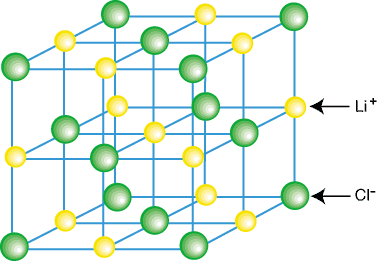
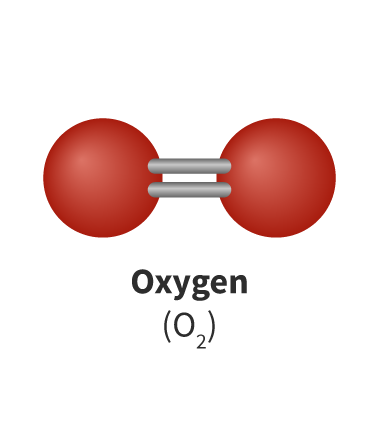
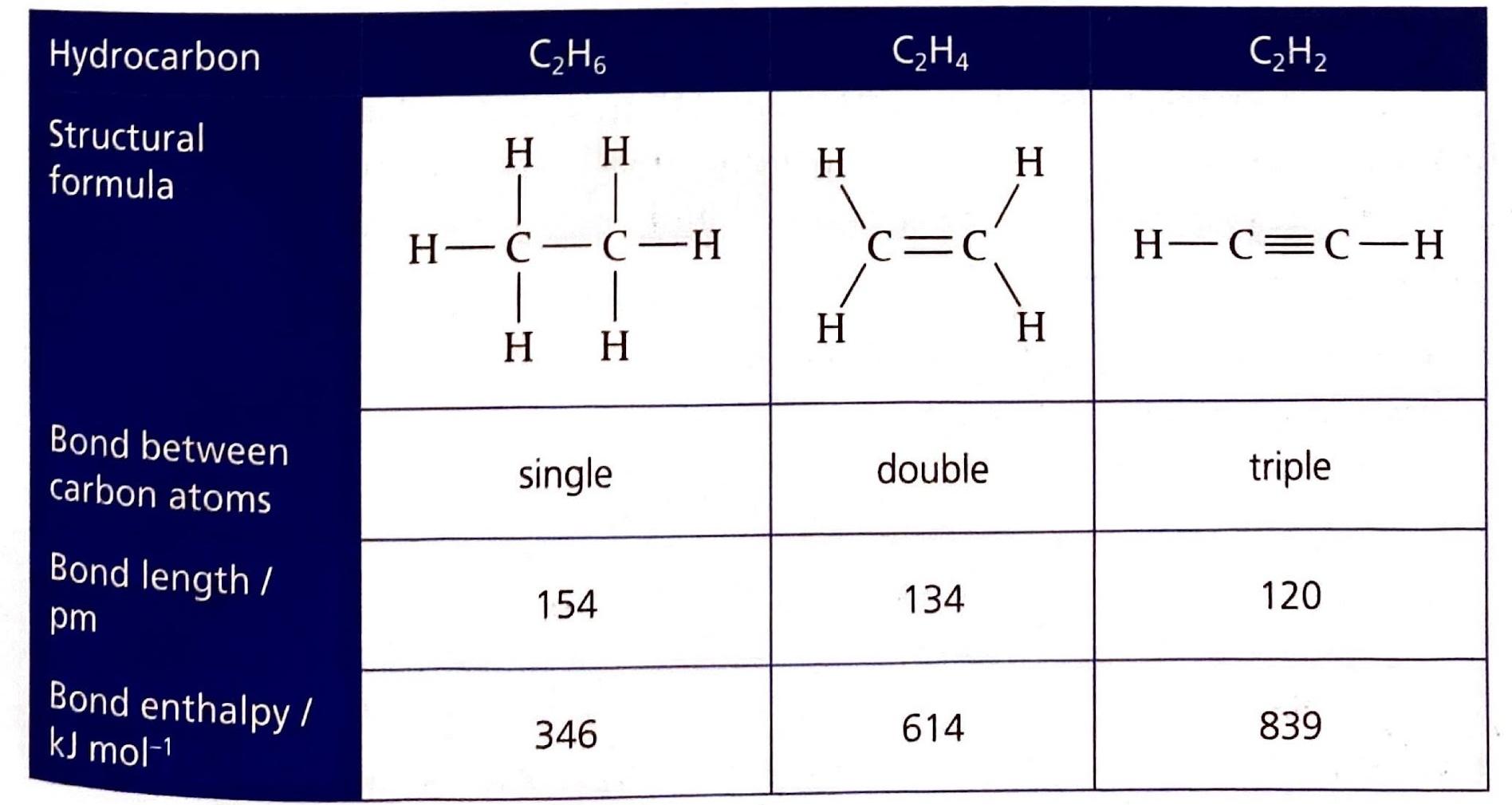
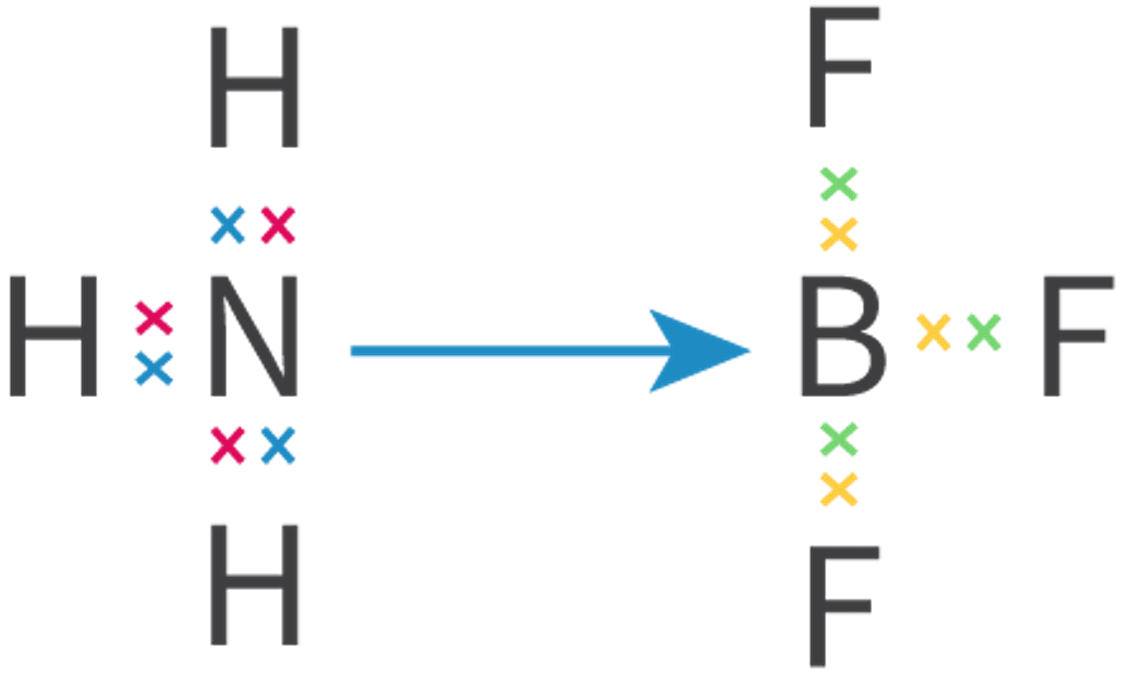
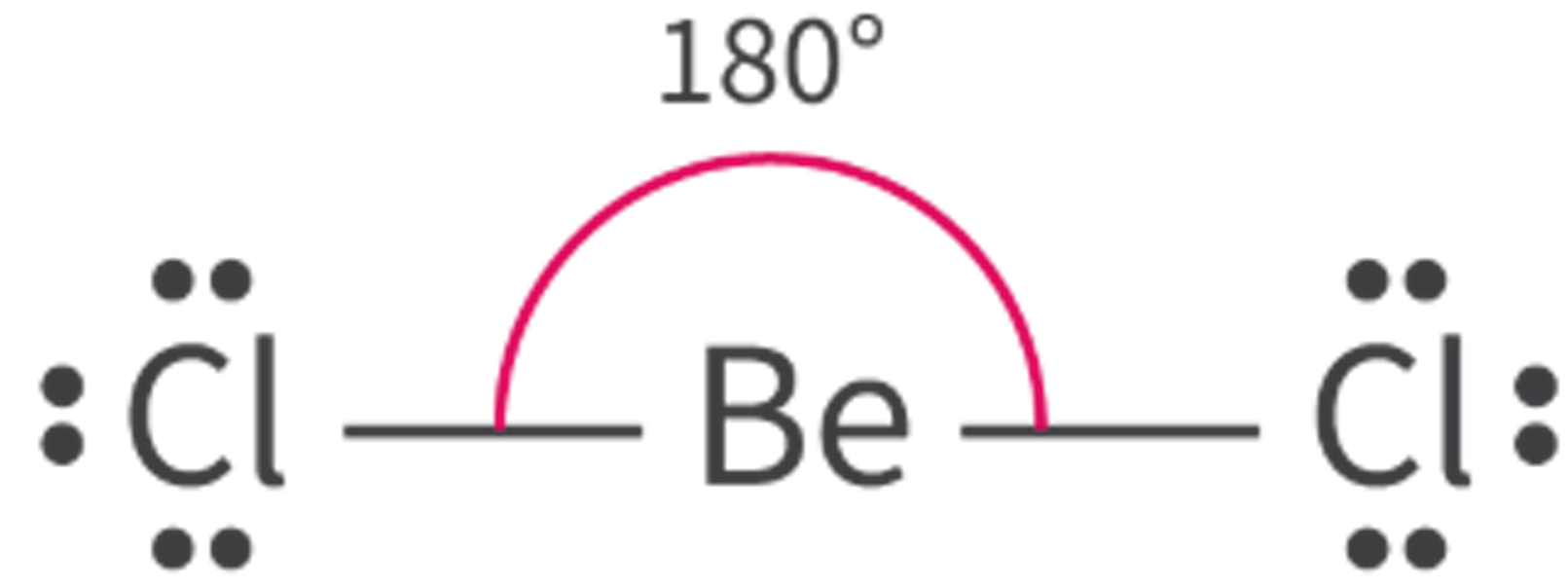
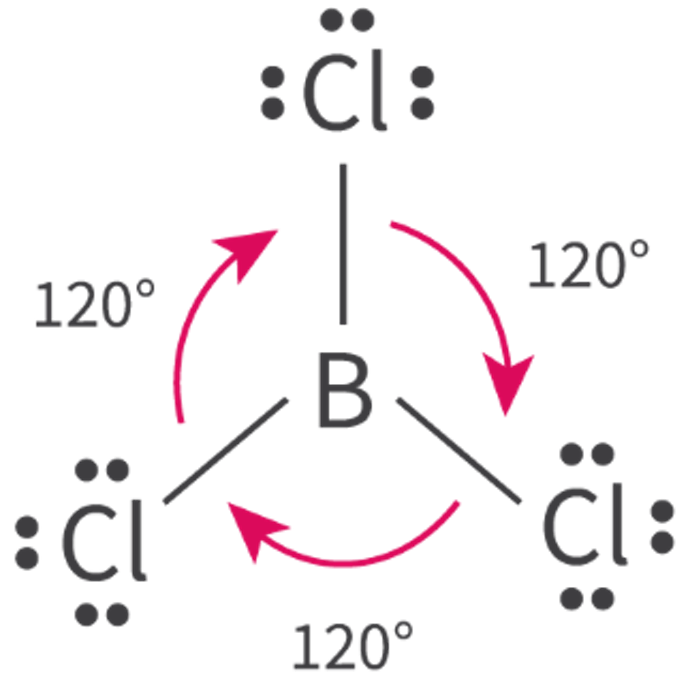
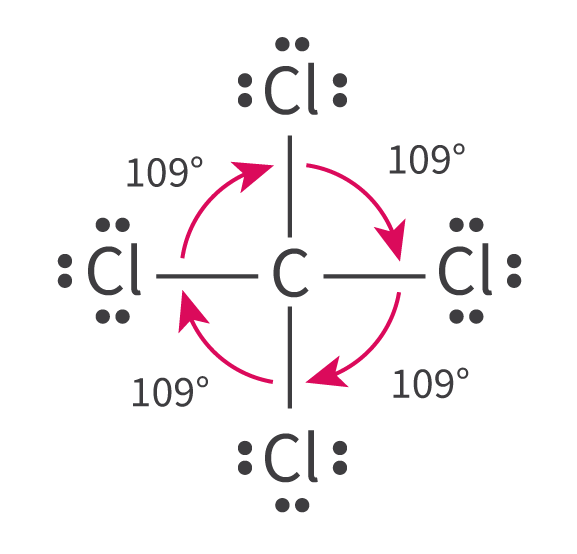
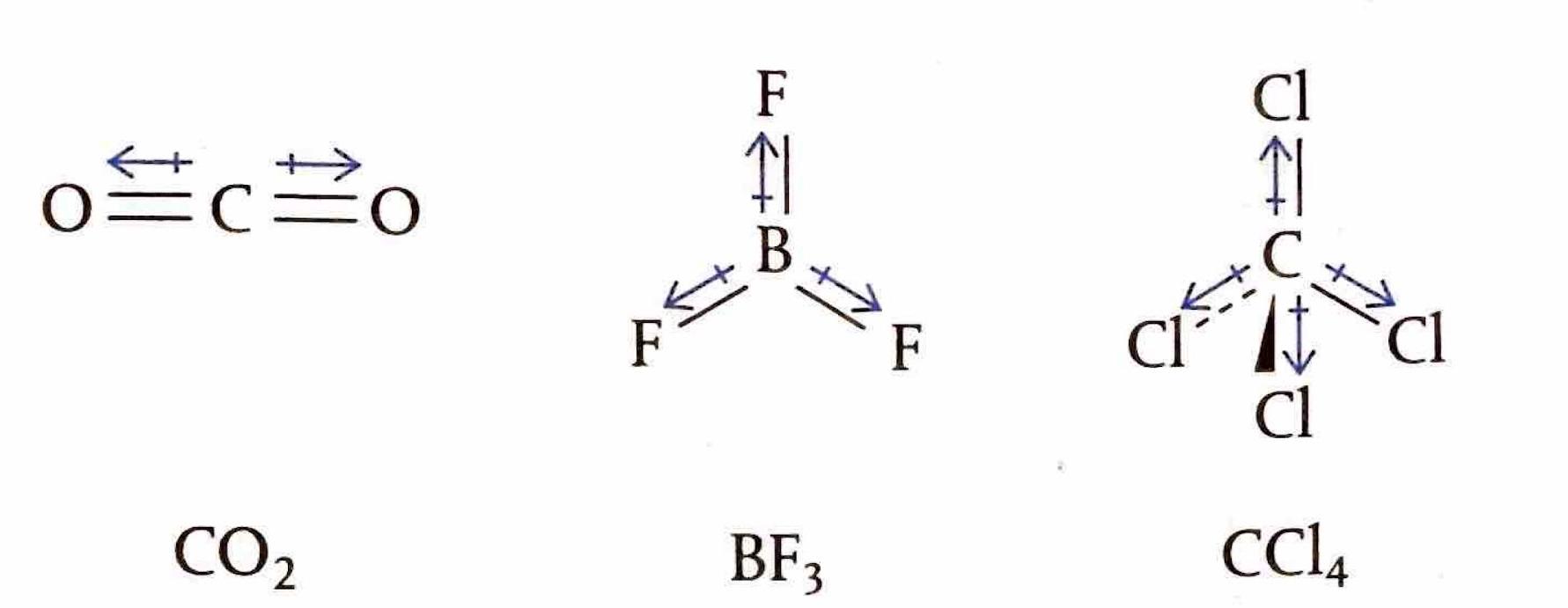
