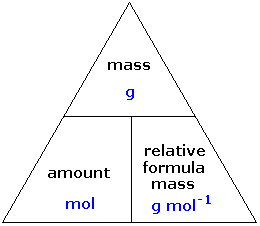Preparing Standard Solutions
To prepare a standard solution, which is a solution with an accurately known concentration, the process begins with precisely measuring the mass of the solute using a balance that can measure to at least two decimal places. This ensures high accuracy in the initial quantity of the substance. The measured solute is then dissolved in a small volume of distilled water within a beaker, using a stirring rod to facilitate dissolution. Once dissolved, the solution is carefully transferred to a volumetric flask using a funnel. To ensure that all of the solute is transferred and none is left behind, the beaker, stirring rod, and funnel are thoroughly rinsed with distilled water, and these rinsings are also poured into the volumetric flask. Finally, distilled water is added to the volumetric flask until the solution reaches the required volume mark. A stopper is then placed on the flask, and it is inverted several times to ensure the solution is thoroughly mixed and homogeneous.
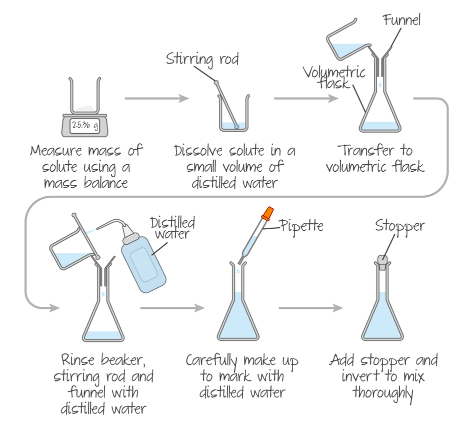
Detailed Procedure for Solution Preparation
The preparation of a solution involves several critical stages to ensure accuracy. The first stage involves transferring a known mass of the solid solute. This is achieved by weighing the solid into a weighing boat and recording its mass to an appropriate precision. The solid is then transferred to a beaker. To account for any solid residue left in the weighing boat, it is re-weighed, and the difference in mass is calculated. Alternatively, the weighing boat can be rinsed directly into the beaker to ensure all solute is transferred.
The second stage focuses on dissolving the solid in distilled water. The minimum amount of distilled water required to dissolve the solid is added to the beaker. The mixture is then stirred with a glass rod until the solid has completely dissolved. It is crucial not to add too much distilled water at this point, as it might leave insufficient volume for rinsing glassware later without overshooting the final desired volume of the solution.
The third and final stage involves rinsing all glassware and making up the solution to its final volume. The dissolved solution is transferred to a volumetric flask, typically 250 cm
3, using a funnel. All glassware that has been in contact with the solution, including the glass rod, beaker, and funnel, must be thoroughly rinsed with distilled water, and these rinsings are added to the volumetric flask. This step is vital to ensure that every particle of solute makes its way into the volumetric flask. Distilled water is then added up to the graduation line on the volumetric flask. After reaching the mark, the flask is stoppered and inverted several times to ensure the final solution is fully mixed and homogeneous before use. This mixing step should only be performed after the solution has been made up to the graduation mark.
Characteristics of Primary Standard Solutions
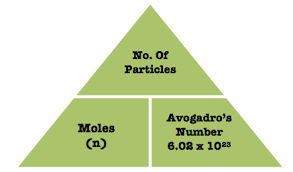
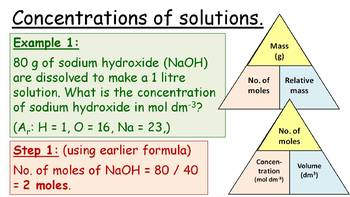
A standard solution is defined as a solution with an accurately known concentration. A primary standard solution is specifically prepared using a primary standard, which is a substance possessing several key properties that make it suitable for this purpose. These properties include high purity, typically 99.9% or greater, a high molar mass to minimize the impact of weighing errors, and low reactivity to ensure stability. Furthermore, a primary standard should not change its composition when exposed to air, preventing reactions with atmospheric components like carbon dioxide or water vapor. Examples of substances commonly used as primary standards include sodium carbonate (Na
2CO
3), oxalic acid (H
2C
2O
4), potassium hydrogen iodate (K(HIO
3)
2), and potassium dichromate (K
2Cr
2O
7).
