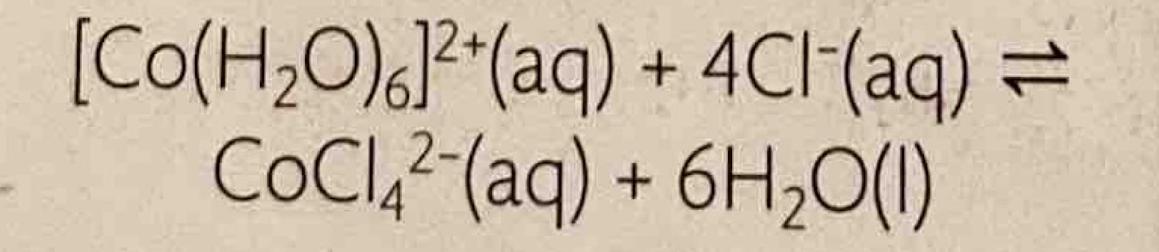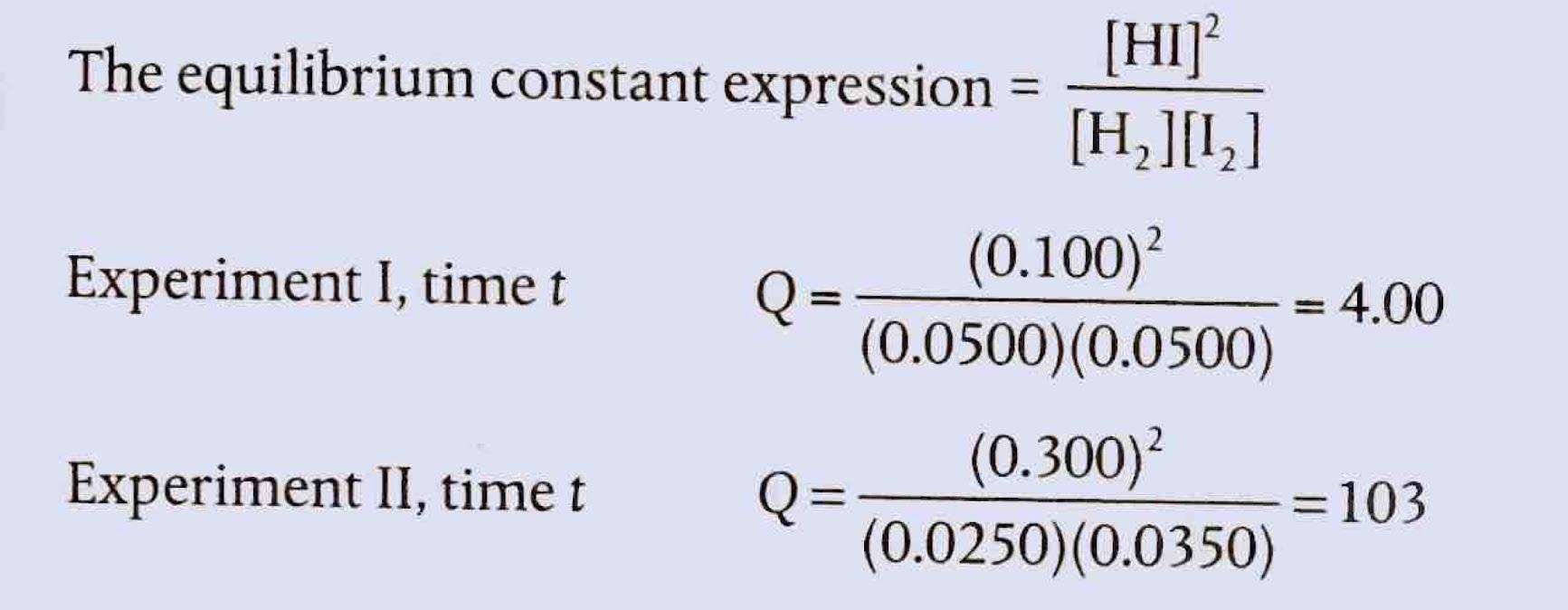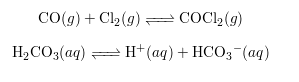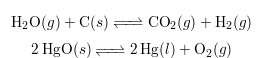
Introduction to the Equilibrium State
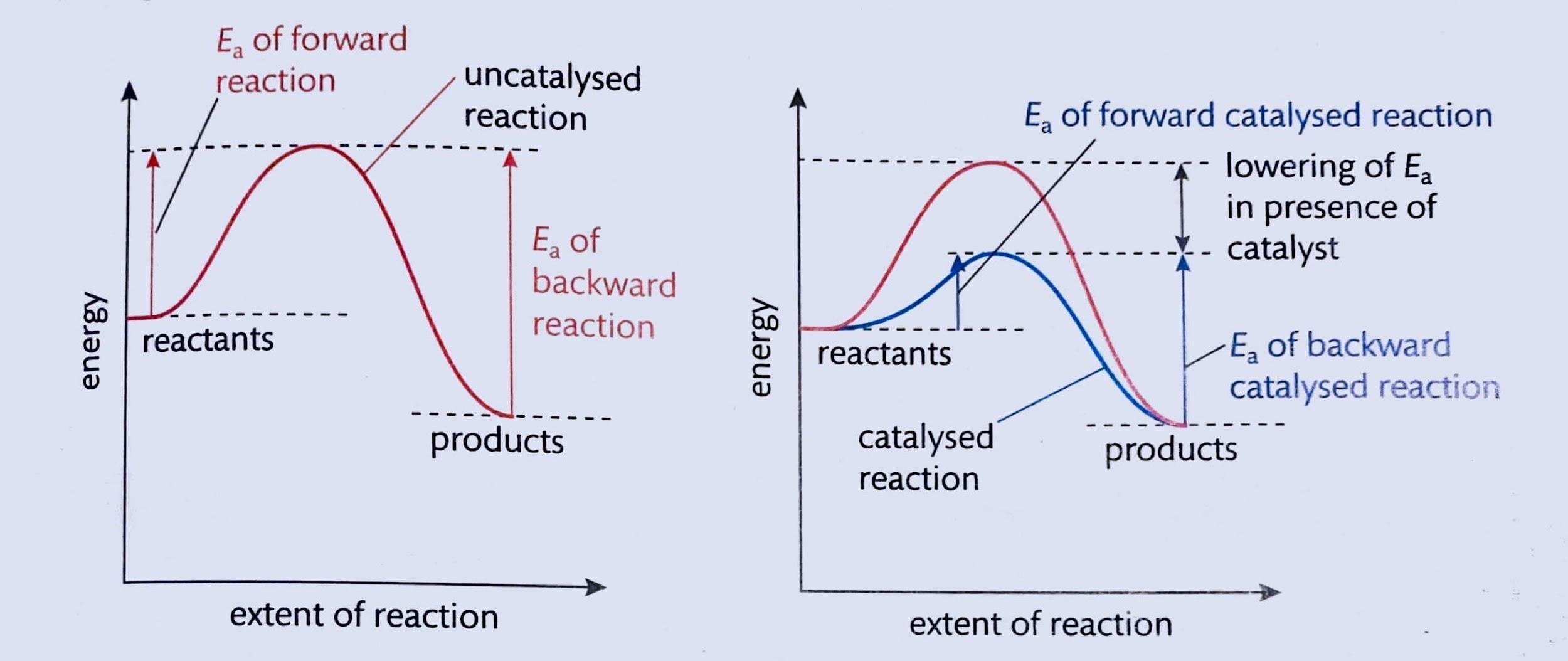
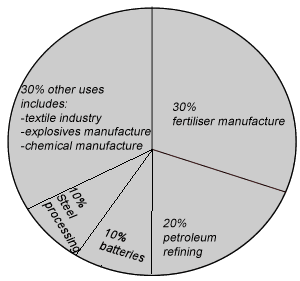
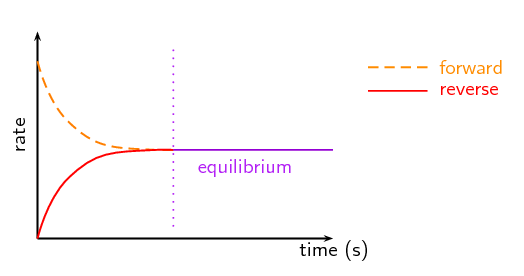
The study of chemical equilibrium is fundamental across various scientific disciplines, playing a crucial role in optimizing industrial processes to maximize product