Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Rate Equations, Activation Energy, Maxwell-Boltzmann Distribution, Rate-Determining Step, Arrhenius Equation
Learning Objectives
Investigate dynamic chemical kinetics, including rate constant calculations and Arrhenius theory models.
Prior Knowledge Needed
Collision theory, energy profiles, stoichiometry (R1.1)
IB Syllabus Reference
R2.2
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
4 Lessons Available
Instantaneous Rates
Maxwell Boltzmann Distribution Curves
Rate Laws
Reaction Mechanisms_Arrhenius Plots
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
rate expression/law
mathematical representation relating reaction rate to changes in amount, concentration, or pressure of reactant or product species per unit time

rate constant
a constant of proportionality between the reaction rate and the concentrations of reactants that appear in the rate law
zero order rate
Rate = k; rate is constant no matter the concentration

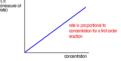
first order rate
rate=k[A]; rate is proportional to the concentration of reactant
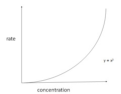
second order rate
Rate=k[A]^2; rate is exponential to the concentration of reactant
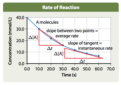
instantaneous rate
The reaction rate at a particular time as opposed to the average rate over an interval of time.
time^-1
units of first order k constant
mol^-1dm^-3s^-1
units of second order k constant
mol^-2dm^6s^-1
units of third order rate contstant
elementary step
an individual step in a reaction mechanism
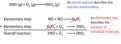
reaction mechanism
a series of elementary reactions that take place during the course of a complex reaction

molecularity
the number of species that must collide to produce the reaction indicated by that step

unimolecular
describes a reaction that involves only one particle that goes on to form products
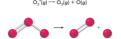
bimolecular
an elementary step in a reaction that involves two particles, either the same species or different, that collide and go on to form products
termolecular
an elementary step of a reaction in which three particles collide and go on to form products

Arrhenius plot
A plot of the natural log of the rate constant (ln k) versus the inverse of the temperature in kelvins (1/T) that yields a straight line with a slope of -Ea/R and a y-intercept of ln A.

Arrhenius constant
Indicates the frequency of collisions and the probability that collisions have proper orienta- tions
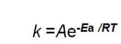
26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments (HL)
Paper 1A: MCQ Practice (HL)
PDF
Paper 1B: Data & Experiments (HL)
PDF
Paper 2: Short Response (HL)
PDF
Official Markscheme (HL)
PDF
17
Cl
35.45
Assignments & Labs Section 08