Understanding Secondary Standard Solutions
In contrast to primary standard solutions, a secondary standard solution is one whose concentration has been determined by standardizing it against a primary standard solution. For instance, a sodium hydroxide solution, which is hygroscopic and reacts with atmospheric carbon dioxide, cannot be prepared directly as a primary standard. Instead, its concentration is accurately determined by titrating it against a primary standard acid, such as oxalic acid. Once standardized, the sodium hydroxide solution can then be used as a secondary standard solution. The use of both primary and secondary standard solutions with accurately known concentrations is fundamental in volumetric analysis, particularly in techniques like titration.
Introduction to Volumetric Analysis
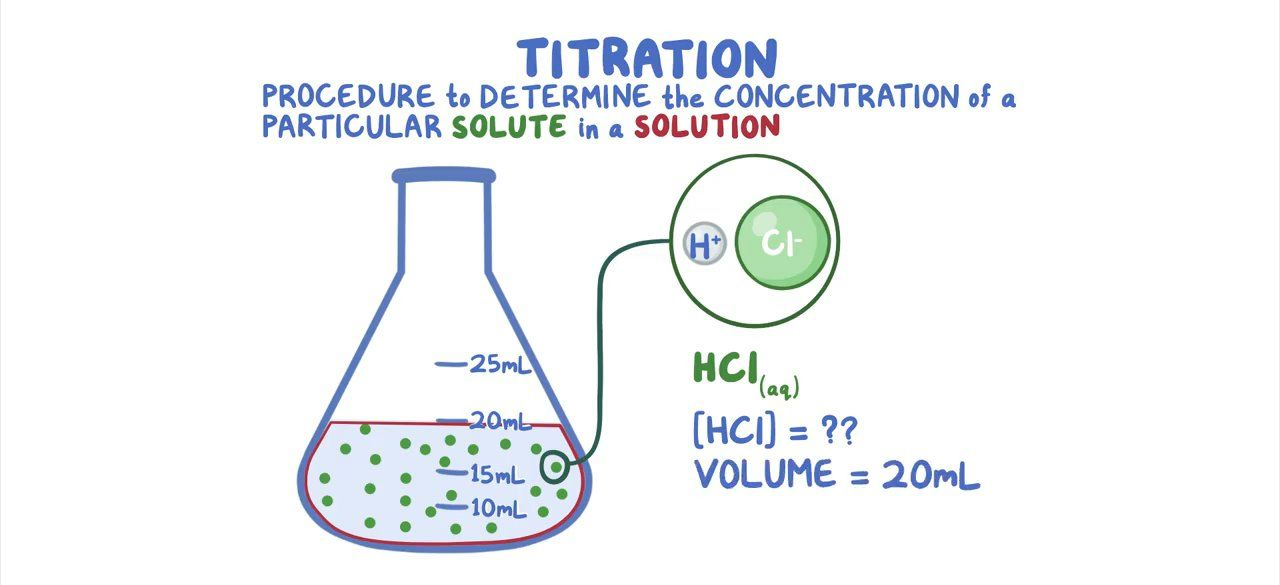
Volumetric analysis is a quantitative analytical method that relies on measuring the volume of a solution of known concentration required to react completely with a solution of unknown concentration. This technique is crucial for determining the concentration of an unknown substance.
The Titration Technique
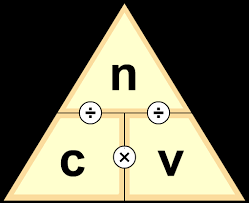
Titration is a specific technique within volumetric analysis used to calculate the unknown concentration (in mol dm
-3) of a solution. This is most commonly achieved through a neutralization reaction between an acid and an alkali. During a titration, a solution of known concentration (the titrant) is gradually added to a solution of unknown concentration (the analyte) until the reaction is complete, indicated by a color change from an appropriate indicator.
The Concept of Neutralization
Neutralization is a chemical reaction that occurs when an acid
and a base (or an alkali, which is a soluble base) react to form a
neutral solution, typically consisting of a salt and water. This
reaction is fundamental to many titration experiments, particularly
acid-base titrations.
Acid-Base Titration Principles
Acid-base titrations are a common application of neutralization
reactions to determine unknown concentrations.