yield. Beyond industrial applications, understanding equilibrium is vital in biochemical contexts, such as predicting the solubility of gases in blood. In environmental science, it helps explain how chemicals react in the atmosphere to form pollutants and elucidates the relationship between water vapor and precipitation under varying temperatures and pressures. Fundamentally, the equilibrium state is achieved when the rates of the forward and reverse reactions become equal.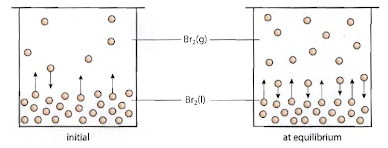
Physical Equilibrium
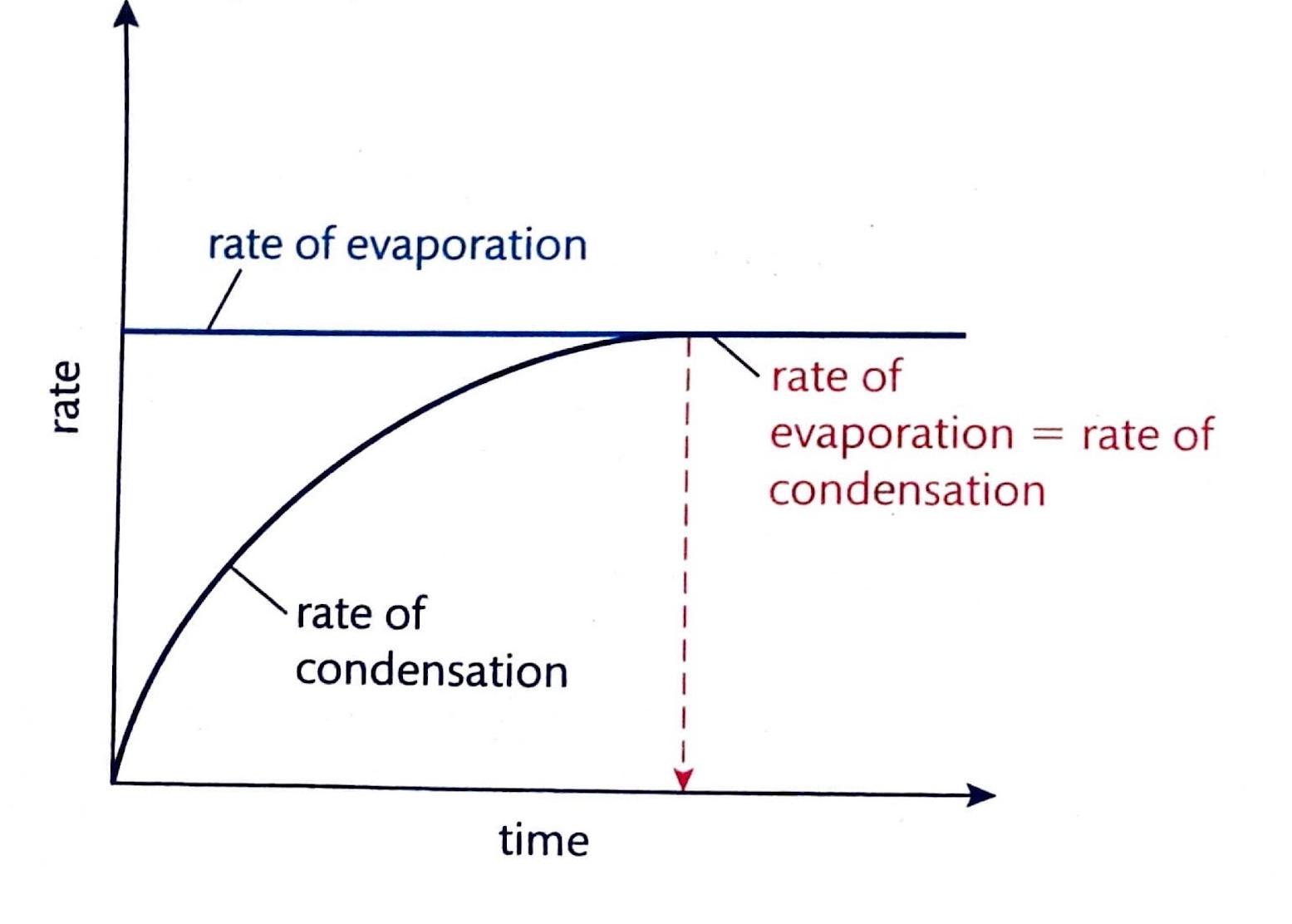
Physical equilibrium is a state that can only be observed in a closed system. This type of equilibrium occurs when two different physical states of the same substance coexist and are in balance. A common example is the equilibrium between liquid water and water vapor in a sealed container, where the rate of evaporation equals the rate of condensation.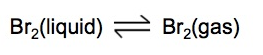
Chemical Equilibrium
