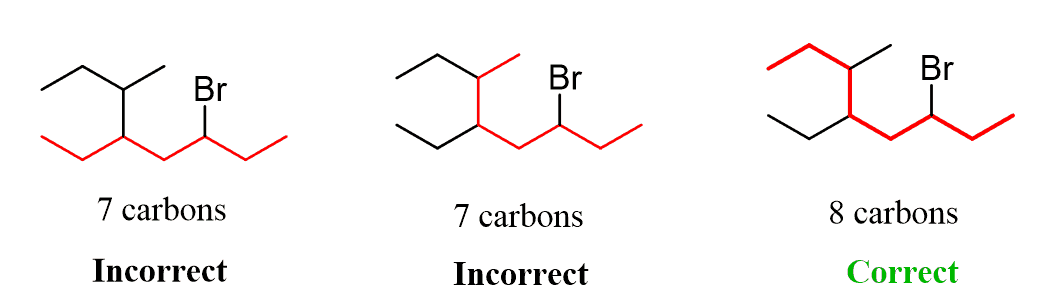
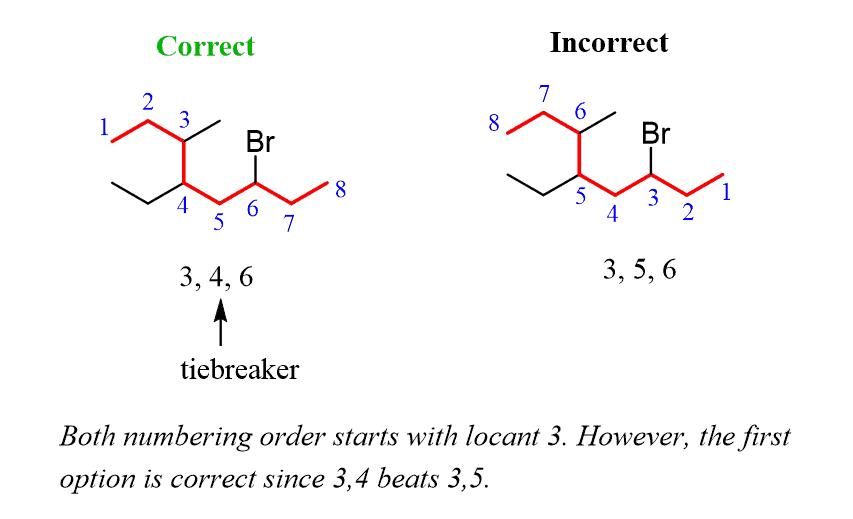
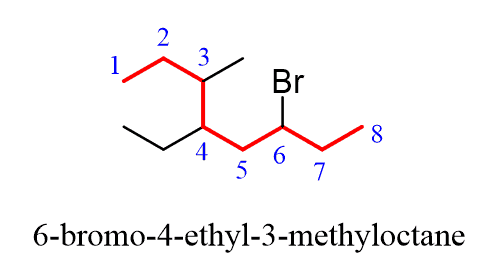
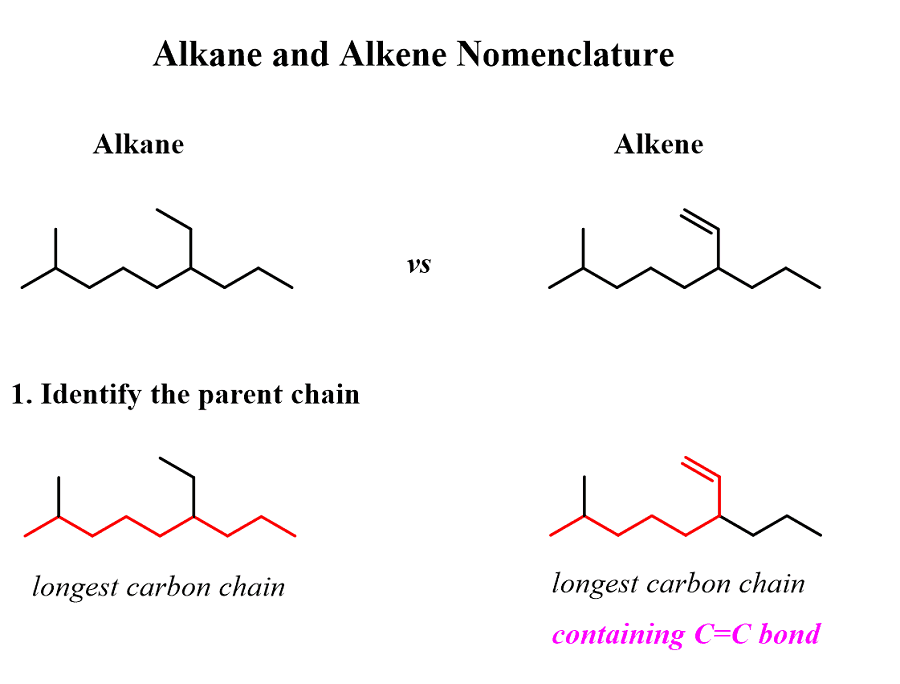
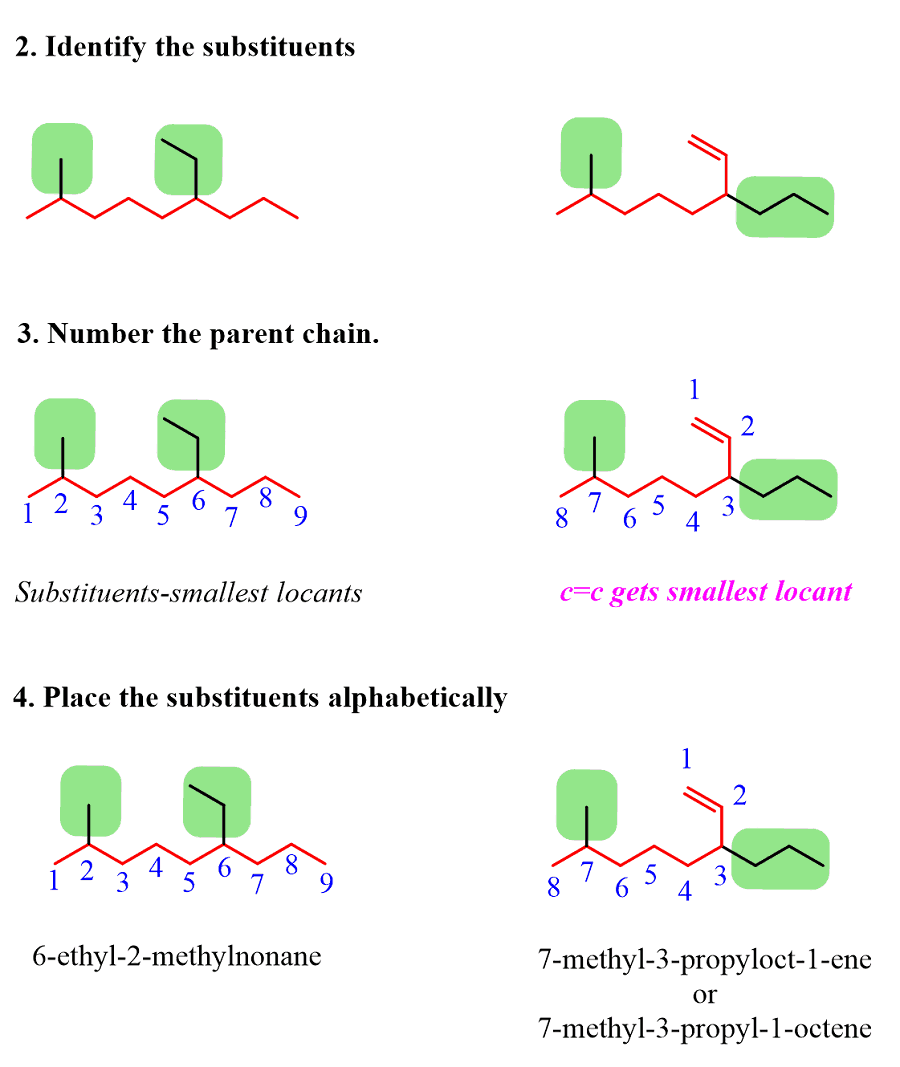
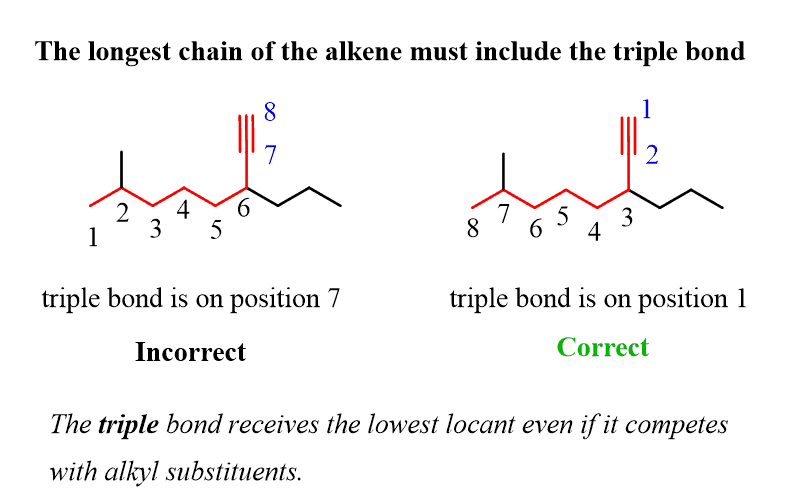
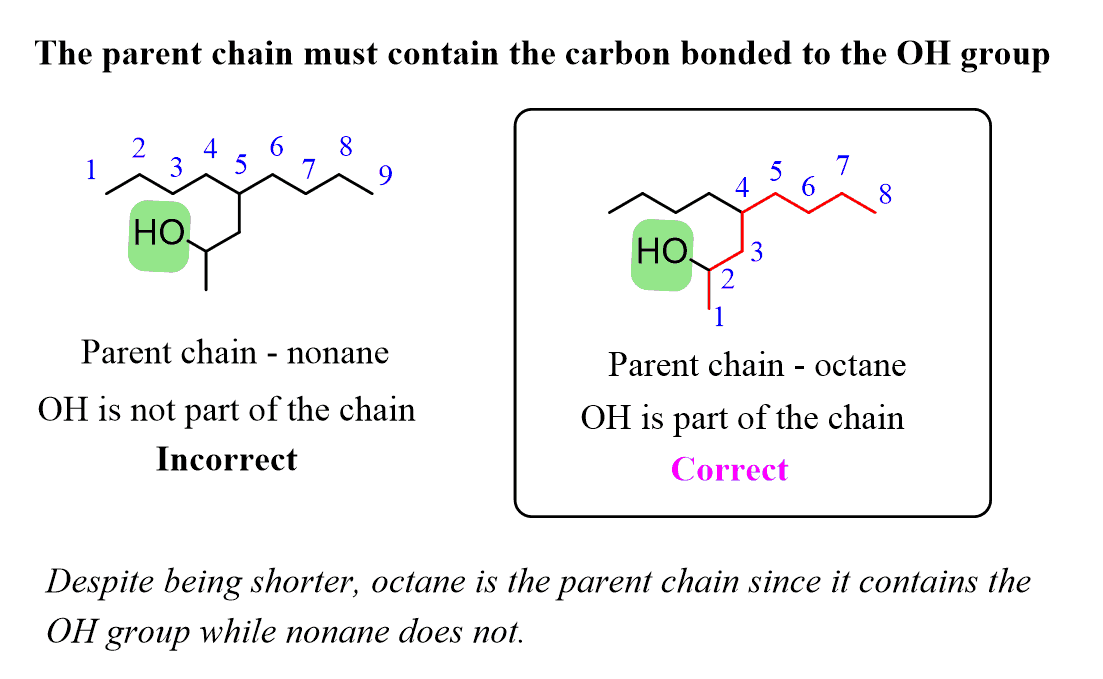
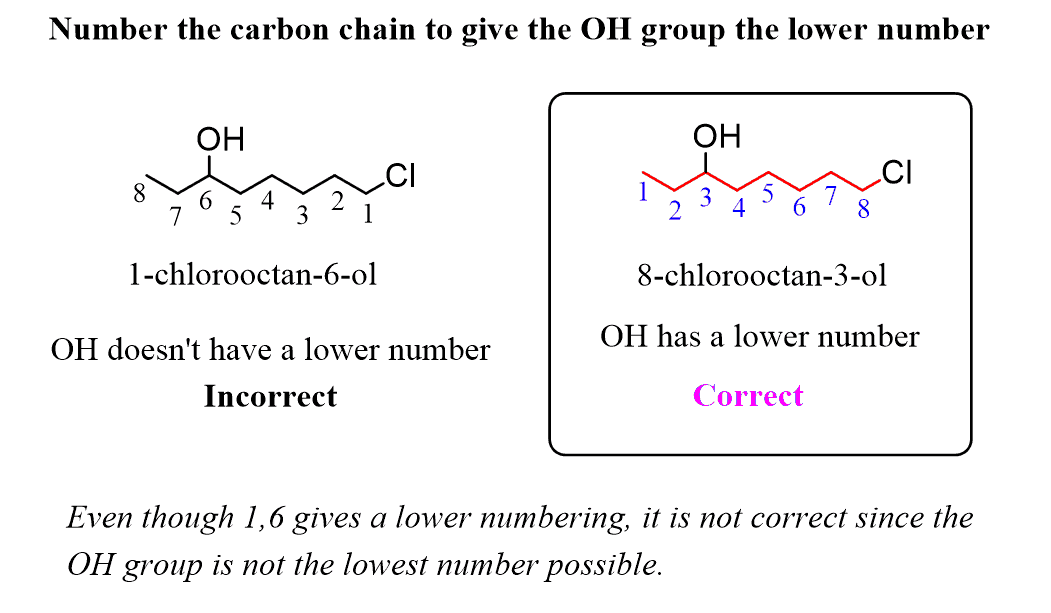
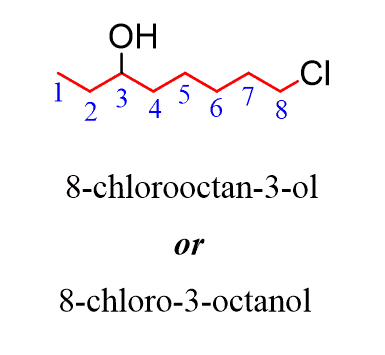
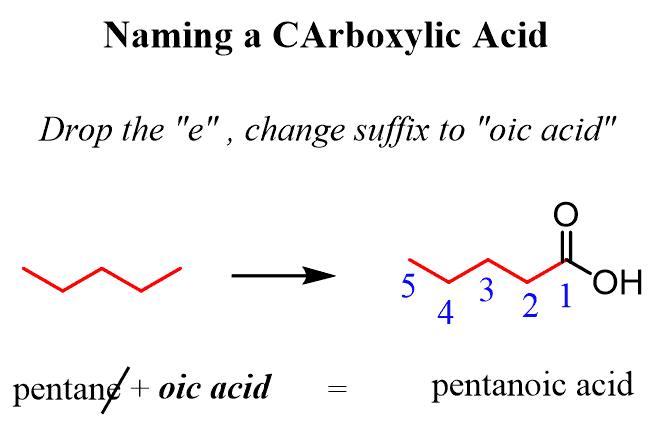
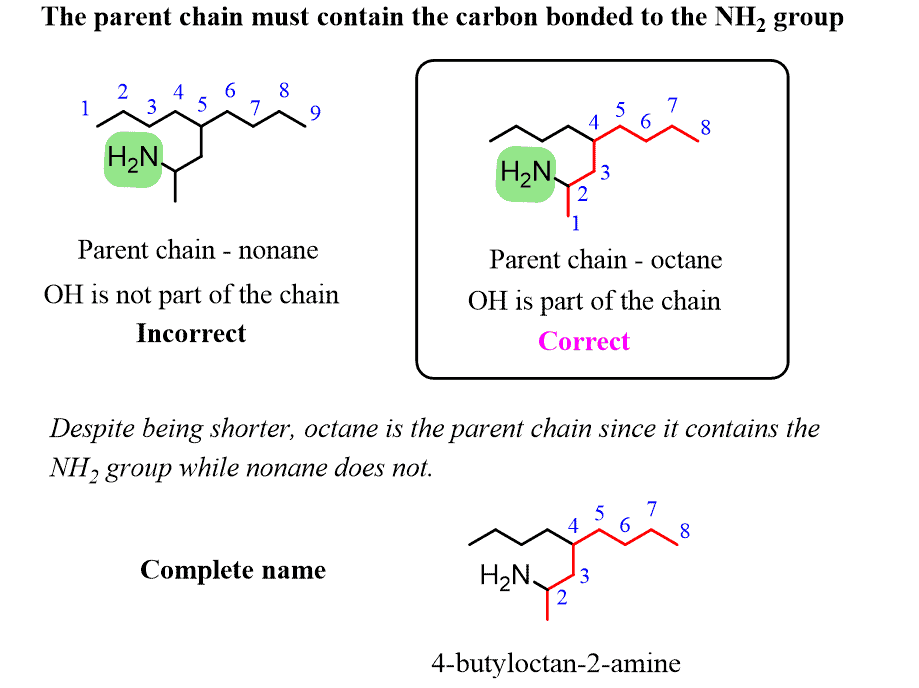
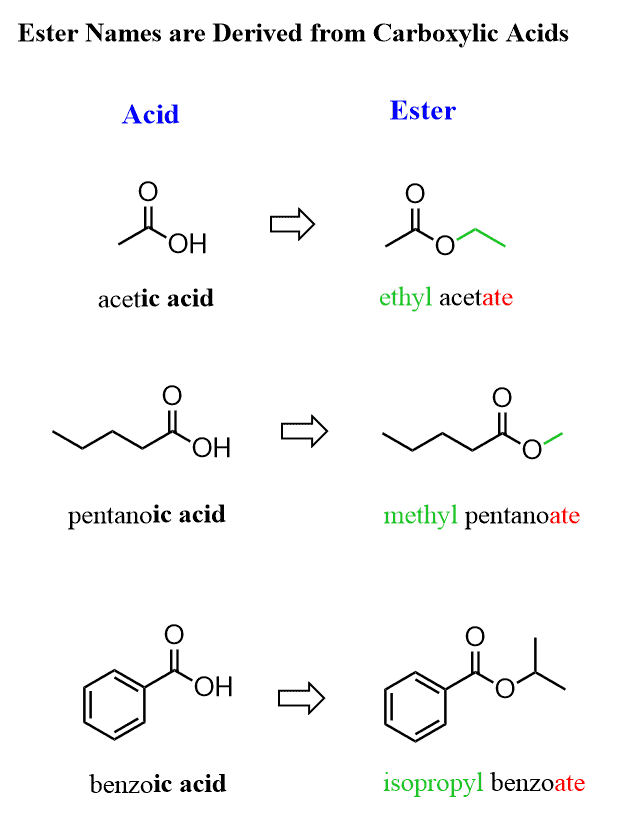
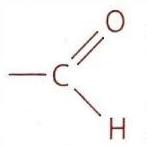
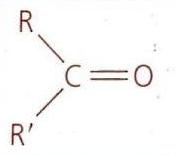
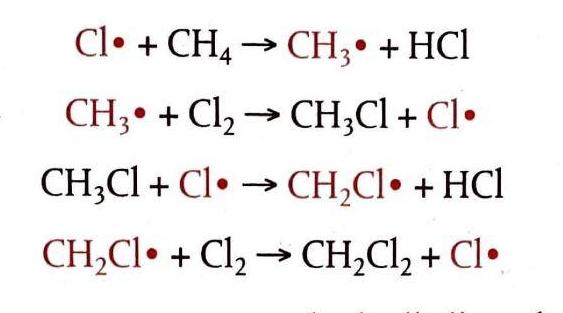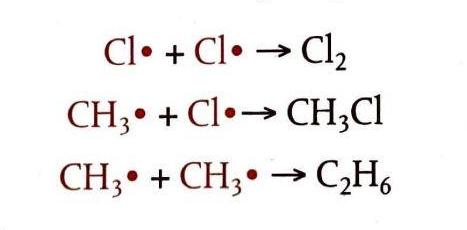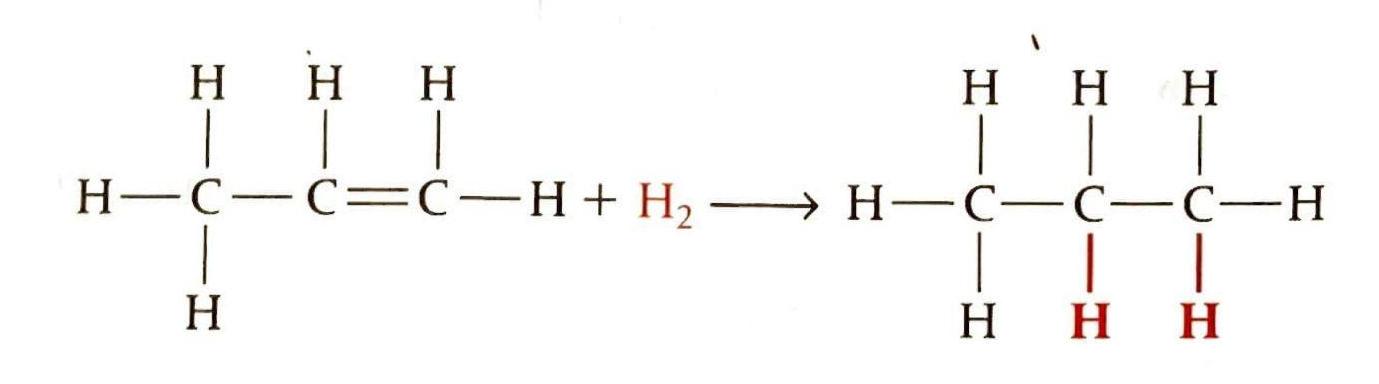Alkanes: Structure and General Reactivity
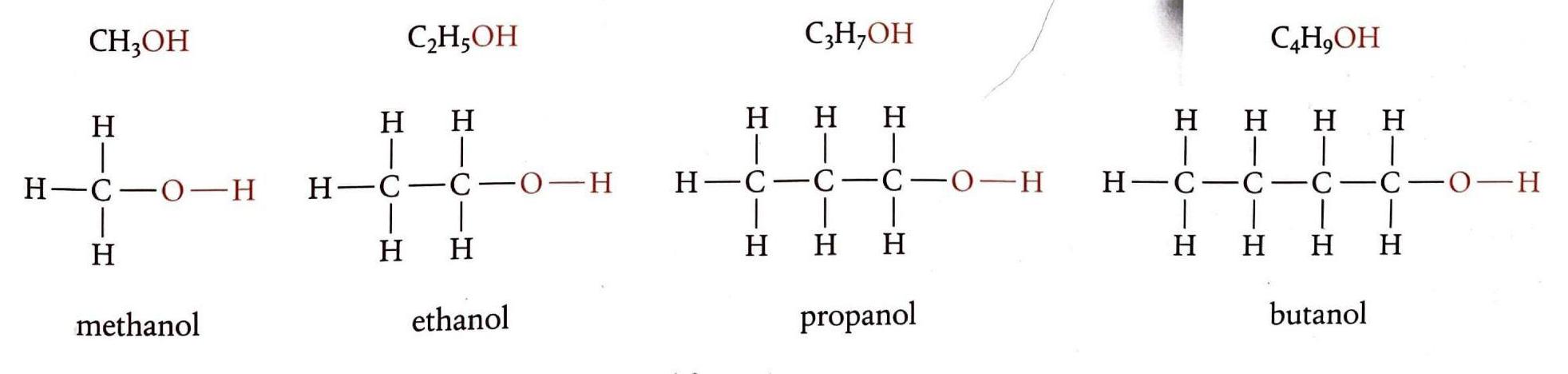
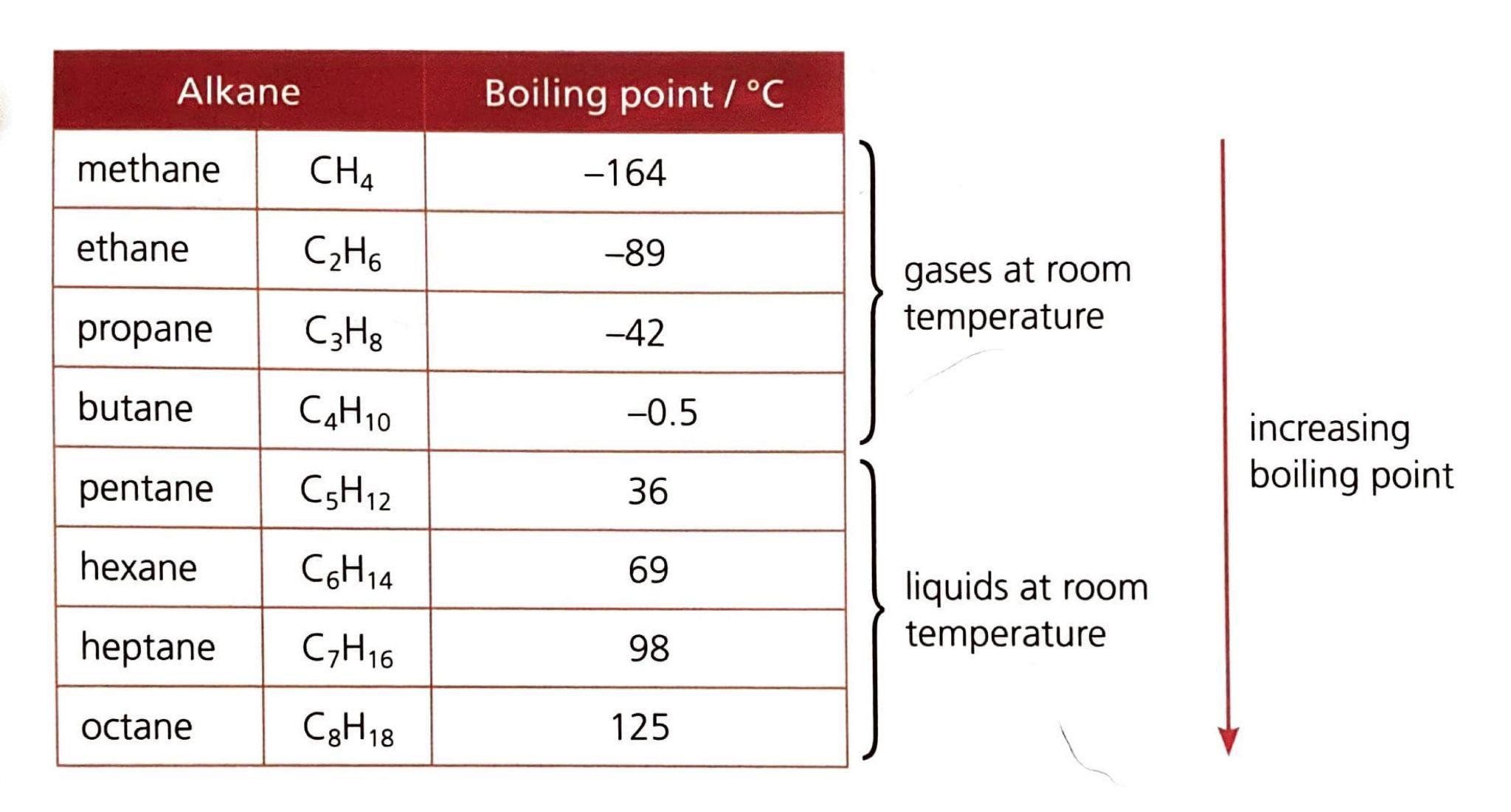
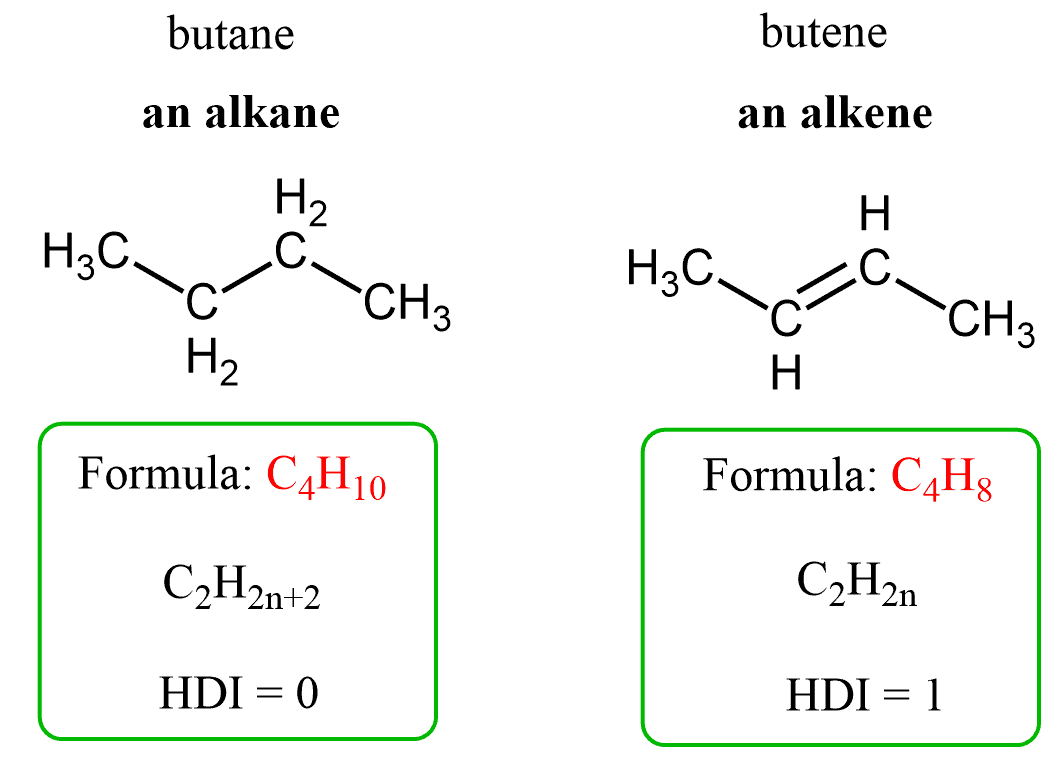
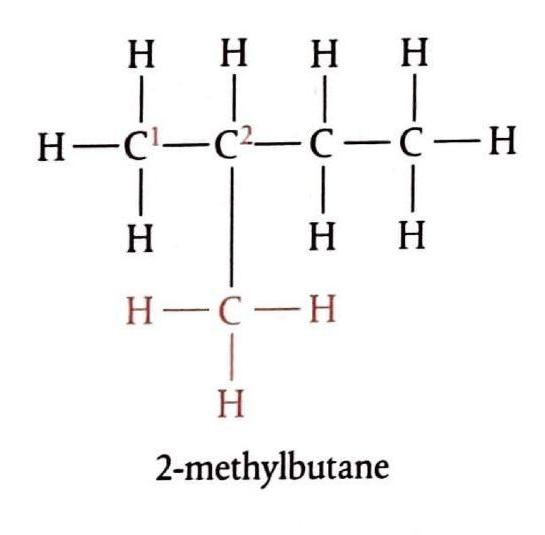
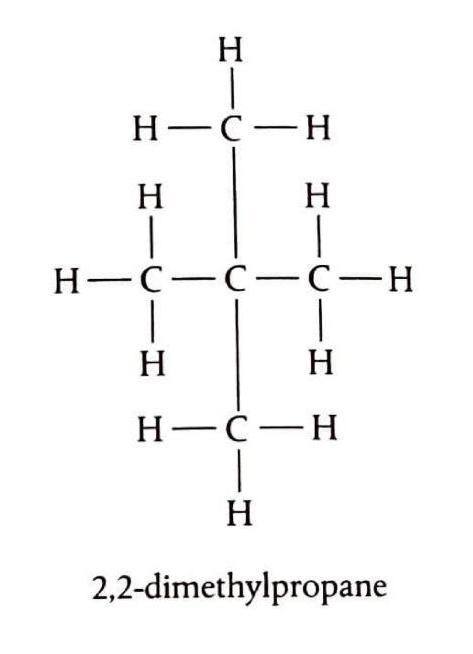
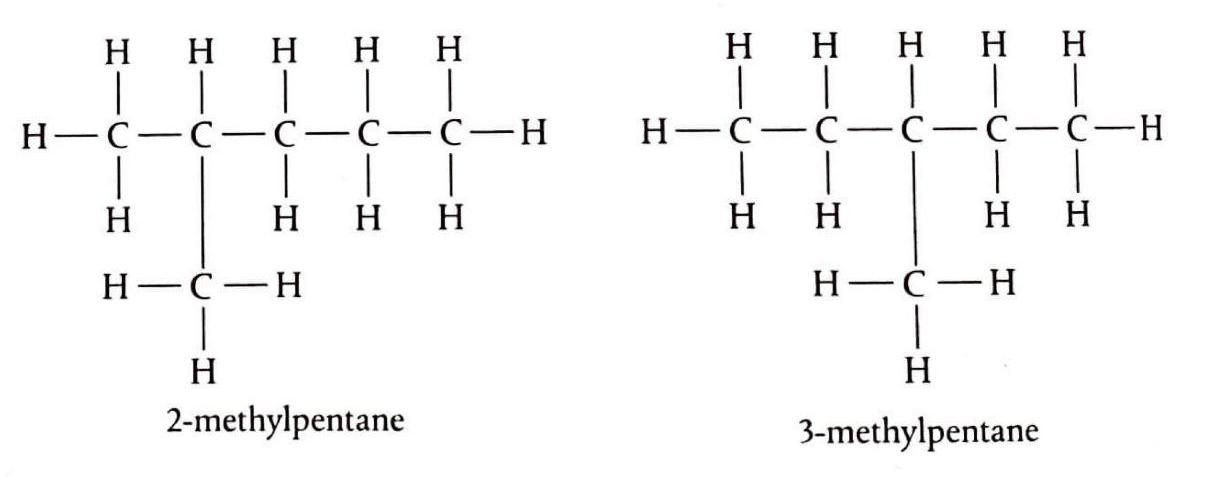