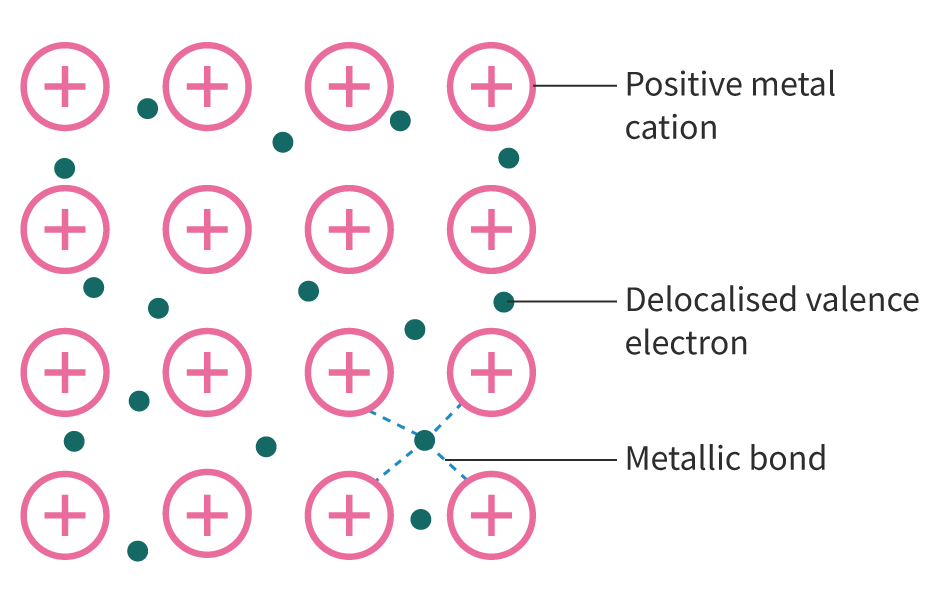
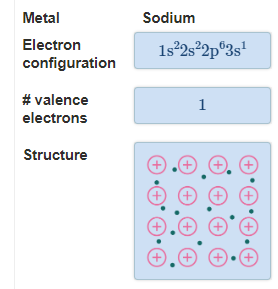
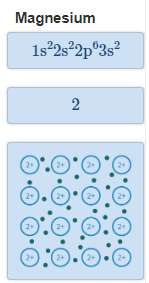
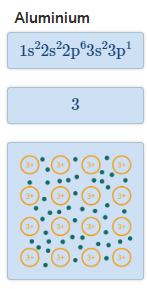
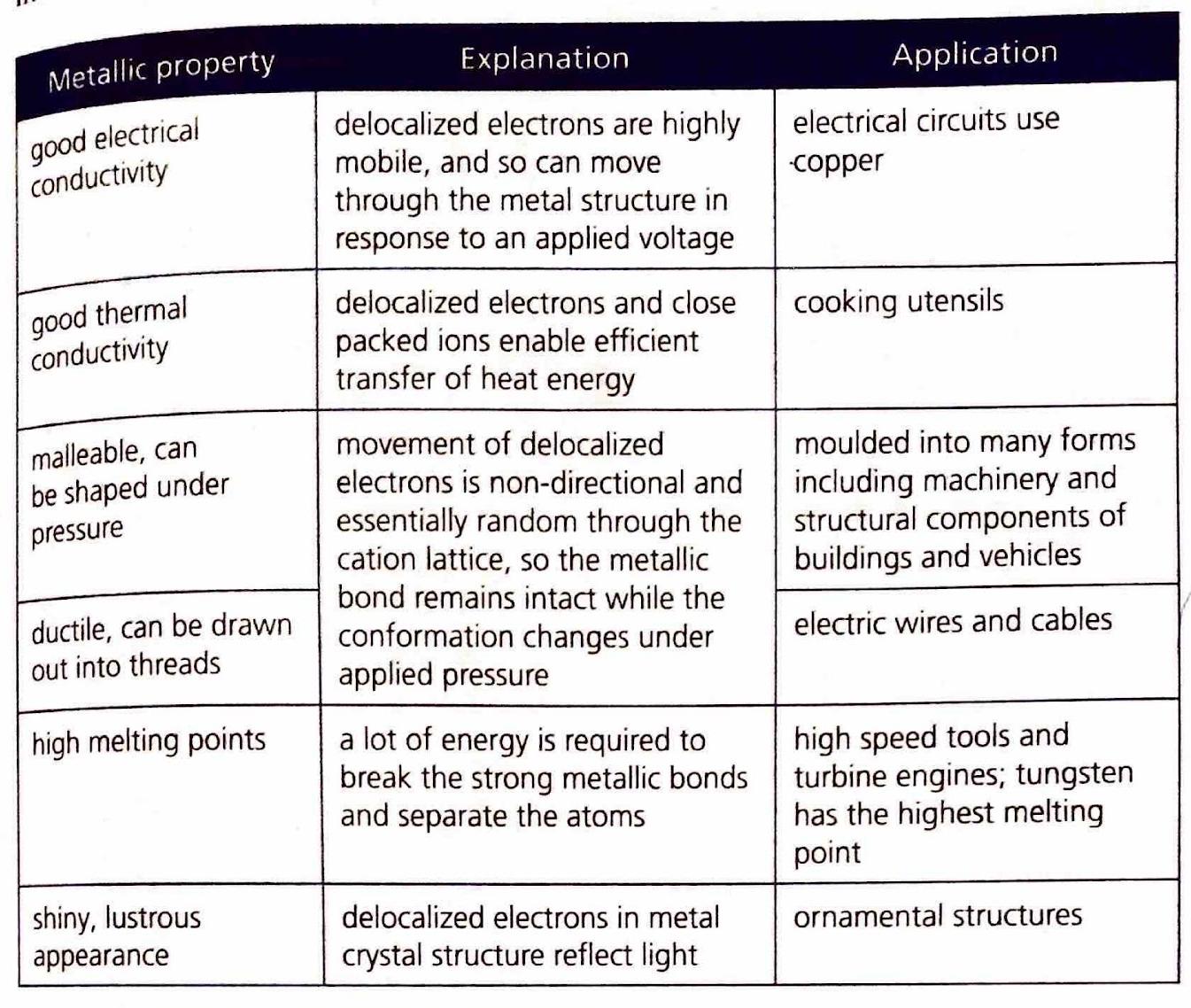
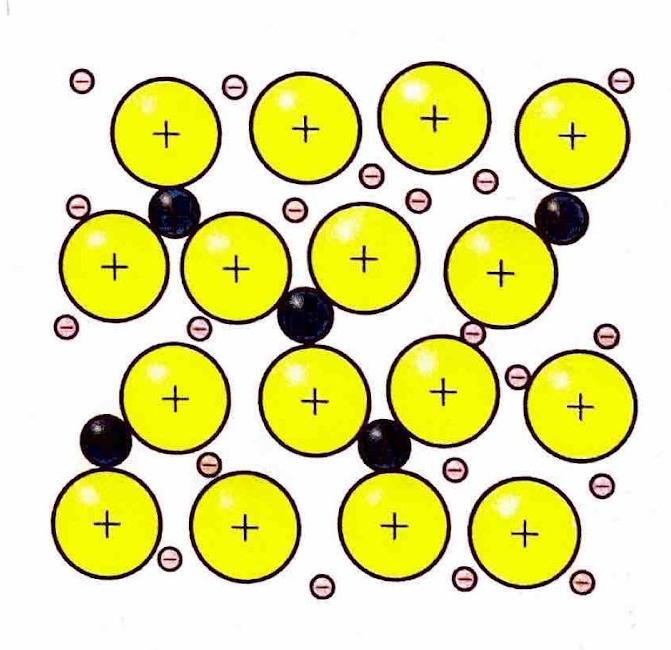
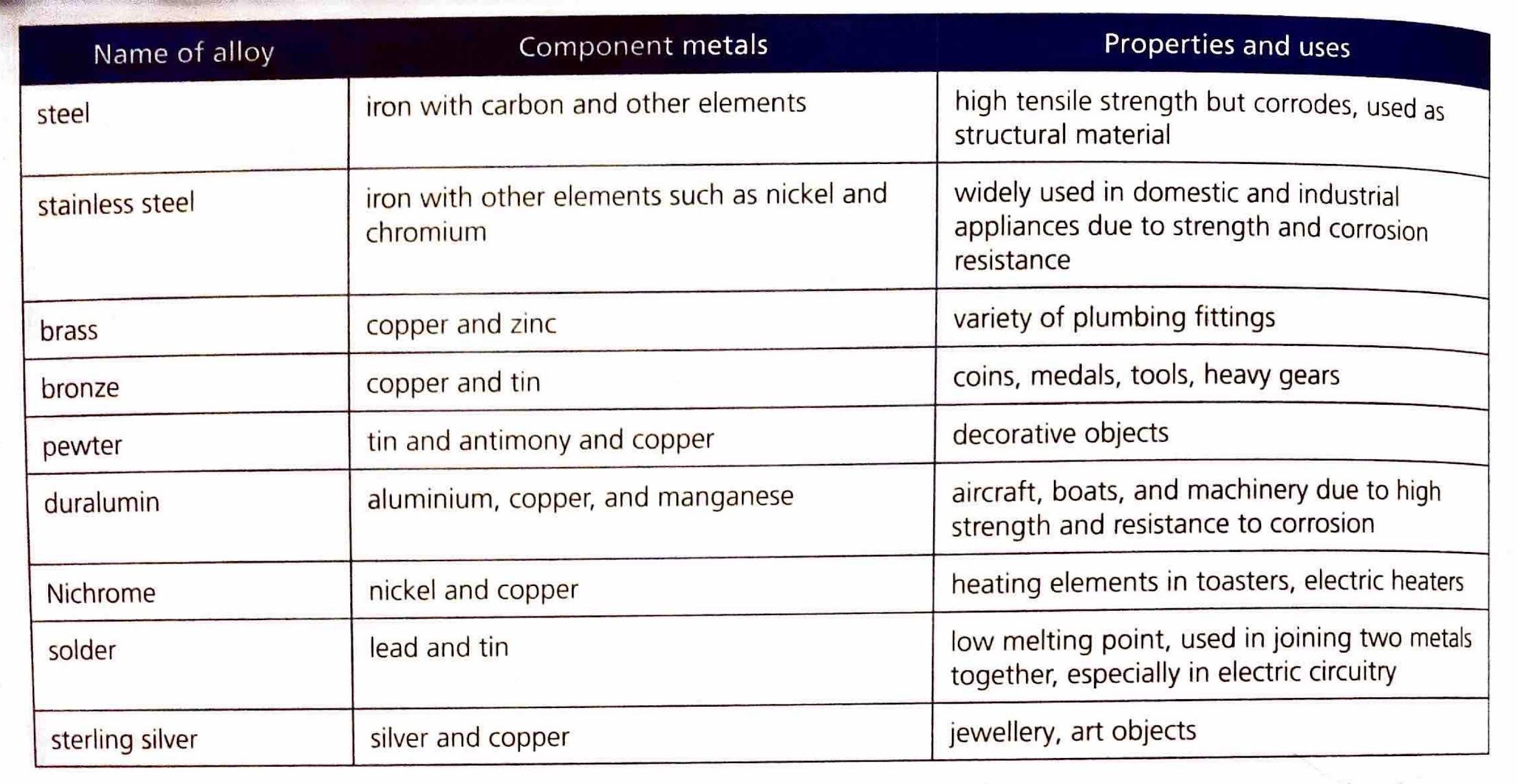
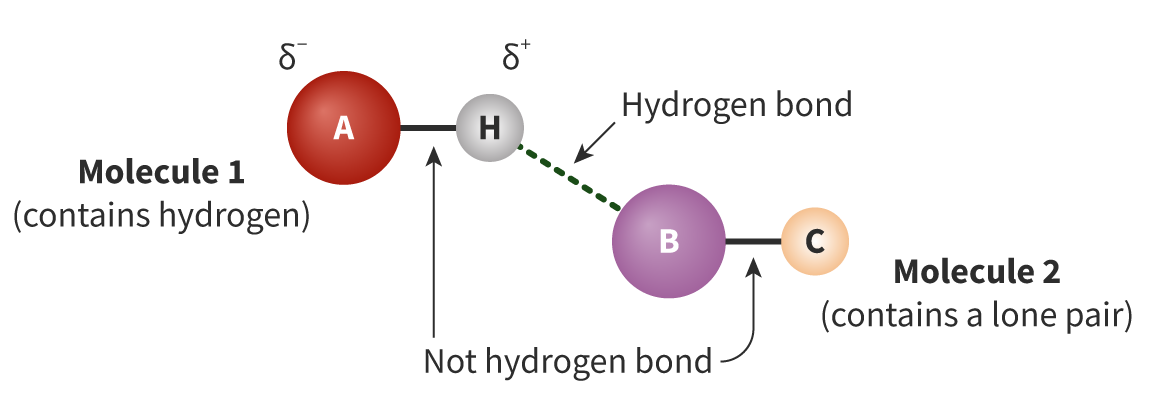