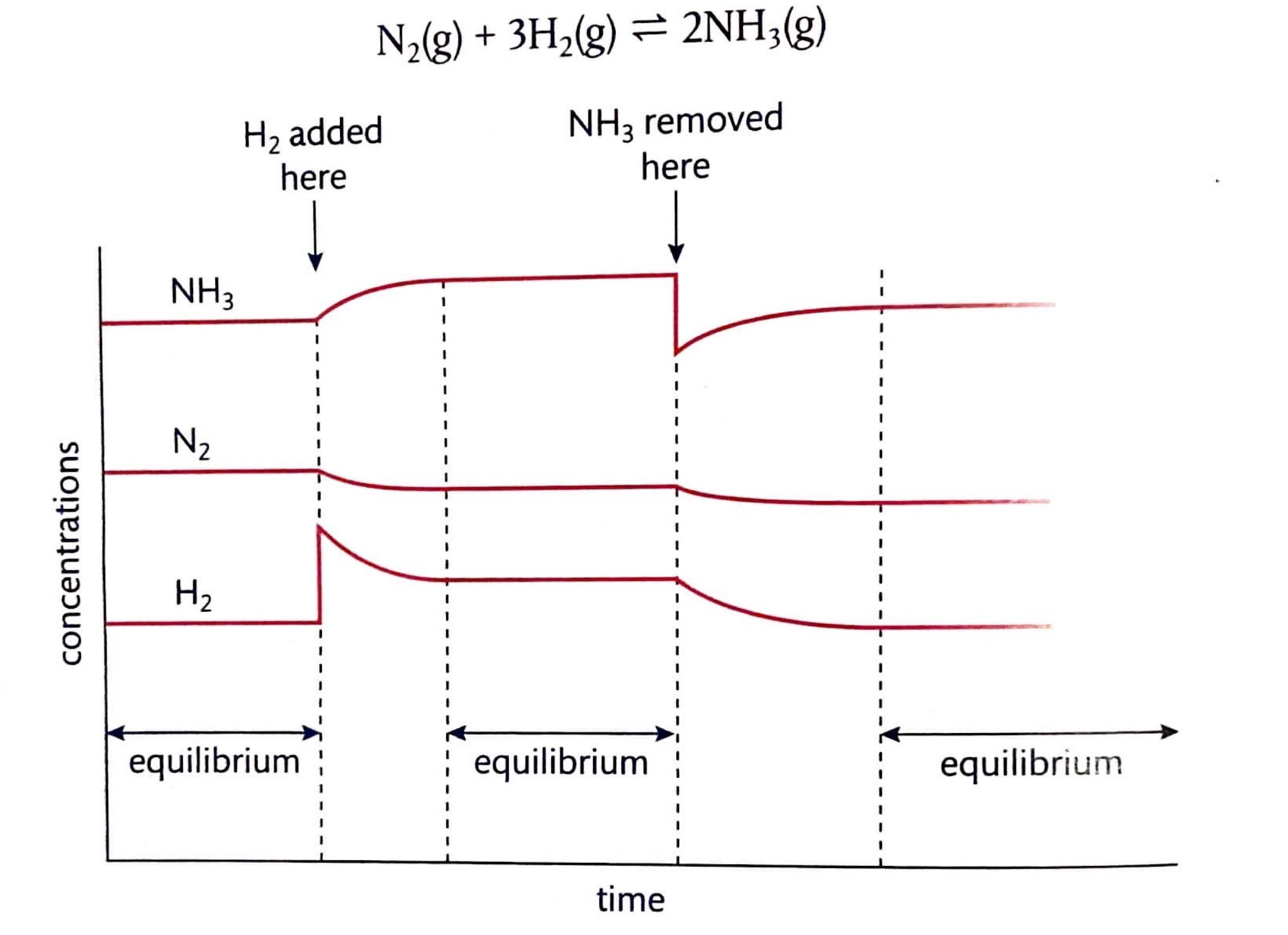
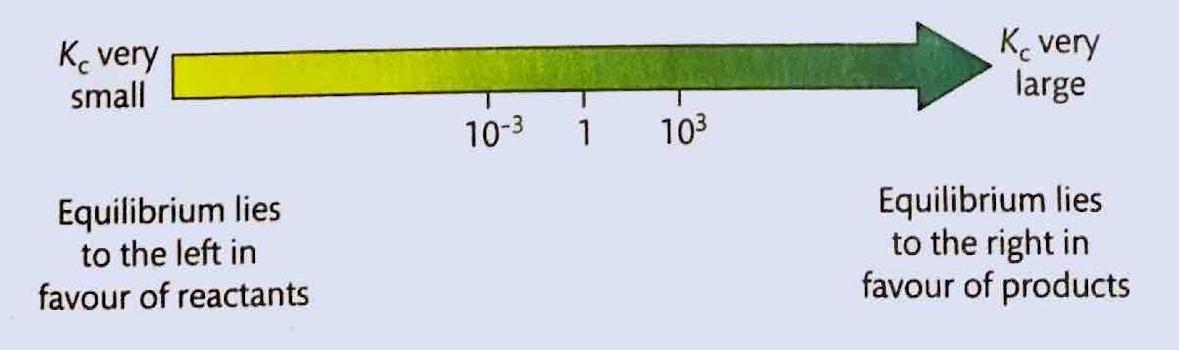
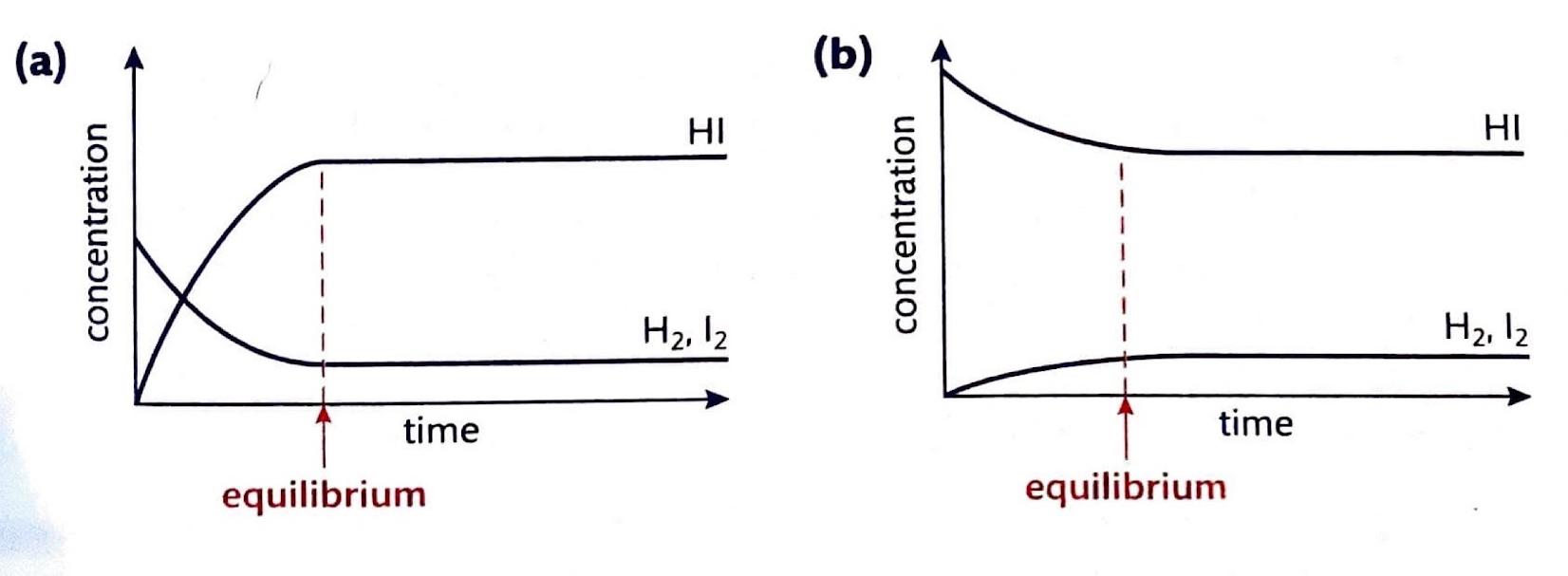
Chemical equilibrium is attained when a chemical reaction reaches a state where the concentrations of reactants and products remain constant over time. This is known as a dynamic equilibrium, meaning that while the macroscopic properties of the system appear static, the forward and reverse reactions are still occurring at equal rates. It is important to note