Alkanes are a class of organic compounds characterized by the general formula CnH2n+2. They are classified as saturated hydrocarbons, meaning they are composed solely of hydrogen and carbon atoms, and all carbon-carbon bonds within their structure are single bonds. This saturation distinguishes them from other hydrocarbon families. The chemical reactivity of alkanes is generally low due to the strength and non-polar nature of their constituent C-C and C-H bonds. These bonds possess significant bond enthalpies, specifically 348 kJ/mol for C-C and 412 kJ/mol for C-H bonds, requiring a substantial energy input to break them. Consequently, alkanes are stable under most conditions, making them suitable for safe storage, transport, and compression. Their non-polar character also renders them resistant to attack by many common reactants.
Combustion of Alkanes
Alkanes are widely utilized as fuels because their combustion releases considerable amounts of energy. When hydrocarbons like alkanes burn in the presence of oxygen, they typically produce carbon dioxide (CO2) and water (H2O). This process is highly exothermic, as a large amount of energy is liberated during the formation of the strong double bonds in CO2 and the bonds in H2O. For example, the complete combustion of propane (C3H8) is represented by the equation:
C3H8 (g) + 5O2 (g) → 3CO2 (g) + 4H2O (g) with a ΔH of -2220 kJ/mol.
It is important to note that the CO2 generated from the combustion of fossil fuels, which are primarily alkanes, is a greenhouse gas contributing to global warming.
In scenarios where the oxygen supply is limited, incomplete combustion occurs, leading to the formation of carbon monoxide (CO) and water. For instance, with limited oxygen, propane combustion can yield: 2C3H8 (g) + 7O2 (g) → 6CO (g) + 8H2O (g). Carbon monoxide is a toxic gas that interferes with the blood's ability to transport oxygen, posing a significant health risk, especially in areas with high traffic or inadequate ventilation. In extremely oxygen-deficient conditions, elemental carbon (soot) may be produced, as shown by: C3H8 (g) + 2O2 (g) → 3C (g) + 4H2O (g).
These unburned carbon particles negatively impact the respiratory system and contribute to smog and global dimming, which is a decrease in the amount of direct solar irradiance reaching Earth's surface.
Halogenation of Alkanes via Free Radical Substitution
Alkanes undergo substitution reactions, specifically halogenation, where a halogen atom replaces a hydrogen atom in the alkane molecule. Examples include the reaction of methane with chlorine to form chloromethane (CH4 (g) + Cl2 (g) → CH3Cl (g) + HCl (g)) and ethane with bromine to form bromoethane (C2H6 (g) + Br2 (g) → C2H5Br (g) + HBr (g)). This process occurs in three distinct stages: initiation, propagation, and termination, and involves free radicals.
Initiation: Homolytic Fission of Halogen Molecules
The initiation step involves the breaking of the diatomic halogen bond, typically by ultraviolet (UV) light. This process, known as photochemical homolytic fission, results in the symmetrical splitting of the shared electron pair, producing two free radicals. Free radicals are highly reactive species possessing an unpaired electron, but no net charge. The movement of single electrons in reaction mechanisms is depicted using 'fish-hook' (single-sided, curly) arrows. These highly reactive free radicals then initiate a chain reaction that ultimately yields a mixture of products, including halogenoalkanes. The entire sequence of these reaction steps is referred to as the reaction mechanism.