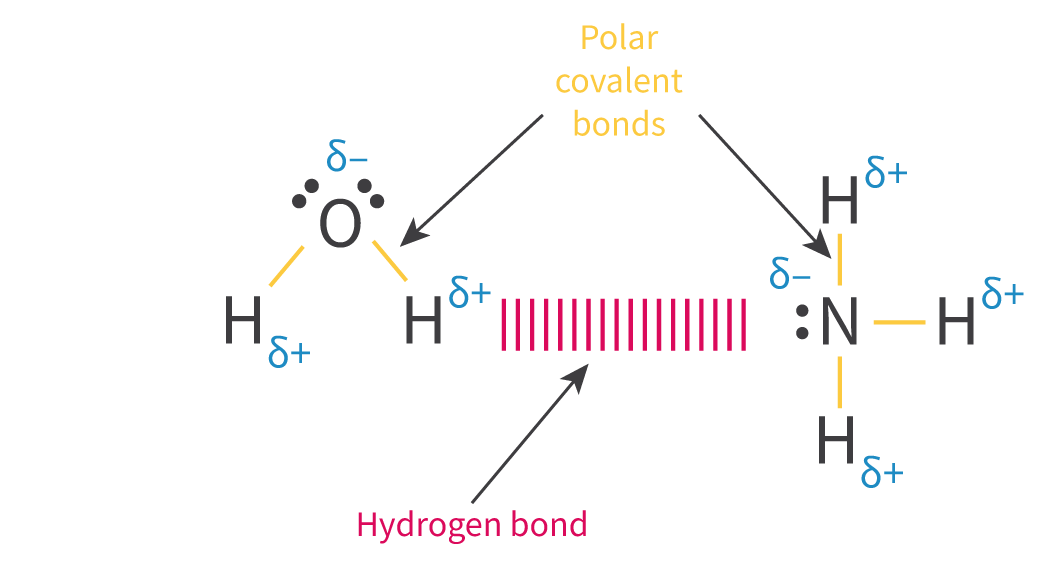
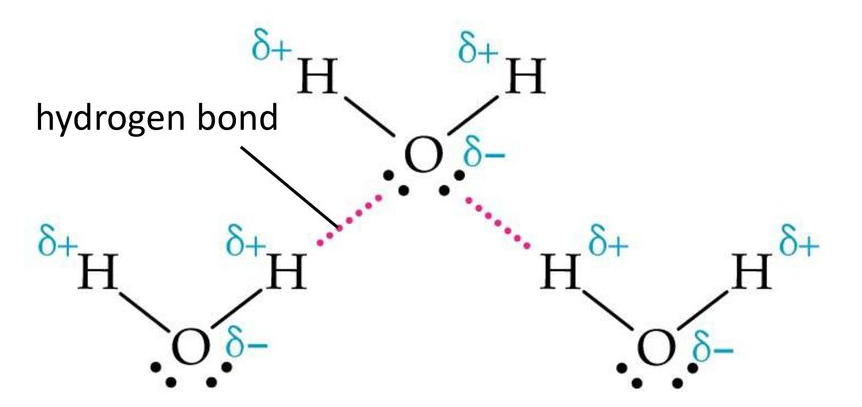
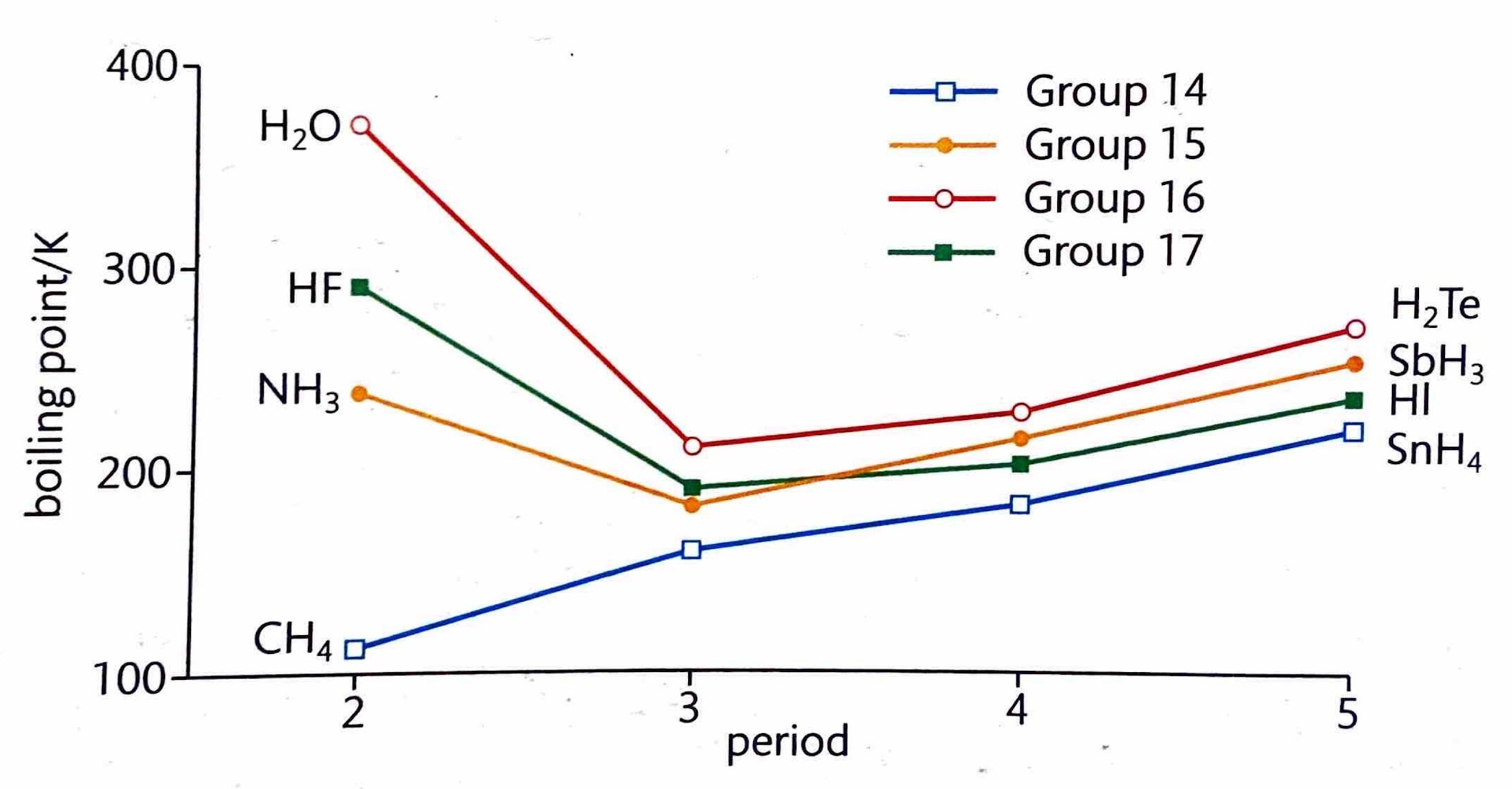
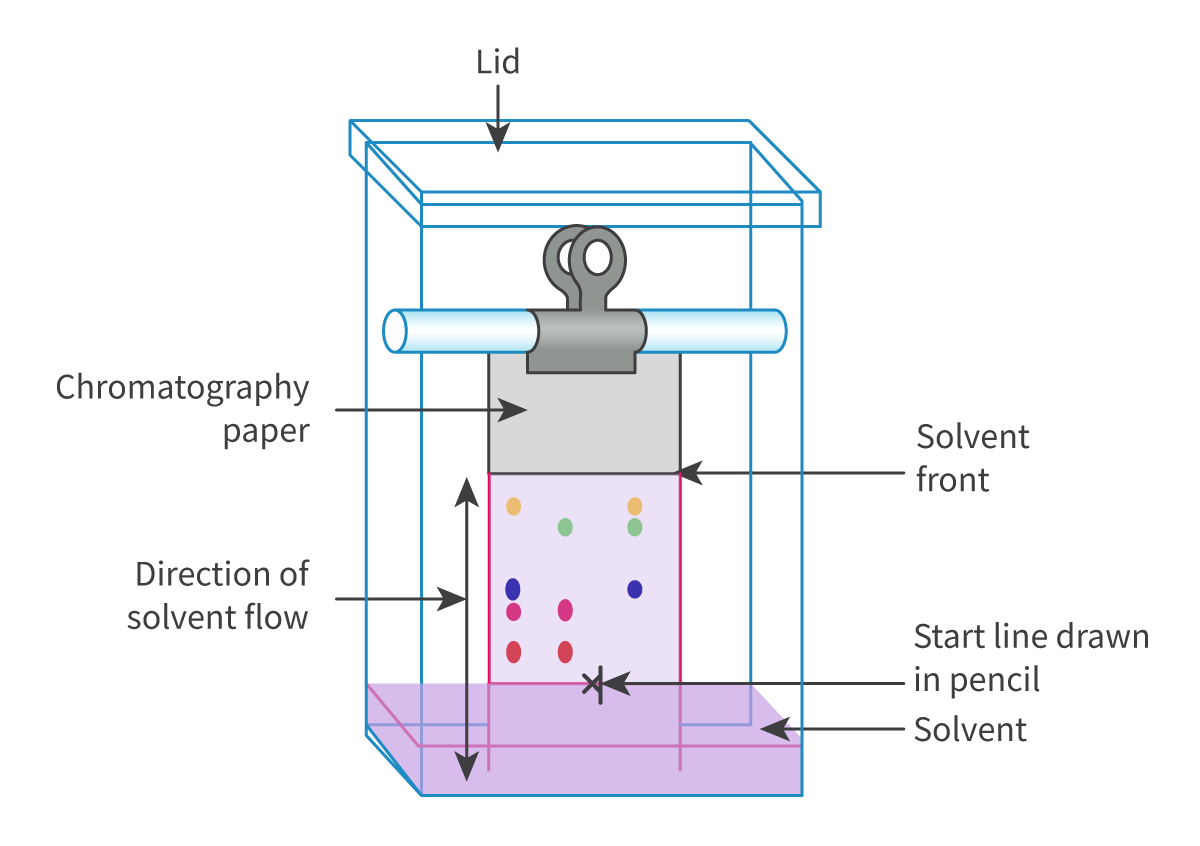
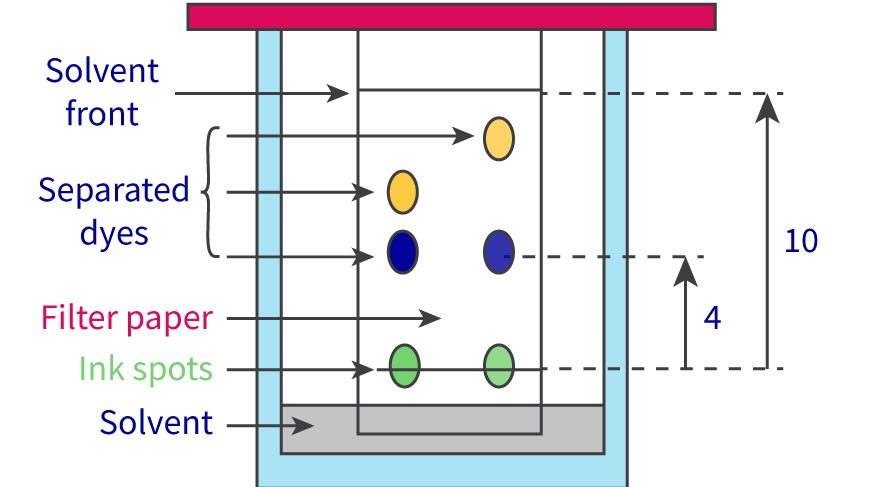
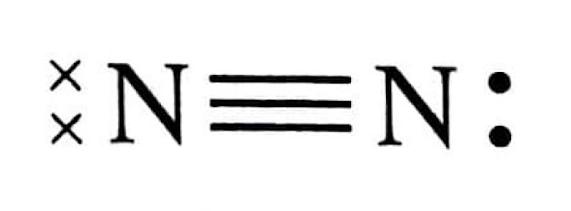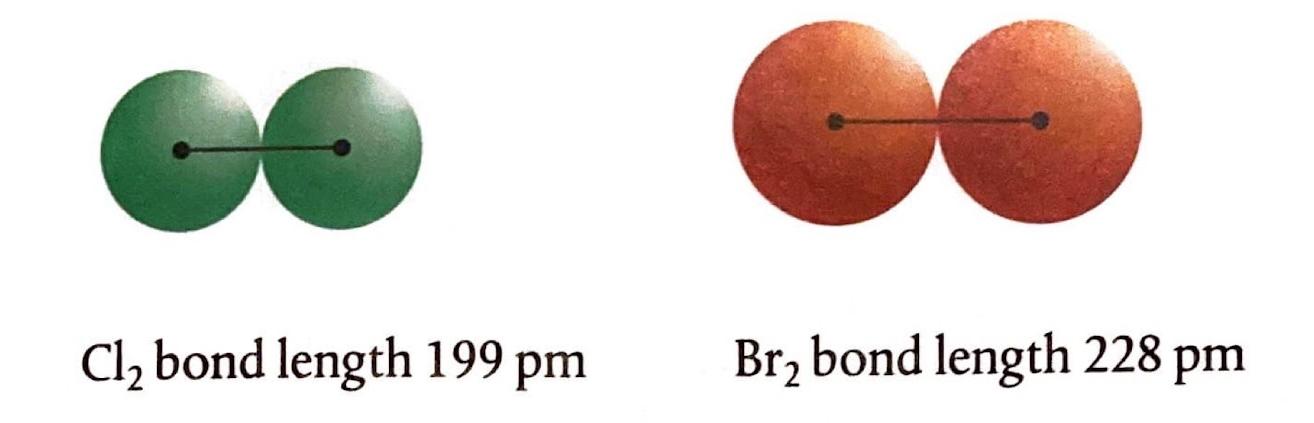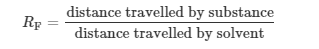