Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Equilibrium Constant (Kc), Reaction Quotient (Q), Le Chatelier’s Principle, Haber/Contact Processes
Learning Objectives
Analyze reversible systems and the factors that influence the position of chemical equilibrium.
Prior Knowledge Needed
Kinetics, enthalpy changes, gas volume calculations
IB Syllabus Reference
R2.3
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
6 Lessons Available
3 Equilibrium Exam Version A edited
3 Equilibrium Test Version B Edited
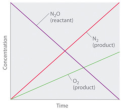
Equilibrium Graphs_Factors that affect Equilibrium
ICE Tables
Relationship between Gibbs free energy and Equilibrium Constant
The Equilibrium Constant and Reaction Quotient
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
ICE table
initial, change, equilibrium, when you have initial concentrations and want to find equilibrium concentrations, can use given eq. molarity to find answer or use given Keq
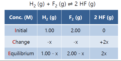
initial concentration
the % concentration of each substrate prior to mixing with the other substrates in solution
equilibrium concentration
The concentration of a substance (usually expressed as molarity) in a system that has reached the equilibrium state.

Kc<<1
reactant favored, you can assume change in reactant concentrations is zero if it is 10^-3 or less

26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments (HL)
Paper 1A: MCQ Practice (HL)
PDF
Paper 1B: Data & Experiments (HL)
PDF
Paper 2: Short Response (HL)
PDF
Official Markscheme (HL)
PDF
17
Cl
35.45
Assignments & Labs Section 08