Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Ionic Lattice, VSEPR Theory, Resonance, Bond Enthalpy, Polar vs Non-polar Bonds
Learning Objectives
Investigate the forces that hold matter together, from ionic and covalent lattices to metallic bonding models.
Prior Knowledge Needed
Valence electrons, Lewis structures, electronegativity (S3.1)
IB Syllabus Reference
S2.1, S2.2
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
6 Lessons Available
Covalent Bonding and Properties of Covalent Compounds
IB Chem 1 Paper Chromatography
Intermolecular Forces
Ionic Bonding and Properties of Ionic Compounds
Lewis Structures_VSEPR Theory_Metallic Bonding and Alloys
Resonance Structures_Formal Charge_Hybridization
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
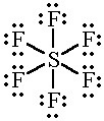
expanded octet
when a molecule has more than 8 electrons around the central atom
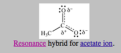
resonance hybrid
the actual structure of a molecule that is intermediate between two or more resonance structures
formal charge
The number of valence electrons in an isolated atom minus the number of electrons assigned to the atom in the Lewis structure
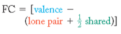
non-equivalent Lewis structure
Lewis structures that contain a different number of single and double bonds, resulting in different electron arrangements and formal charges
bond order
the number of bonds between atoms: 1 for a single bond, 2 for a double bond, and 3 for a triple bond

delocalized electrons
the electrons involved in a molecule that are free to move easily from one atom to the next and are not attached to a particular atom
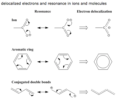
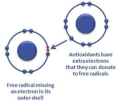
free radical
an atom or a group of atoms that has one unpaired electron
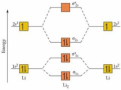
molecular orbital
Region in a molecule where atomic orbitals overlap, resulting in either a stable low-energy bonding orbital or an unstable high-energy antibonding orbital.
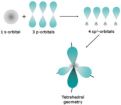
orbital hybridization
orbitals involved in covalent bonding in an atom are hybridized so that they are identical in properties, and those properties are intermediate to the properties of the original orbitals
sigma bond
a bond formed when two atomic orbitals combine to form a molecular orbital that is sym- metrical around the axis connecting the two atomic nuclei
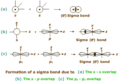
pi bond
a bond that is formed when parallel orbitals overlap to share electrons.

A type of hybridization that results from the combination of the s orbital and all three p orbitals in the second energy level of carbon, resulting in four hybrid orbitals and occurs when a carbon atom is bonded to four other atoms. The geometric arrangement of those four hybrid orbitals is called tetrahedral.

sp2 hybridization
A type of hybridization that results from the combination of the s orbital and two p orbitals in the second energy level of carbon, resulting in three hybrid orbitals and occurs when a carbon atom is bonded to three other atoms. The geometric arrangement of those three hybrid orbitals is called trigonal planar.

sp hybridization
A type of hybridization that results from the combination of the s orbital and one p orbital in the second energy level of carbon, resulting in two hybrid orbitals and occurs when a carbon atom is bonded to two other atoms. The geometric arrangement of those two hybrid orbitals is called linear.

26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments (HL)
Paper 1A: MCQ Practice (HL)
PDF
Paper 1B: Data & Experiments (HL)
PDF
Paper 2: Short Response (HL)
PDF
Official Markscheme (HL)
PDF
17
Cl
35.45
Assignments & Labs Section 08