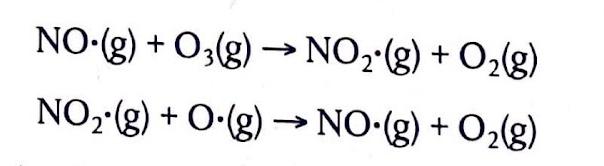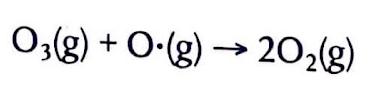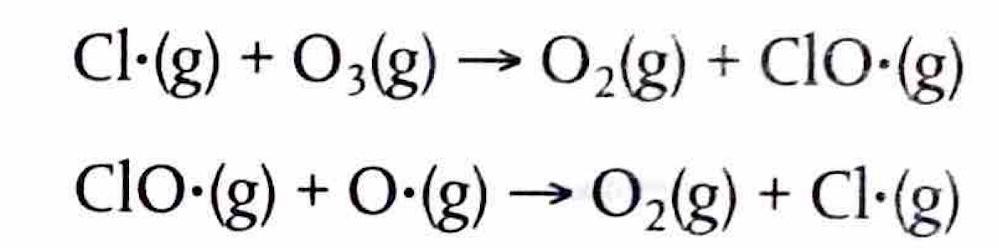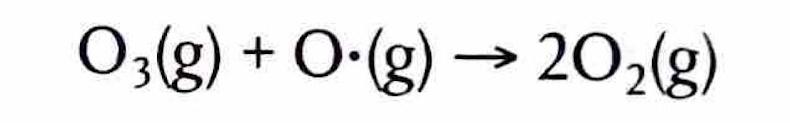The Extent of Ionic Character
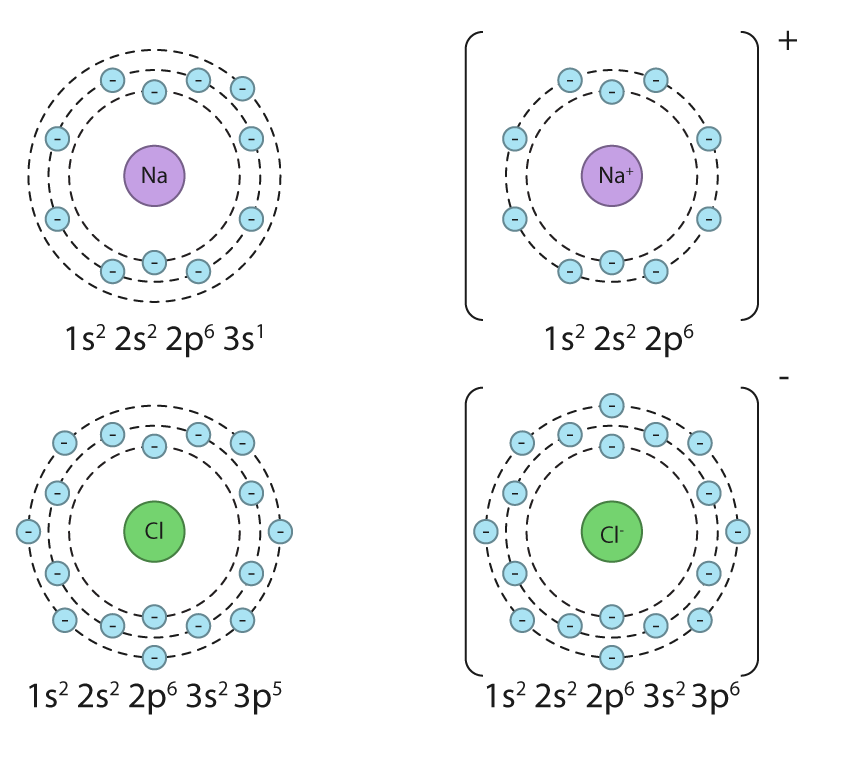
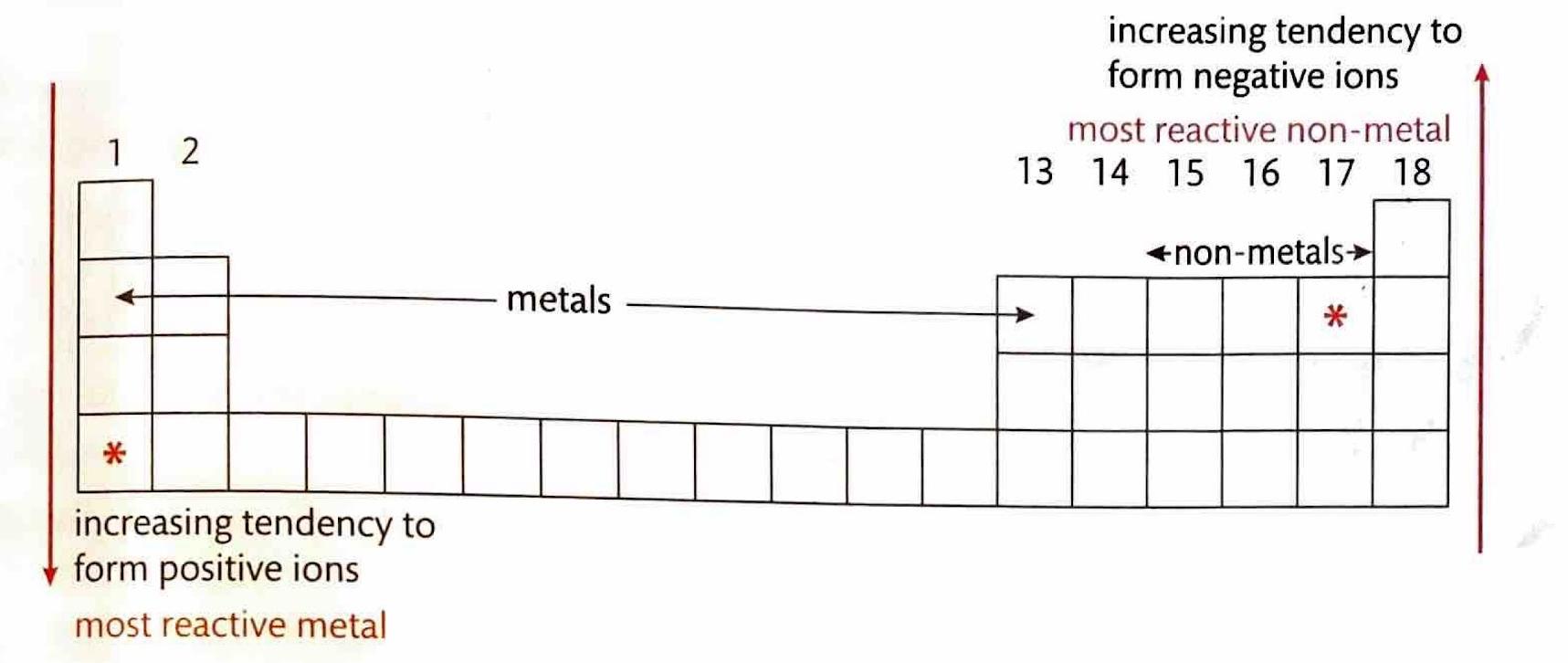
The ease with which elements form ionic compounds is largely determined by their positions on the periodic table. Elements located in the bottom-left corner, which are typically metals, readily react with nonmetals found in the top-right corner to form ionic bonds. This is due to the significant difference in their tendencies to gain or lose electrons.
