Has a high purity. - Has a high molar mass. - Is stable in air.

Volumetric analysis
a technique for determining the concentration of a certain substance by doing a titration
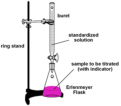
titration
a measured amount of a solution of unknown concentration is added to a known volume of a second solution until the reaction between them is complete

neutralization reaction
a reaction in which an acid and a base react in an aqueous solution to produce a salt and water
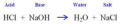
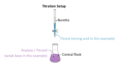
titrant
A solution of known concentration that is used to titrate a solution of unknown concentration

analyte
solution of unknown concentration in a titration
parts per million (ppm)
a method of expressing low concentrations; 1 ppm is equivalent to 1 milligram per liter.
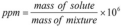
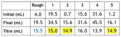
titre

concordant titre
A set of titres that vary within a narrow range, e.g. within 0.20 mL from smallest to largest titre.
26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments
Paper 1A: MCQ Practice
PDF
Paper 1B: Data & Experiments
PDF
Paper 2: Short Response
PDF
Official Markscheme
PDF
17
Cl
35.45
Assignments & Labs Section 08