that at equilibrium, the concentrations of reactants and products do not necessarily have to be equal; rather, they are constant. For instance, in the reaction between hydrogen and iodine to form hydrogen iodide, H2(g) + I2(g) ⇌ 2HI(g), hydrogen and hydrogen iodide are colorless, but iodine gas is purple. As the reaction proceeds towards equilibrium, the purple color of iodine will stabilize, indicating that its concentration, and thus the concentrations of the other species, has become constant.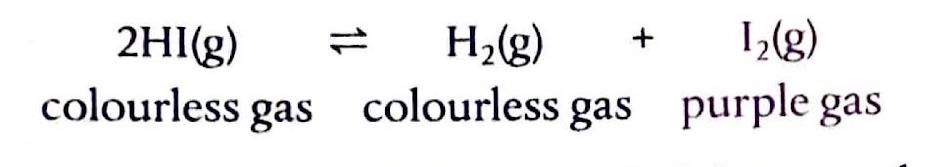

Reversible vs. Non-reversible Reactions
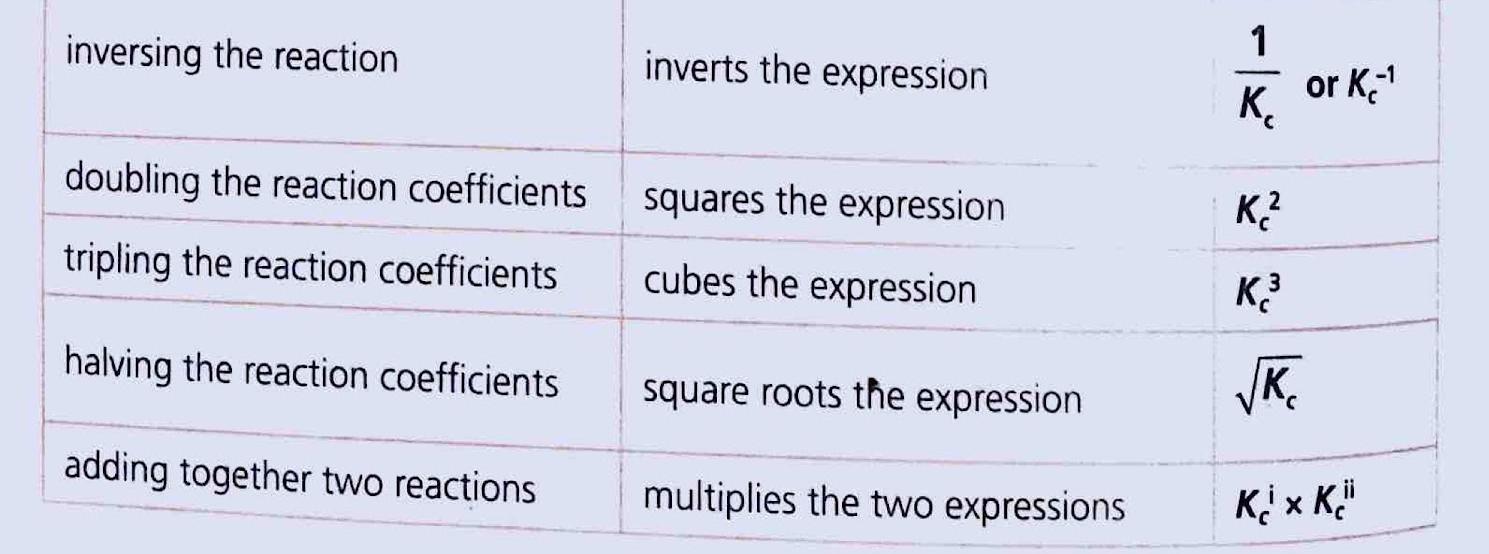
Chemical reactions can be categorized as either reversible or non-reversible, depending on whether the products can revert back to reactants under suitable conditions.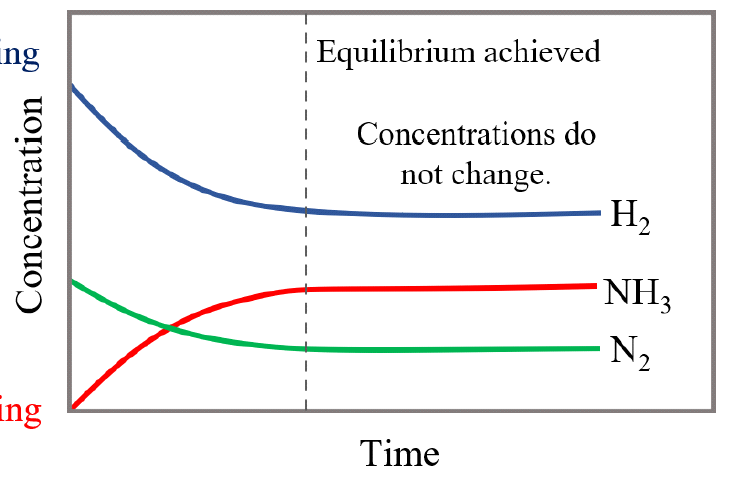
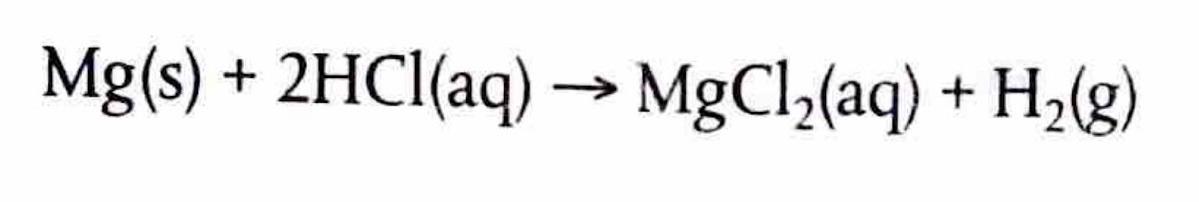
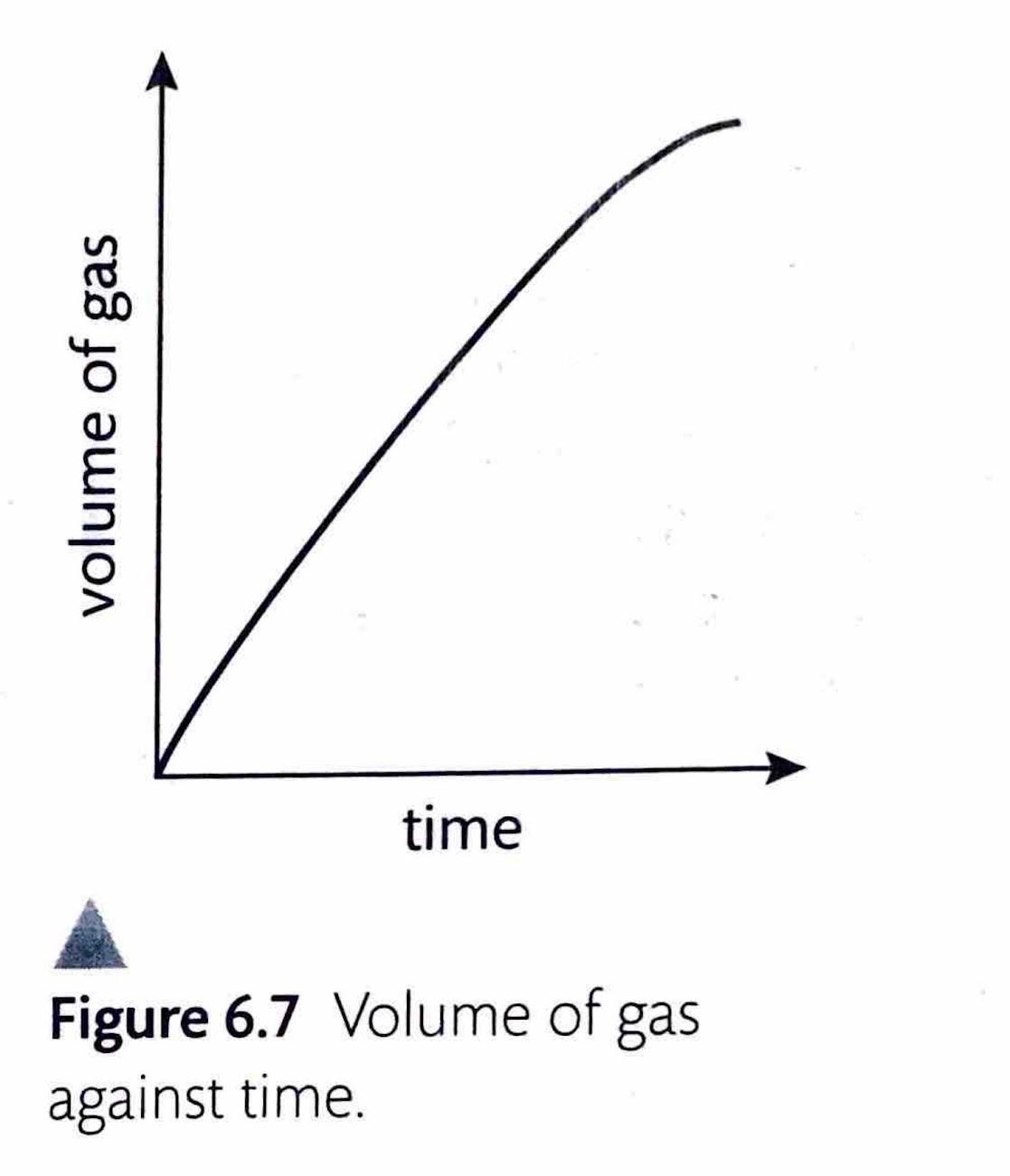
The key distinctions between these two types of reactions are summarized in the table below:
| Reversible Reaction | Non-reversible Reaction |
|---|---|
| It can be reversed under suitable conditions. | It cannot be reversed (products can not change back into reactants). |
| Both forward and reverse reactions take place simultaneously (indicated by equilibrium arrows). | Unidirectional. It proceeds only in forward direction (indicated by unidirectional reaction arrow). |
| Attains equilibrium. | Equilibrium is not attained. |
| The reactants cannot be converted completely into products. | The reactants can be converted completely into products. |
| It is relatively slow. | It is fast. |
Summary of the Equilibrium State
In summary, the equilibrium state, whether physical or chemical, represents a dynamic balance where opposing processes occur at equal rates, leading to constant macroscopic properties. This fundamental concept is critical for understanding and predicting the behavior of systems in various scientific and industrial contexts.