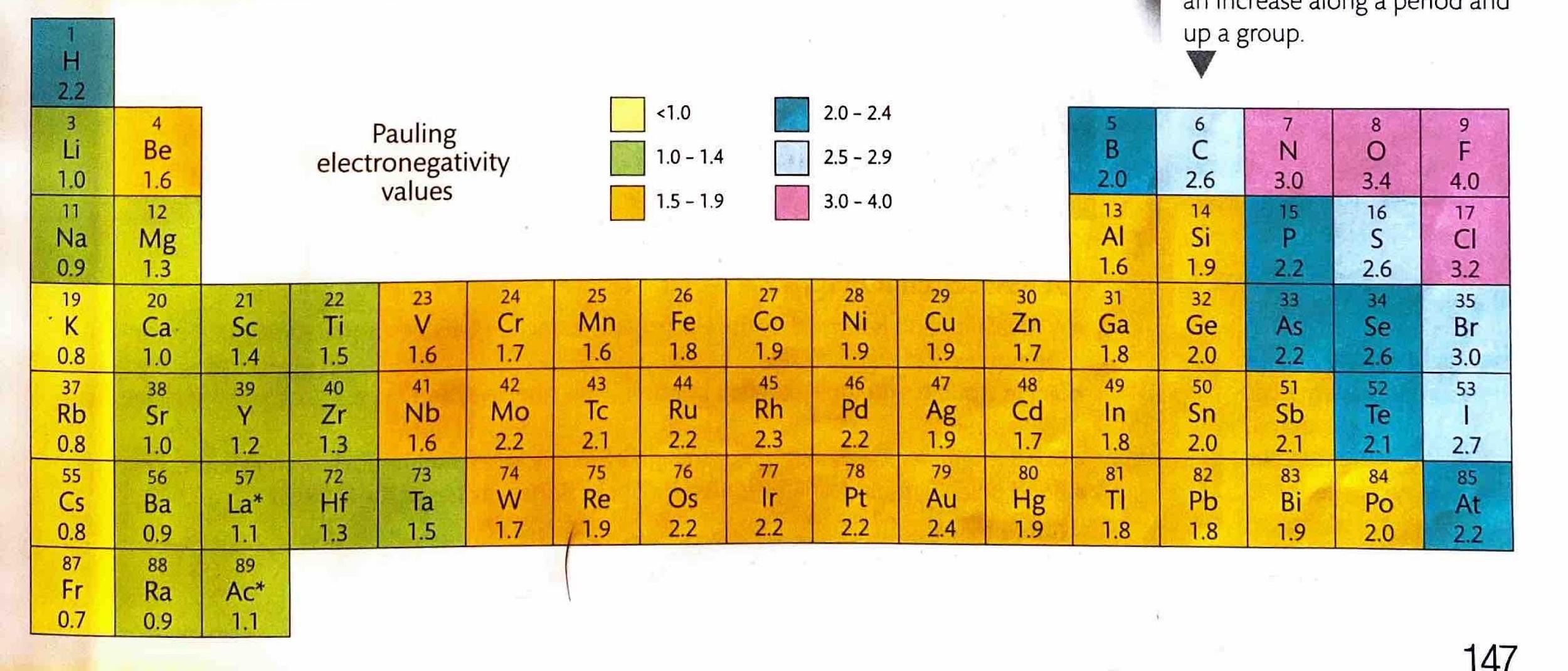
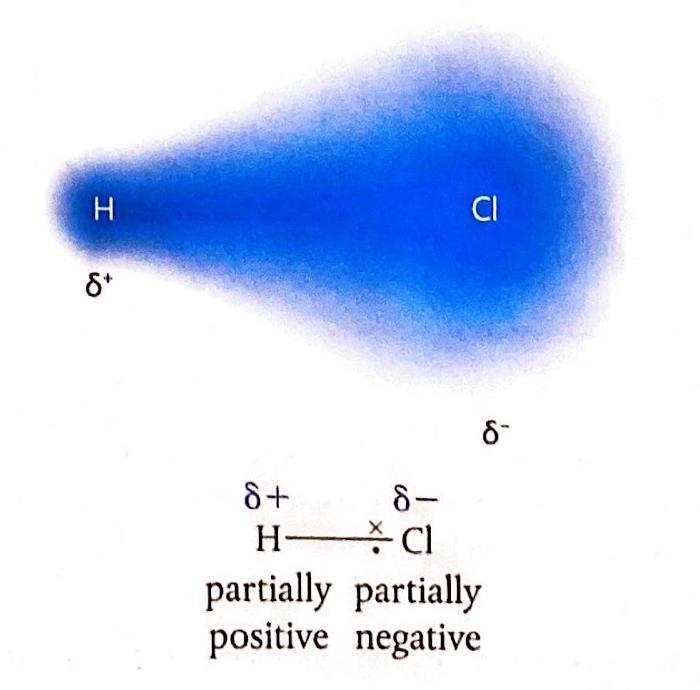
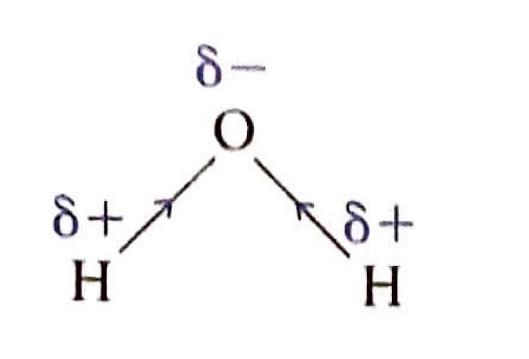
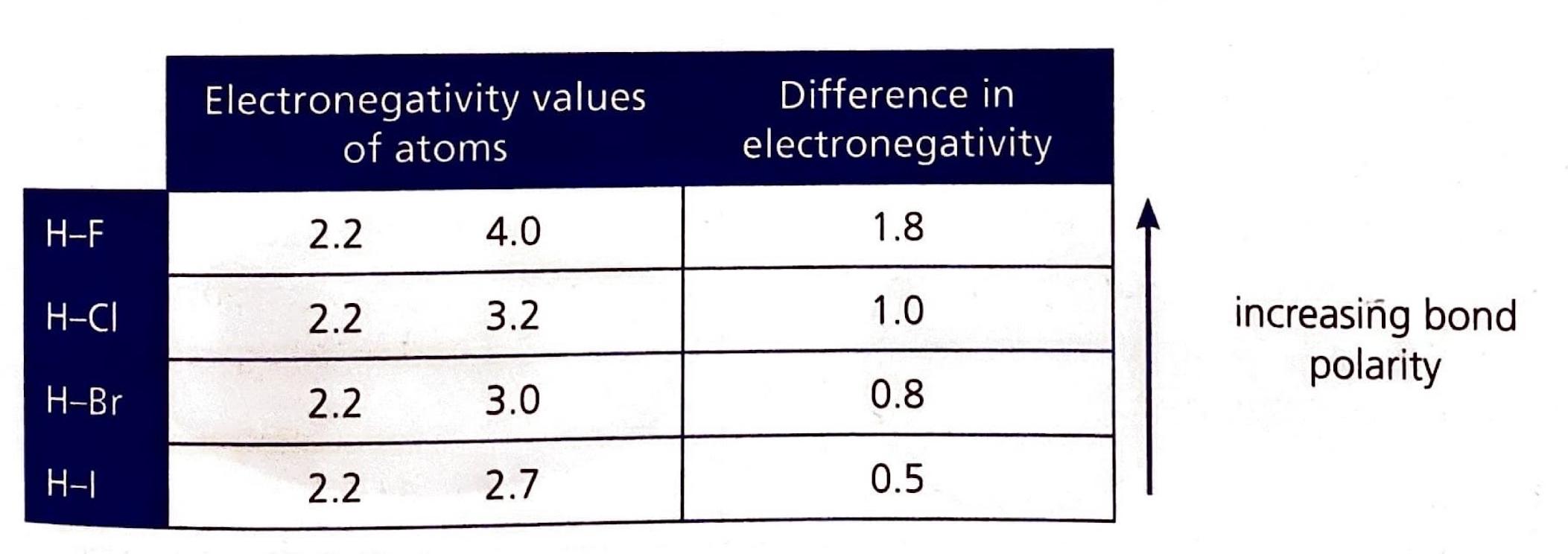
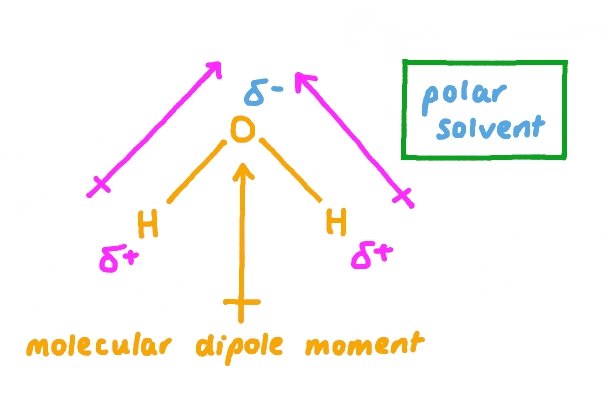
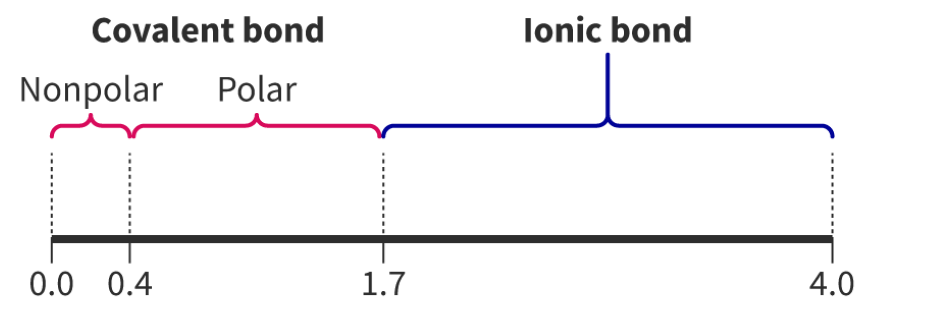
A crucial factor in determining the extent of ionic character in a bond is the difference in electronegativity (ΔEN) between the two bonded atoms. Bonds with a ΔEN greater than 1.8 are generally considered to be predominantly ionic. Electronegativity