Propagation: Chain Reactions Involving Free Radicals
In the propagation stage, free radicals are both consumed and generated, allowing the reaction to continue in a chain-like manner. For instance, a chlorine radical can react with methane, leading to a series of steps that ultimately produce dichloromethane. This continuous cycle of free radical formation and consumption sustains the overall reaction.
Termination: Removal of Free Radicals
Termination reactions bring the chain reaction to an end by removing free radicals from the system. This occurs when two free radicals combine, pairing up their unpaired electrons. Numerous possible termination steps can occur, leading to a variety of products in the reaction mixture.
Alkenes: Structure and Addition Reactions
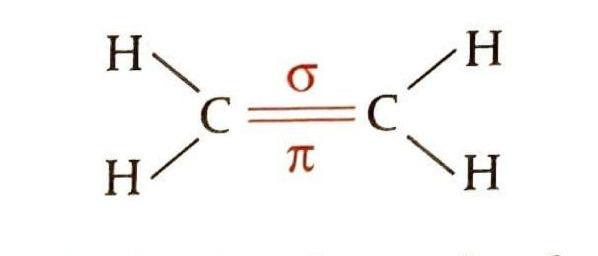
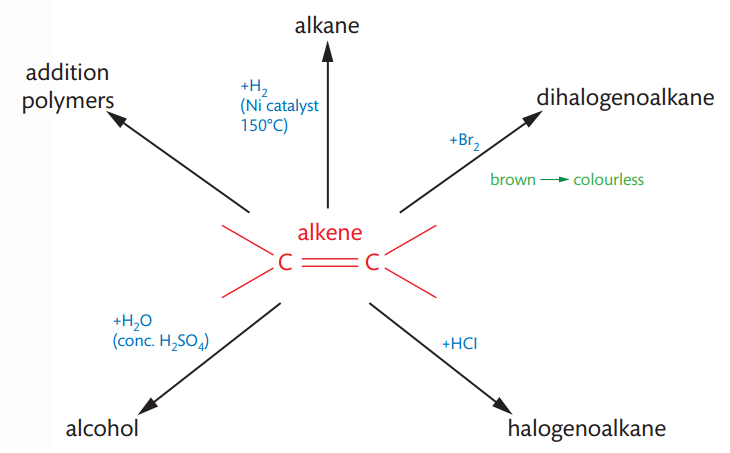
Alkenes are unsaturated hydrocarbons with the general formula CnH2n. Their defining feature is the presence of a carbon-carbon double bond. This double bond consists of one strong sigma (σ) bond and one weaker pi (π) bond. The carbon atoms involved in the double bond are sp2 hybridized, resulting in bond angles of approximately 120 degrees. The π bond in alkenes is relatively easily broken, creating two new bonding positions on the carbon atoms, which makes alkenes highly reactive towards addition reactions. These reactions convert the unsaturated alkene into a saturated product.
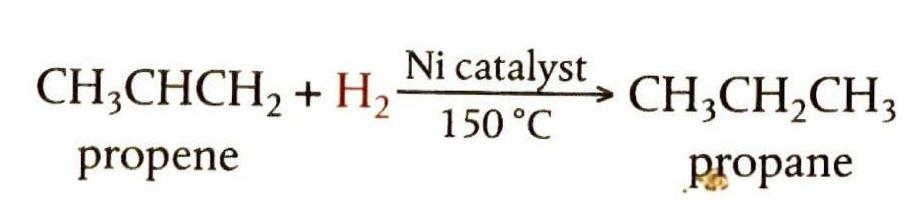
Addition of Hydrogen (Hydrogenation)
Alkenes react with hydrogen gas in a process called hydrogenation to form alkanes. This reaction typically requires a nickel catalyst and a temperature of about 150°C. During hydrogenation, the double bond is broken, and hydrogen atoms add across the carbon atoms, converting the unsaturated alkene into a saturated alkane. This process is industrially significant, particularly in the margarine industry, where unsaturated oils are hydrogenated to produce more saturated compounds with higher melting points.
Addition of Halogens
Alkenes readily react with halogens (e.g., bromine, chlorine) to form dihalogeno compounds. These reactions are typically fast and occur at room temperature. A characteristic observation is the decolorization of the reacting halogen, such as the red-brown color of bromine water disappearing. Since two halogen atoms are added to the product, the naming convention requires assigning numbers to indicate their positions.
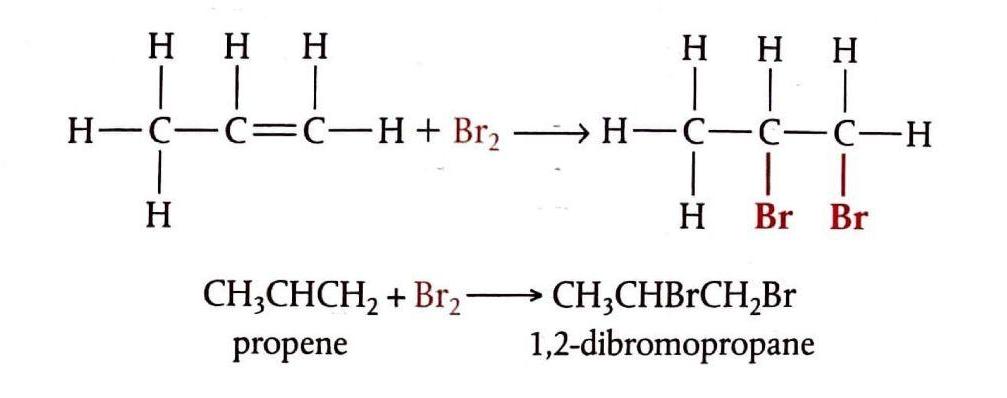
Addition of Hydrogen Halides
Alkenes react with hydrogen halides (e.g., HCl, HBr, HI) to produce halogenoalkanes. These reactions occur in solution at room temperature. The reactivity of hydrogen halides follows the order HI > HBr > HCl, with hydrogen iodide reacting most readily due to its weaker H-I bond.
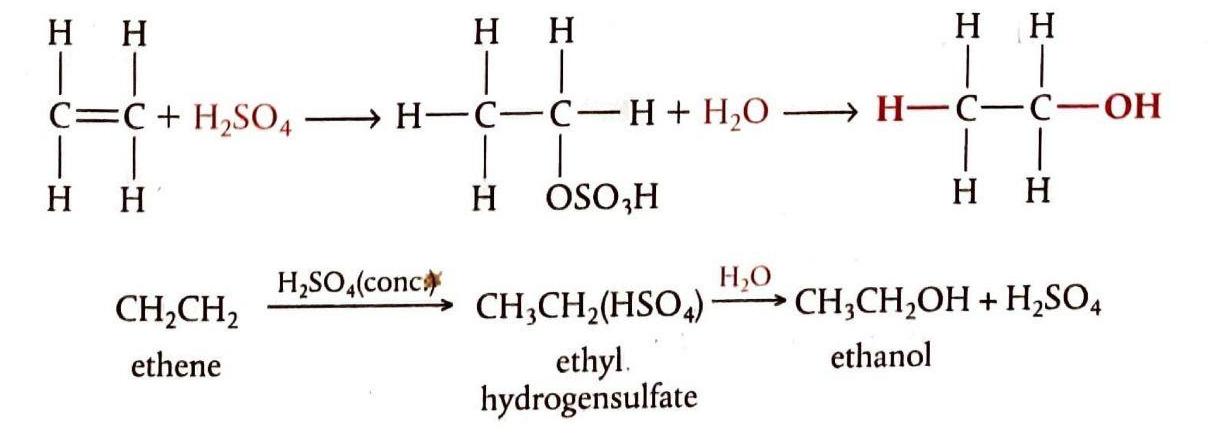
Addition of Water (Hydration)
The addition of water to an alkene, known as hydration, converts the alkene into an alcohol. This reaction is typically catalyzed by sulfuric acid. It is crucial not to confuse hydration with hydrogenation. The hydration of alkenes holds industrial importance, as it is a method for synthesizing ethanol, a widely used solvent. Modern industrial ethanol synthesis often employs catalytic hydration of ethene over a phosphoric acid catalyst absorbed on silica.