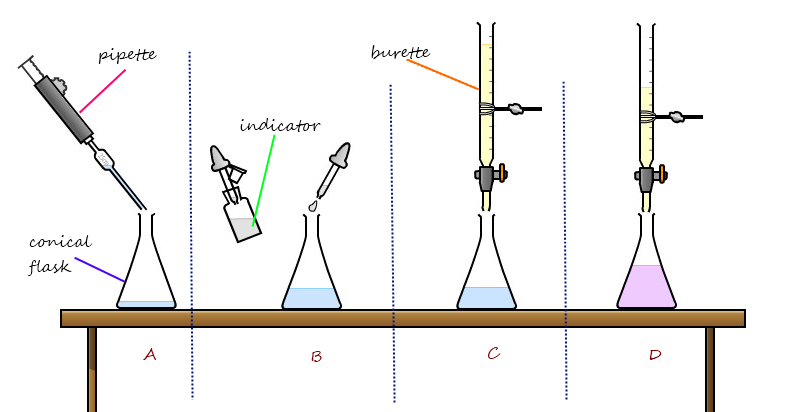
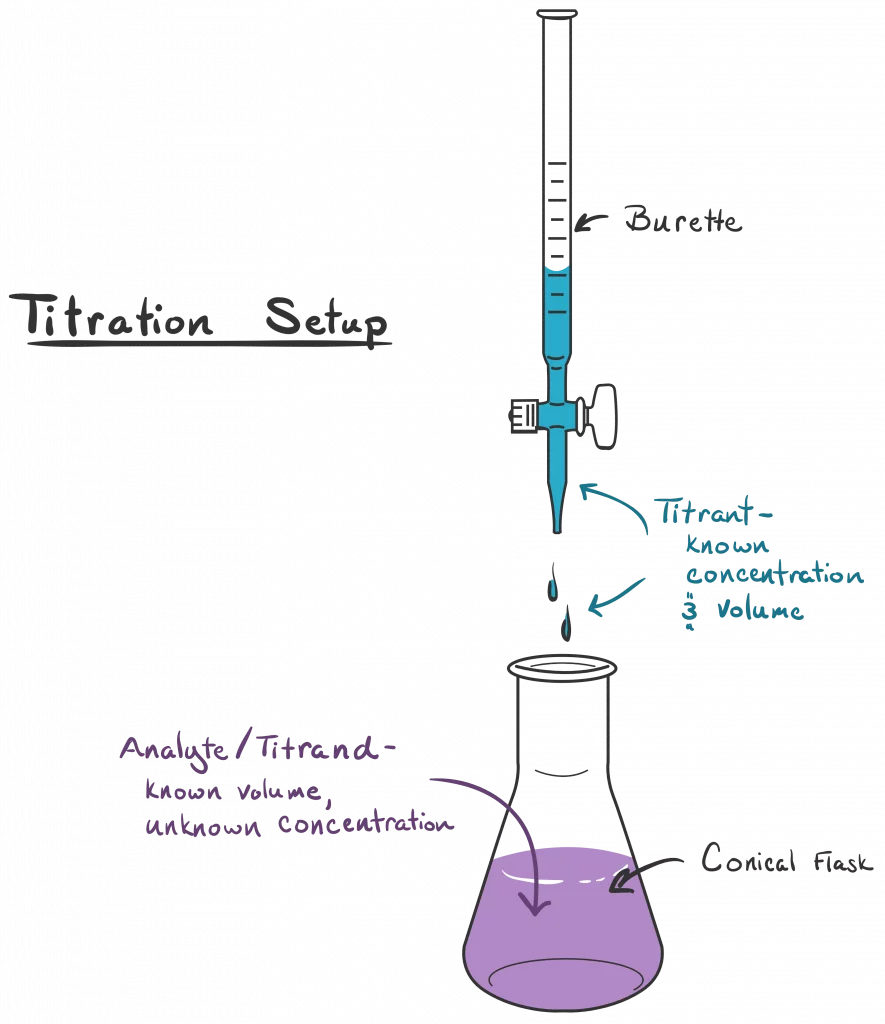
Proper Use of Pipettes and Burettes in Titration
Accurate titration relies on the correct use of specialized glassware, specifically pipettes and burettes. The solution of known concentration, referred to as the titrant, is typically placed in the burette, while the unknown solution, known as the analyte, is placed in the conical flask. It is crucial to ensure that the top of the burette is at eye level or below when filling to avoid errors in reading the volume. The titration proceeds by adding the titrant from the burette to the analyte in the flask until the endpoint is reached, which is indicated by a distinct color change of an added indicator.