values, which quantify an atom's ability to attract electrons in a chemical bond, can be found in the Data Booklet.
The Continuum of Chemical Bonding
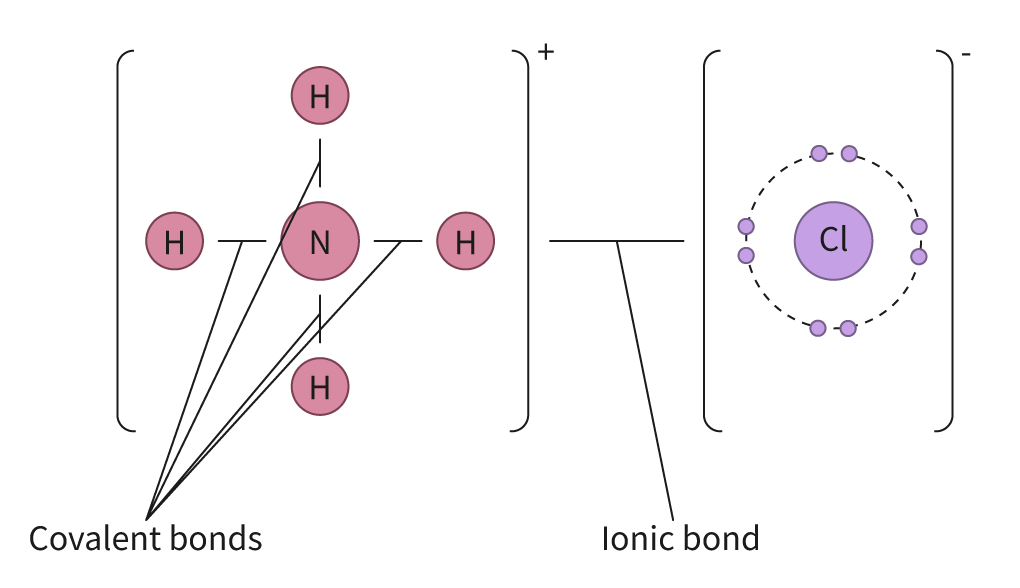
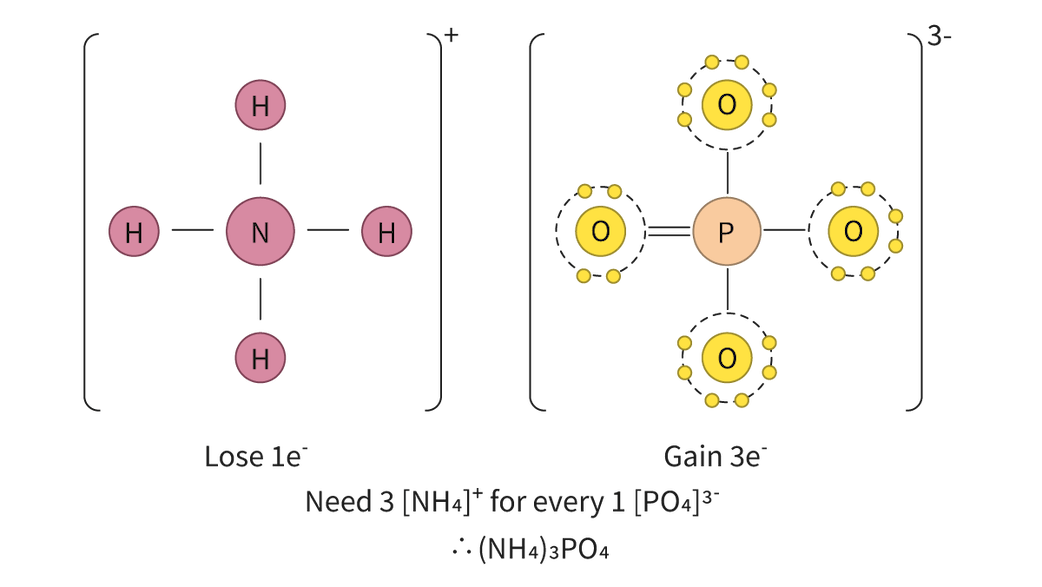
It is important to understand that the distinction between ionic and covalent bonds is not absolute but rather represents a continuum. Many bonds exhibit characteristics of both ionic and covalent bonding, falling somewhere along this spectrum.