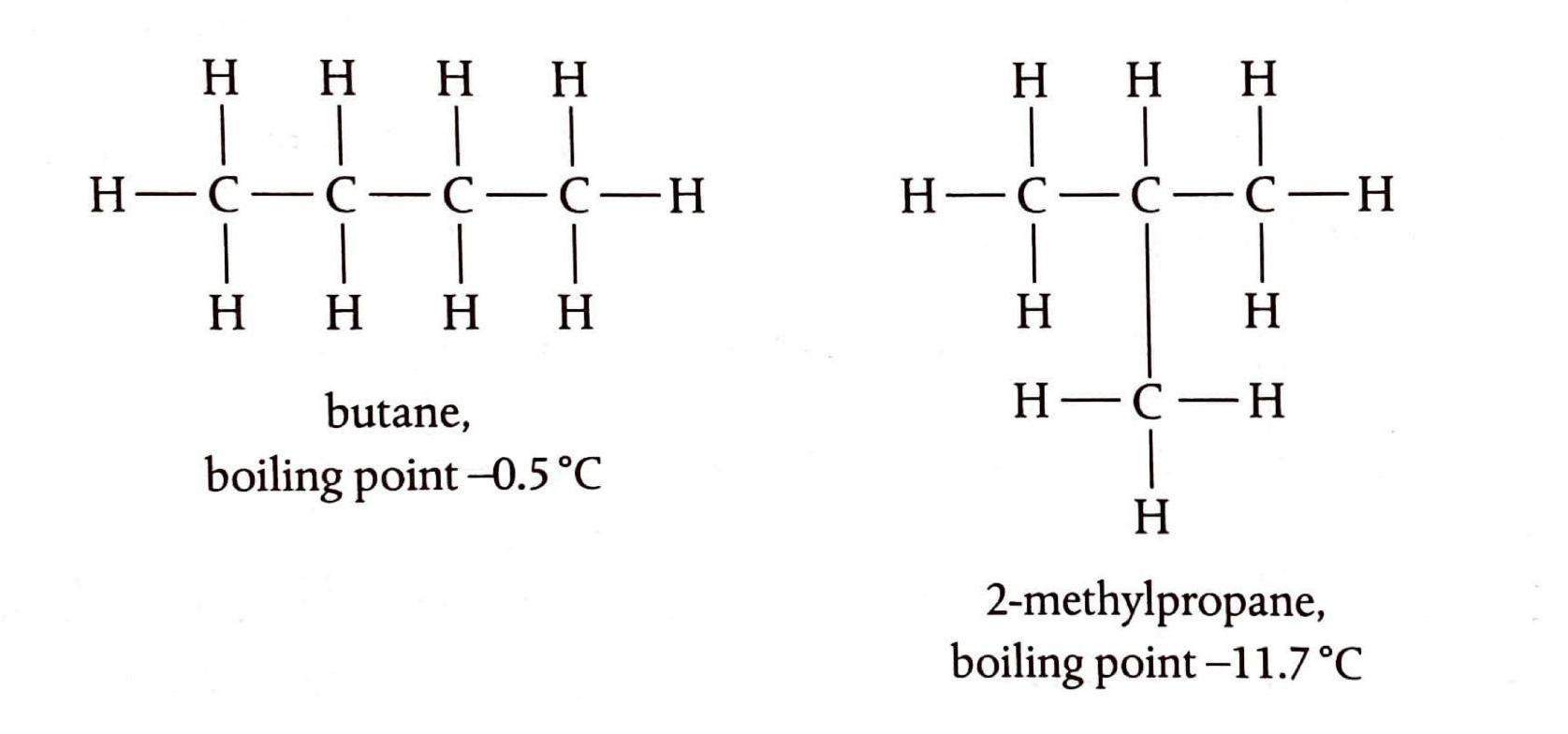
Distinguishing Alkanes and Alkenes
There are two primary observable differences that can be used to distinguish between alkanes and alkenes. Firstly, alkenes readily decolorize bromine water at room temperature without the need for UV light. If both an alkene and an alkane are shaken with red-brown bromine water, the bromine water will immediately lose its color in the presence of the alkene, but not the alkane. Alkanes only undergo substitution reactions with halogens in the presence of UV light. Secondly, alkenes tend to burn with a dirtier, smokier flame compared to alkanes. This is attributed to their higher carbon-to-hydrogen ratio, which leads to incomplete combustion and the production of soot. Compounds containing benzene rings, being even more unsaturated, exhibit an even smokier flame. Furthermore, alkenes readily participate in addition reactions, a characteristic not shared by alkanes.
Polymerization of Alkenes
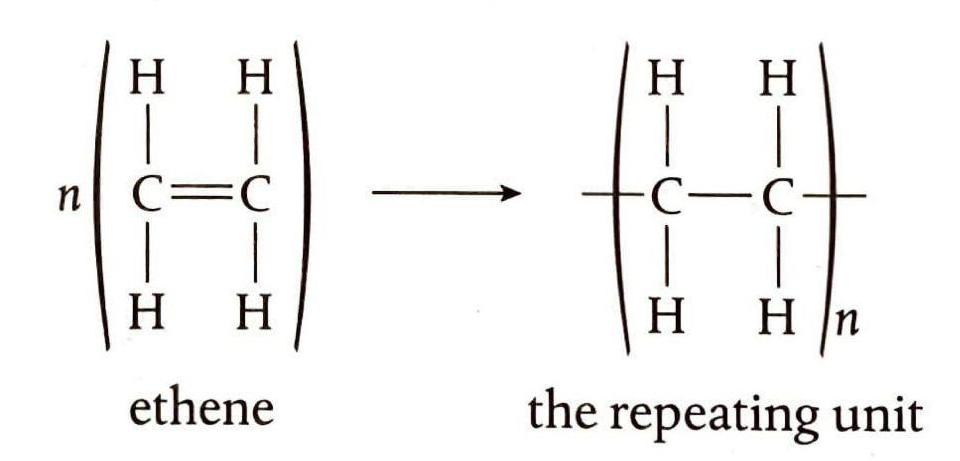
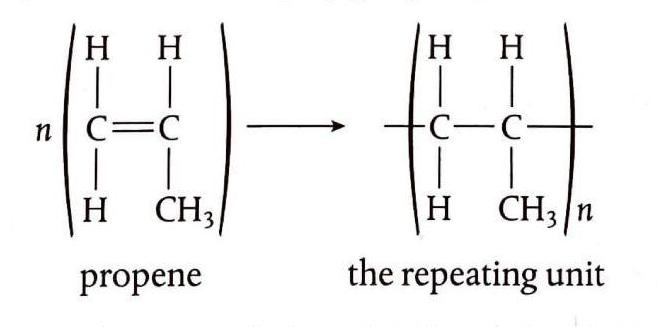
Alkenes are capable of undergoing addition reactions by breaking their double bonds, allowing them to join together to form long chains called polymers. The specific chemical nature of the monomer (the alkene used in the reaction) dictates the properties of the resulting polymer. Polymers, typically composed of thousands of monomer units, are significant products of the chemical industry, forming the basis of many plastics. During polymerization, the double bonds of the alkene monomers break to facilitate the formation of the polymer chain, as seen in the formation of poly(ethene) from ethene.
Examples of Alkene Polymers
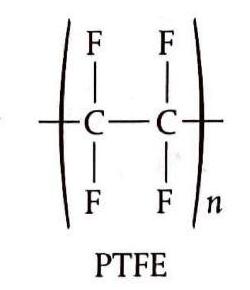
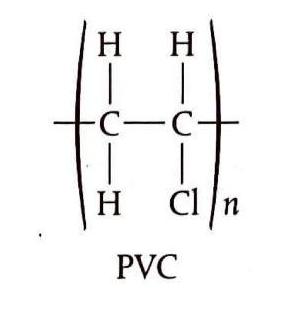
Propene polymerizes to form polypropene, commonly known as polypropylene, which finds applications in the manufacture of clothing, particularly thermal wear. Polychloroethene, or PVC (polyvinyl chloride), is another crucial plastic used extensively in construction materials, packaging, and electrical cable sheathing. However, its synthesis can produce dioxins, which are toxic and linked to various cancers. Polytetrafluoroethene, often marketed as Teflon, is known for its non-stick properties.
Disposal of Plastics
The disposal of plastics presents a major global environmental challenge due to their impermeability to water, low reactivity, and often non-biodegradable nature. As a result, plastics can persist in landfills for indefinite periods, and approximately 10% of plastics end up in the
ocean, posing significant hazards to marine life. Efforts to address
this problem include promoting more efficient recycling, developing
biodegradable plastics, and exploring the use of plastic-feeding
microorganisms. For instance, biodegradable plastics often
incorporate starch granules that absorb water and expand when
buried, breaking the plastic into smaller pieces and increasing the
surface area for bacterial digestion.
Summary of Alkene Reactions
Alcohols: Structure and Reactions
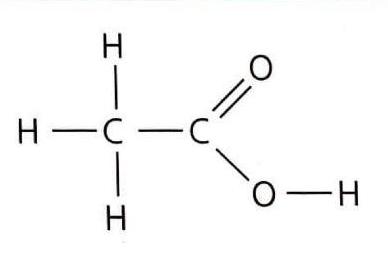
Alcohols are organic compounds characterized by the general formula CnH2n+1OH. They contain the hydroxyl (-OH) functional group. The presence of this polar -OH group significantly increases their solubility in water compared to alkanes of similar molar mass. Alcohols participate in several key reactions, including combustion, oxidation, and esterification.
Combustion of Alcohols
Alcohols react with oxygen in combustion reactions to produce carbon dioxide and water, releasing substantial amounts of energy. The amount of energy released per mole generally increases as one moves up a homologous series of alcohols due to the formation of more CO2. For example, the burning of methanol (2CH3OH (l) + 3O2 (g) → 2CO2 (g) + 4H2O (g), ΔH°c = -726.1 kJ mol-1) releases less energy than the burning of pentanol (2C5H11OH (l) + 15O2 (g) → 10CO2 (g) + 12H2O (g), ΔH°c = -3330.9 kJ mol-1). Similar to hydrocarbons, if the oxygen supply is limited, incomplete combustion will occur, leading to the production of carbon monoxide instead of carbon dioxide.