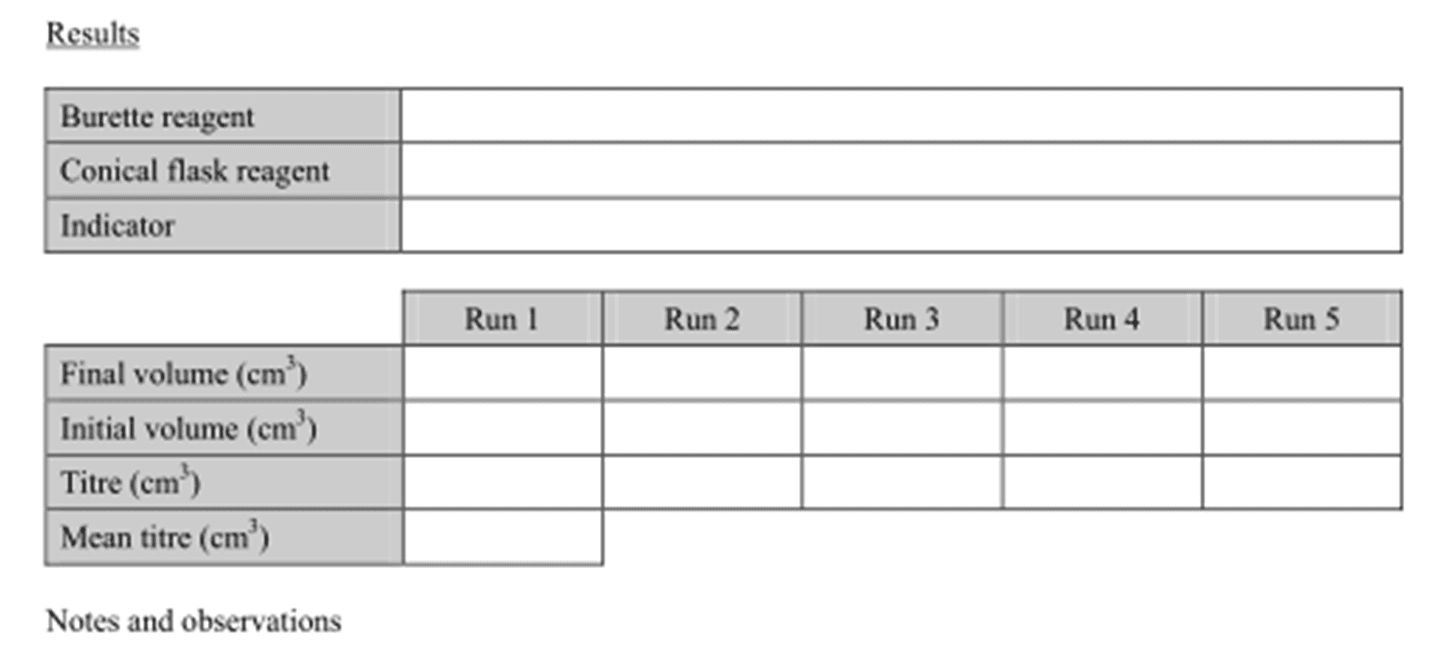
Recording Titration Results
All titration readings, including initial and final volumes, must be recorded to two decimal places to maintain precision. For example, readings should be recorded as 26.30 cm
3, 26.35 cm
3, or 26.40 cm
3.
Step-by-Step Titration Procedure
Performing a titration involves several distinct stages to ensure accurate results.
Stage 1: Preparing the Conical Flask
First, a solution with an unknown concentration is accurately transferred into a conical flask using a glass pipette. Subsequently, several drops of an appropriate indicator are added to the conical flask. Any solution adhering to the sides of the flask is rinsed down into the main body of the solution with distilled water.
Stage 2: Preparing the Burette
The burette is filled with the solution of known concentration (the titrant) using a funnel. Before starting the titration, a small amount of the solution is run through the burette into a waste container to ensure that there are no air bubbles present in the tip and that the burette walls are conditioned with the solution. The funnel is then removed from the burette.
Stage 3: Performing the Titration
The conical flask is placed on a white piece of paper directly beneath the burette to clearly observe any color changes. The starting volume of the burette is recorded, which is often 0.00 cm
3. The titrant is then slowly added from the burette to the conical flask, while continuously swirling the flask, until a permanent color change is observed, indicating the endpoint of the reaction. The final volume in the burette is recorded, and the titre (the volume of solution required to reach the endpoint) is calculated. This entire process is repeated until concordant results are achieved, typically within 0.20 cm
3 for at least four runs. A mean titre is then calculated from these concordant results.
Calculating Unknown Concentrations from Titration Data
To calculate the unknown concentration of a solution from titration data, a systematic approach is followed. First, it is helpful to sketch the experimental setup, noting all known volumes and concentrations. A balanced chemical equation for the reaction is then written. The next step involves calculating the moles of the solution whose concentration was known (the titrant). Using the stoichiometry from the balanced chemical equation, the moles of the reacting substance in the unknown solution (the analyte) are determined. Finally, using the calculated moles and the known volume of the unknown substance, its concentration can be determined.