Reactions Forming Ionic Compounds: Group 1 Elements
Group 1 alkali metals are highly reactive and readily form ionic compounds. For instance, when alkali metals react with water, they produce hydrogen gas and a metal hydroxide. The reactivity of these metals increases as one moves down the group, primarily because the effective nuclear charge experienced by the valence electrons decreases, making it easier to lose an electron. A typical example is the reaction of potassium with water:
2K(s) + 2H
2O(l) → 2KOH(aq) + H
2(g)
This can also be represented in its ionic form:
2K(s) + 2H
2O(l) → 2K
+(aq) + 2OH
-(aq) + H
2(g)
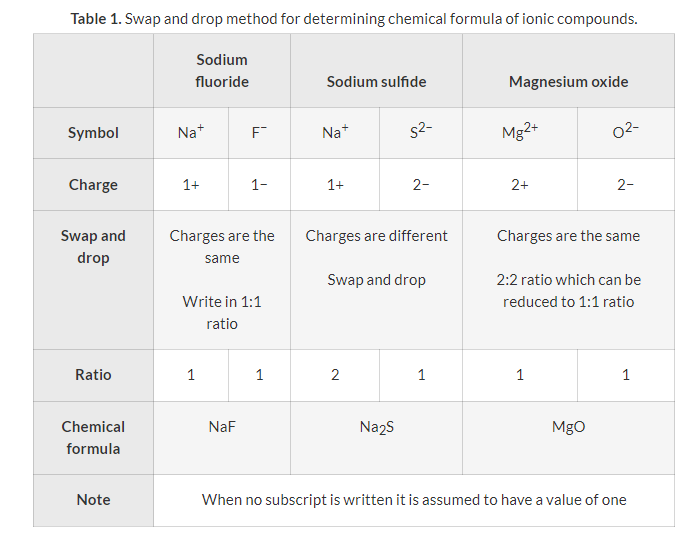
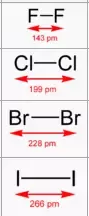
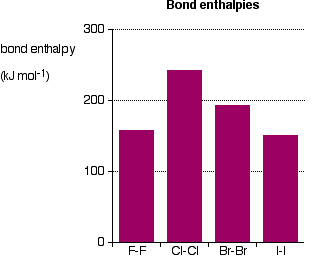
Alkali metals also react vigorously with halogens to form ionic halides. The most energetic reactions occur between elements that are furthest apart on the periodic table, such as those at the bottom of Group 1 and the top of Group 17. For example, sodium reacts with chlorine to form sodium chloride:
2Na(s) + Cl
2(g) → 2NaCl(s)
Displacement Reactions Involving Halogens
The relative reactivity of elements can be observed through displacement reactions, where elements compete for an electron. In the reaction between potassium bromide and chlorine, for example:
2KBr(aq) + Cl
2(aq) → 2KCl(aq) + Br
2(aq)
The net ionic equation for this reaction is:
2Br
-(aq) + Cl
2(aq) → 2Cl
-(aq) + Br
2(aq)
In this case, the chlorine nucleus exerts a stronger attraction for an electron compared to bromine, primarily due to its smaller atomic radius. Consequently, chlorine gains an electron from the bromide ion to form a chloride ion, while the bromide ion loses an electron to form bromine. Similar displacement reactions occur with other halogens:
2I
-(aq) + Cl
2(aq) → 2Cl
-(aq) + I
2(aq)
2I
-(aq) + Br
2(aq) → 2Br
-(aq) + I
2(aq)
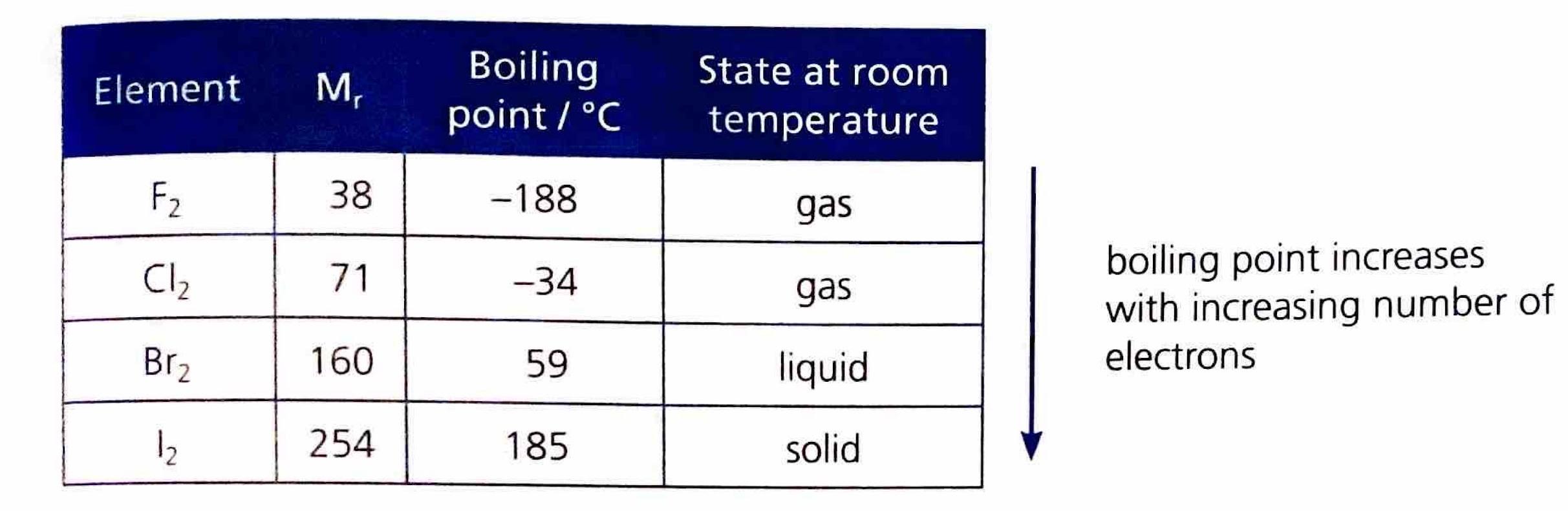
Generally, a more reactive halogen will displace the ion of a less reactive halogen from its compounds.
It is also noteworthy that halogens form insoluble salts with silver. Therefore, adding a solution containing a halide ion to a solution of silver ions will result in the formation of a precipitate:
Ag
+(aq) + X
-(aq) → AgX(s)
Bonding Characteristics of Period 3 Oxides
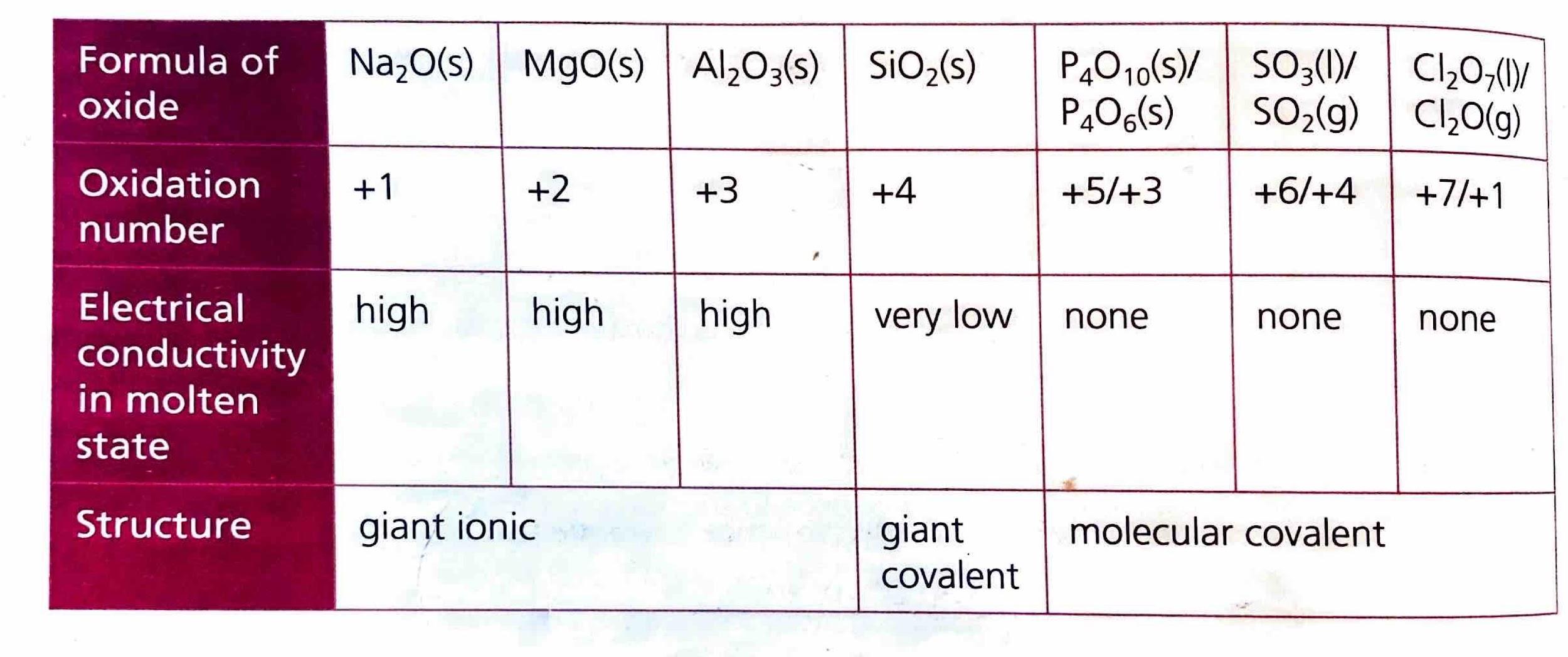
Elements in Period 3 frequently combine with oxygen to form various oxides. The ionic character of these oxides generally decreases from left to right across the period. This trend occurs because, while electronegativity increases across the period, the difference in electronegativity (ΔEN) between the Period 3 element and oxygen tends to decrease, leading to more covalent character. Conversely, the ionic character of oxides increases down a group, as the electronegativity of the metal decreases, resulting in larger ΔEN values with oxygen. The electrical conductivity of molten oxides provides an experimental measure of their ionic character; higher conductivity indicates a more ionic compound.
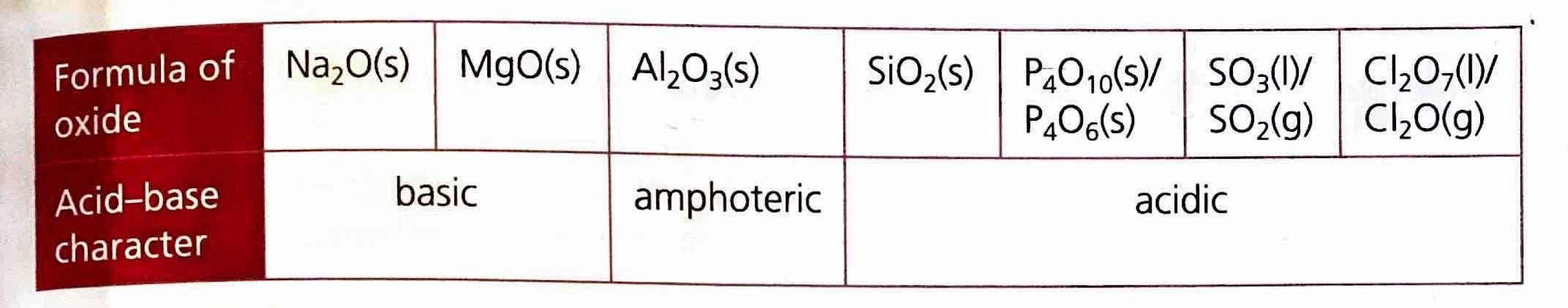
Acid-Base Properties of Period 3 Oxides
The acid-base properties of Period 3 oxides are intrinsically linked to their bonding and structural characteristics. Metals, which typically form ionic oxides, tend to produce basic oxides. In contrast, nonmetals, which form covalent oxides, generally yield acidic oxides. Aluminum oxide (Al
2O
3) presents a unique case, exhibiting both ionic and covalent character. Consequently, it can act as both an acid and a base, a property known as amphoterism.