Oxidation of Alcohols
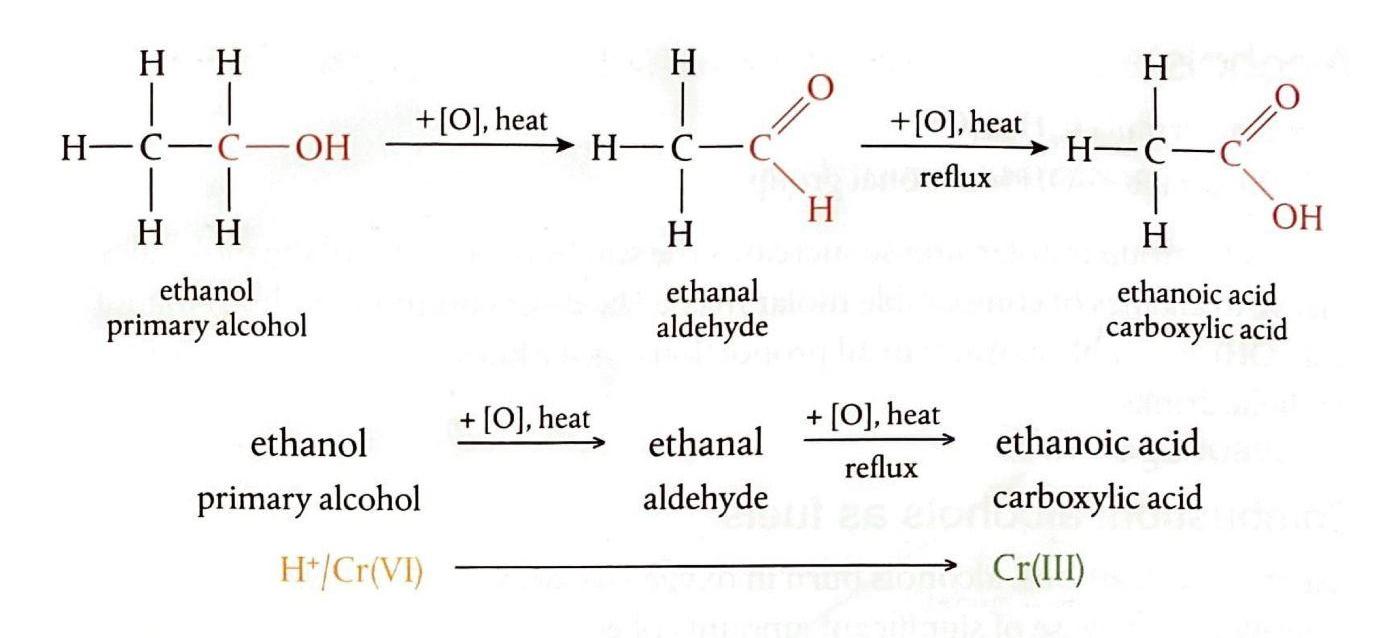
While combustion represents the complete oxidation of an alcohol molecule, it is also possible to selectively oxidize the carbon atom bearing the -OH group, leaving the rest of the carbon skeleton intact. This selective oxidation is typically achieved using acidified oxidizing agents, with acidified potassium dichromate(VI) being the most common. Potassium dichromate(VI) is bright orange and changes to green as it is reduced to chromium(III) during the reaction. In chemical equations, the oxidizing agent is often represented by +[O] placed above the reaction arrow.
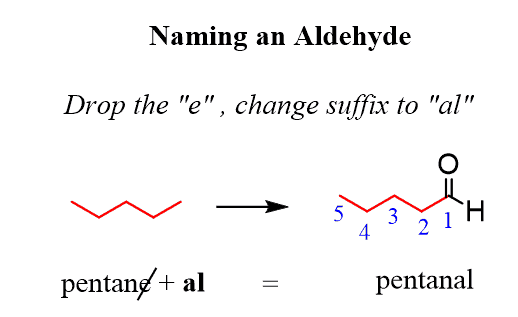
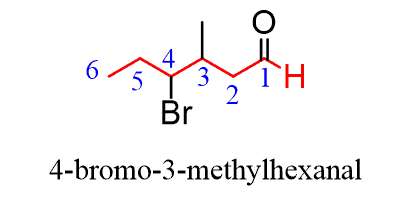
Oxidation of Primary Alcohols
Primary alcohols undergo a two-step oxidation process. The first step yields an aldehyde, and if the reaction proceeds further, the aldehyde is then oxidized to a carboxylic acid.
Separating Products of Alcohol Oxidation
Distillation is a crucial technique for separating the components of alcohol oxidation mixtures, as it exploits differences in boiling points. Aldehydes, lacking hydrogen bonding, have lower boiling points than alcohols and carboxylic acids, allowing them to be separated by distillation. To obtain the carboxylic acid, the aldehyde is kept in contact with the oxidizing agent for an extended period, often using a reflux apparatus. Refluxing involves continuously heating the reaction mixture while condensing and returning any volatile vapors to the reaction vessel, ensuring that volatile components remain in the reaction long enough for the reaction to go to completion.
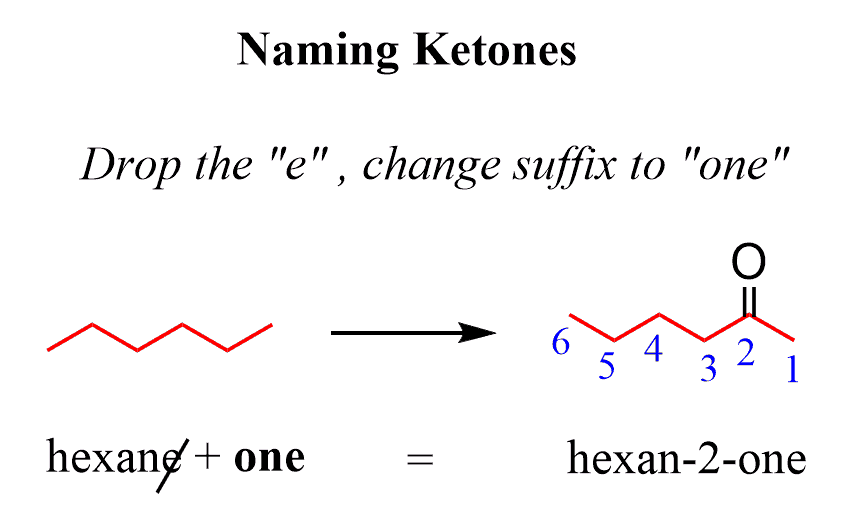
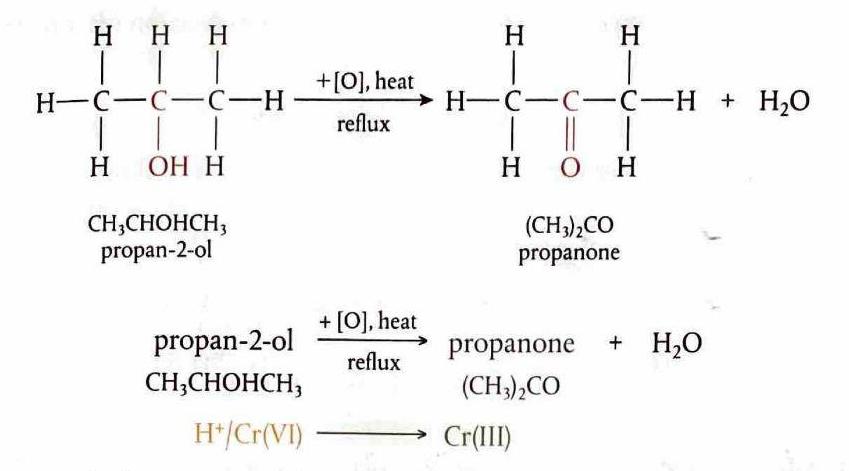
Oxidation of Secondary Alcohols
Secondary alcohols are oxidized to ketones. This reaction is analogous to the oxidation of primary alcohols, but due to the structure of secondary alcohols, the oxidation stops at the ketone stage.
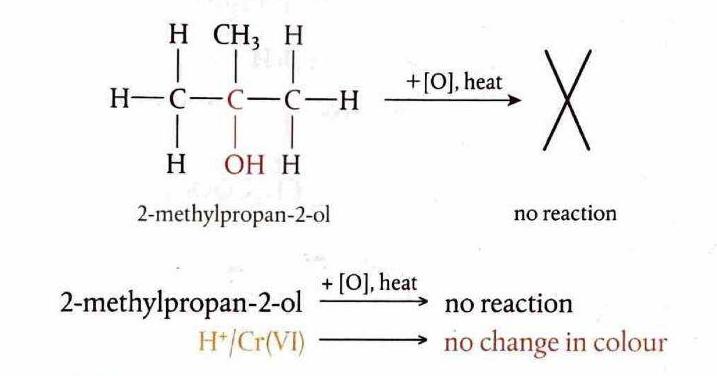
Oxidation of Tertiary Alcohols
Tertiary alcohols generally do not undergo oxidation under comparable conditions. This is because the carbon atom bonded to the hydroxyl group is also bonded to three other carbon atoms, meaning there are no hydrogen atoms directly attached to this carbon that can be removed during oxidation.
Summary of Alcohol Oxidation
In summary, oxidation with potassium dichromate(VI) solution effectively oxidizes primary and secondary alcohols, but not tertiary alcohols. The characteristic color change from orange Cr(VI) to green Cr(III) serves as an indicator of the reaction. Primary alcohols can be oxidized to carboxylic acids, secondary alcohols to ketones, while tertiary alcohols show no reaction.