Example Calculation of Unknown Concentration
Consider the neutralization reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH):
HCl (aq) + NaOH (aq) → NaCl (aq) + H
2O (l)
In an experiment, 100 cm
3 of NaOH solution with an unknown concentration is titrated with 0.120 mol dm
-3 HCl. The titration results are recorded in the table below:
| Volume /± 0.05 cm3 |
Trial 1 |
Trial 2 |
Trial 3 |
| Initial Volume |
22.10 |
42.90 |
22.80 |
| Final Volume |
1.20 |
22.15 |
2.00 |
| Titre |
20.90 |
20.75 |
20.80 |
To calculate the mean titre, we consider the concordant results. Let's assume the concordant results are Trial 2 and Trial 3, which are 20.75 cm
3 and 20.80 cm
3 respectively. The mean titre would be (20.75 + 20.80) / 2 = 20.775 cm
3. For calculation purposes, we will use the provided mean titre from the next table.
|
Trial 1 |
Trial 2 |
Trial 3 |
| Initial Volume /± 0.05 cm3 |
22.10 |
42.90 |
22.80 |
| Final Volume /± 0.05 cm3 |
1.20 |
22.15 |
2.00 |
| Titre /± 0.10 cm3 |
20.90 |
20.75 |
20.80 |
| Mean titre /± 0.10 cm3 |
20.82 |
|
|
Using the mean titre of 20.82 cm
3 (or 0.02082 dm
3), we can calculate the moles of the titrant (HCl):
Moles of HCl = Concentration × Volume = 0.120 mol dm
-3 × 0.02082 dm
3 = 0.0024984 mol HCl.
From the balanced chemical equation, 1 mole of HCl reacts with 1 mole of NaOH. Therefore, the moles of NaOH that reacted in the flask are also 0.0024984 mol.
The original volume of NaOH in the flask was 100 cm
3 (or 0.100 dm
3). Now, we can calculate the concentration of NaOH:
Concentration of NaOH = Moles / Volume = 0.0024984 mol / 0.100 dm
3 = 0.024984 mol dm
-3.
Rounding to an appropriate number of significant figures, the concentration of NaOH is approximately 0.0250 mol dm
-3.
Propagation of Uncertainty in Titration Calculations
When performing calculations involving experimental data, it is crucial to propagate uncertainties to determine the overall uncertainty in the final result.
For the reaction HCl (aq) + NaOH (aq) → NaCl (aq) + H
2O (l), let's assume the uncertainty of the HCl concentration was ±0.002 mol dm
-3 and the uncertainty of the volume of titrant (mean titre) was ±0.10 cm
3 (or ±0.0001 dm
3). When quantities are multiplied, their percentage uncertainties are added.
First, calculate the percentage uncertainty for the HCl concentration:
% uncertainty HCl concentration = (0.002 / 0.120) × 100% = 1.667%
Next, calculate the percentage uncertainty for the volume of HCl:
% uncertainty volume HCl = (0.0001 / 0.02082) × 100% = 0.4803%
Adding these percentage uncertainties gives the total percentage uncertainty for the moles of HCl, which is also the percentage uncertainty for the moles of NaOH due to the 1:1 stoichiometric ratio:
Total % uncertainty = 1.667% + 0.4803% = 2.1473% ≈ 2.15%
Further Propagation of Uncertainty for Final Concentration
To calculate the uncertainty of the final concentration of NaOH, the percentage uncertainties of the moles of NaOH and the volume of NaOH must be added.
Assuming the absolute uncertainty of the volume of NaOH (100 cm
3) was ±0.05 cm
3 (or ±0.00005 dm
3):
Calculate the percentage uncertainty for the volume of NaOH:
% uncertainty volume NaOH = (0.00005 / 0.100) × 100% = 0.05%
Now, add the percentage uncertainty for the moles of NaOH (calculated previously as 2.15%) and the percentage uncertainty for the volume of NaOH:
Total % uncertainty for NaOH concentration = 2.15% + 0.05% = 2.20%
Finally, convert this percentage uncertainty back to an absolute uncertainty for the NaOH concentration:
Absolute uncertainty = 0.0250 mol dm
-3 × (2.20 / 100) = 0.00055 mol dm
-3.
Therefore, the concentration of NaOH can be reported as 0.0250 mol dm
-3 ± 0.0006 mol dm
-3 (rounded to one significant figure for the uncertainty).
Standard Titration Results Table Format
When recording titration results, it is essential to use a consistent and clear format. All titration readings, including initial and final volumes, should always be recorded to two decimal places, for example, 26.30 cm
3, 26.35 cm
3, or 26.40 cm
3. A standard table format helps organize the data effectively.