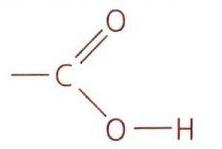
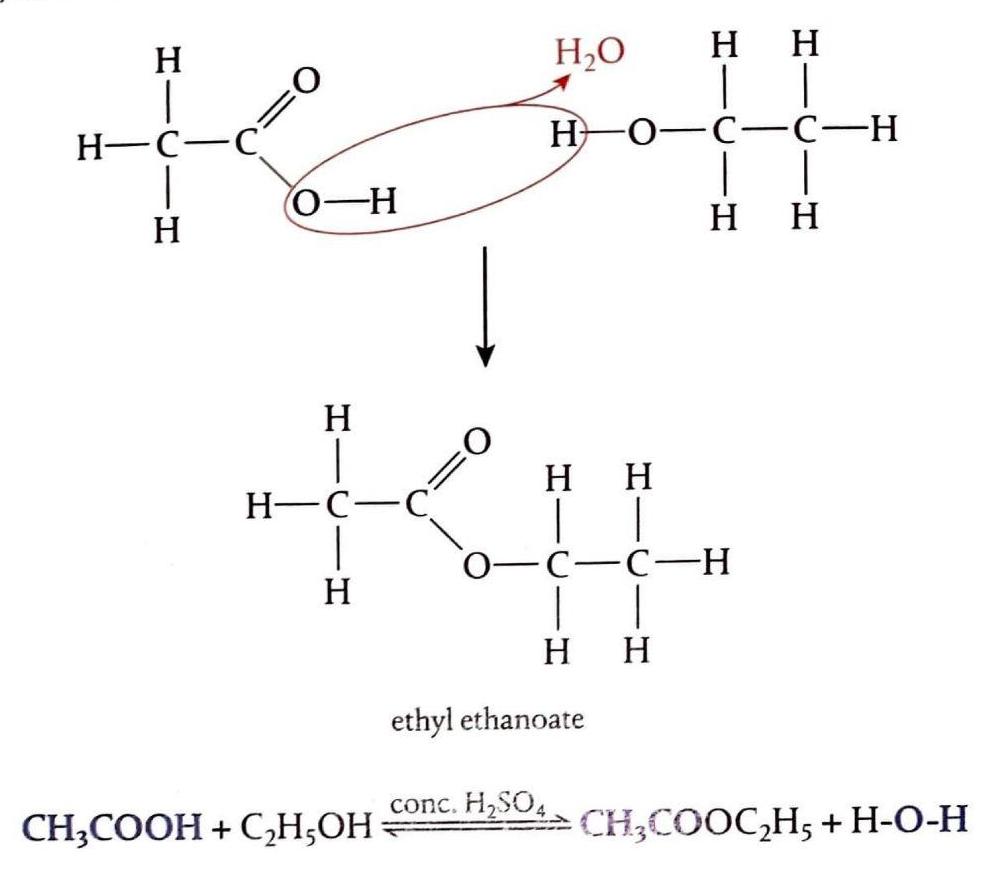
Esterification of Alcohols
Esterification is a condensation reaction where an alcohol reacts with a carboxylic acid to form an ester and typically water. The name of the ester is derived from the alkyl portion of the alcohol and the name of the acid salt. For example, the reaction between ethanol and ethanoic acid produces ethyl ethanoate. This reaction is an equilibrium process and requires warming the mixture of alcohol and carboxylic acid in the presence of concentrated sulfuric acid, which acts as a catalyst. Condensation reactions involve the joining of two molecules to form a product, usually with the elimination of a small molecule like H2O, HCl, or NH3.
Properties of Esters
Many esters are known for their sweet, fruity smells and are widely used as food flavorings and in perfumes. Unlike their parent alcohols and carboxylic acids, esters lack a free -OH group, which means they cannot form hydrogen bonds with each other. This characteristic makes them more volatile and less soluble in water compared to the corresponding alcohols and acids. Naturally occurring fats and oils are also esters, specifically triglycerides, which contain three ester linkages formed from the reaction of an alcohol with three -OH groups (glycerol) and three carboxylic acids (fatty acids).
Halogenoalkanes: Structure and Nucleophilic Substitution
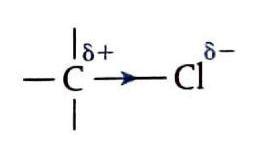
Halogenoalkanes are saturated molecules with the general formula CnH2n+1X, where X represents a halogen atom (fluorine, chlorine, bromine, or iodine) bonded to the carbon skeleton. Unlike non-polar alkanes, halogenoalkanes possess a polar bond due to the halogen atom's higher electronegativity compared to carbon. This electronegativity difference causes the halogen to exert a stronger pull on the shared electrons in the carbon-halogen bond, resulting in a partial negative charge on the halogen and a partial positive charge on the carbon. This electron-deficient carbon is a key factor in the reactivity of halogenoalkanes, particularly their susceptibility to nucleophilic substitution reactions.
Nucleophilic Substitution Reactions
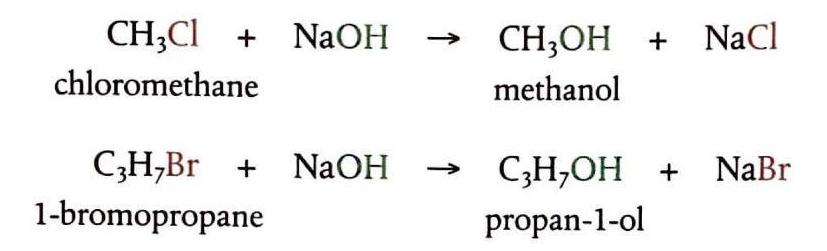
Nucleophiles are electron-rich species that are strongly attracted to electron-deficient atoms. They possess a lone pair of electrons and may also carry a negative charge, examples include H2O, OH-, NH3, and CN-. In halogenoalkanes, the nucleophiles are attracted to the electron-deficient carbon atom, leading to a reaction where the halogen atom is substituted by the nucleophile. These are known as nucleophilic substitution reactions.
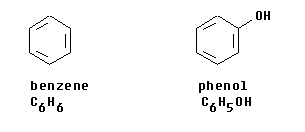
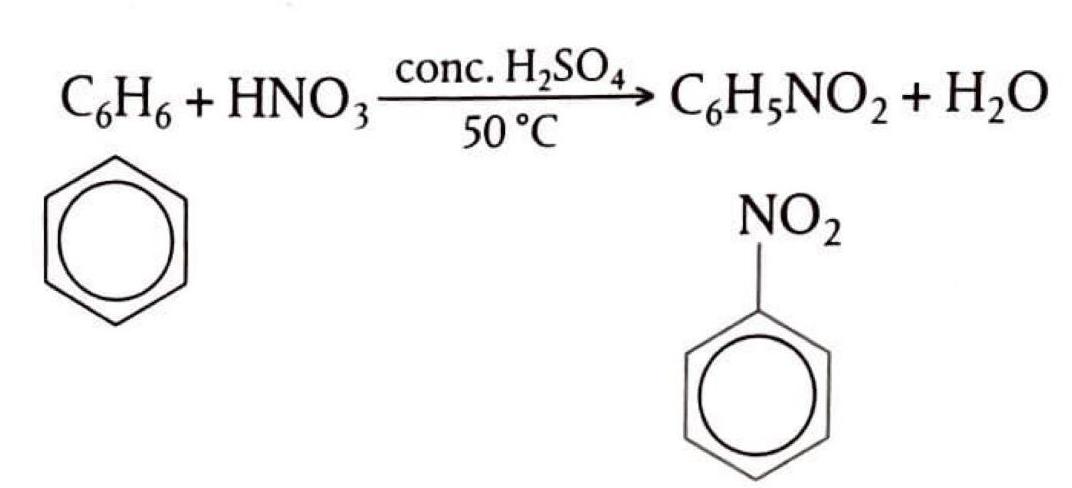
Benzene: Electrophilic Substitution
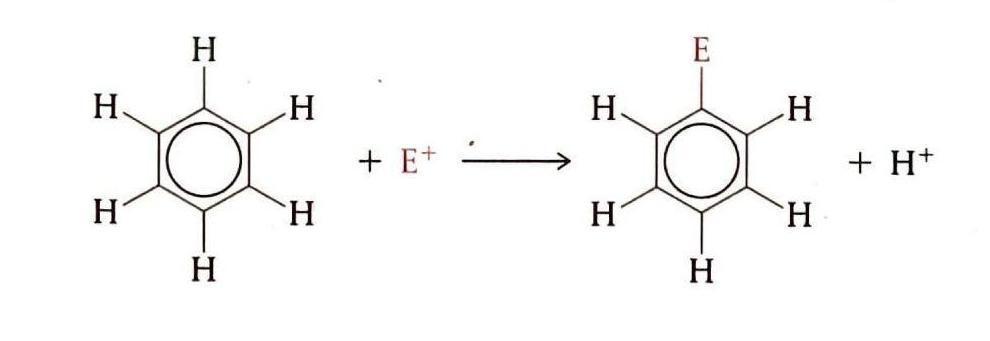
Benzene, an aromatic hydrocarbon, exhibits unique reactivity. Due to the high stability of its delocalized electron ring, addition reactions are not favored. Instead, benzene primarily undergoes substitution reactions, specifically electrophilic substitution, where one or more hydrogen atoms on the ring are replaced by an incoming group. This type of substitution occurs within the delocalized electron ring but importantly preserves the aromaticity and stability of the ring.
Electrophilic Substitution Reactions
Electrophiles are electron-deficient species, typically possessing a positive or partial positive charge. These electrophiles (E+) are attracted to the electron-rich benzene ring, initiating electrophilic substitution reactions. Examples include the reaction of benzene with NO2+ (nitration) and with halogens (halogenation).