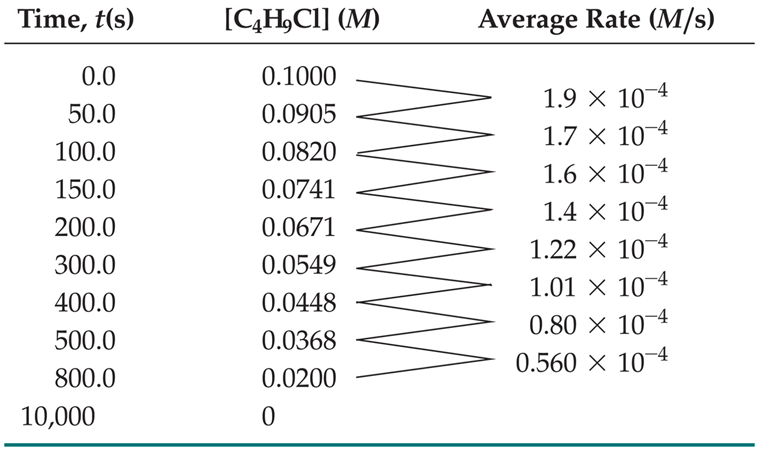
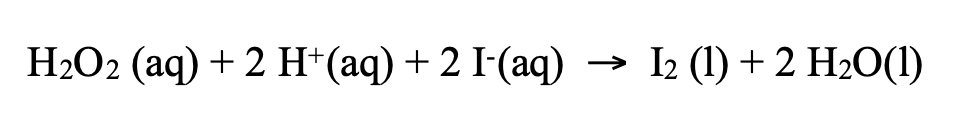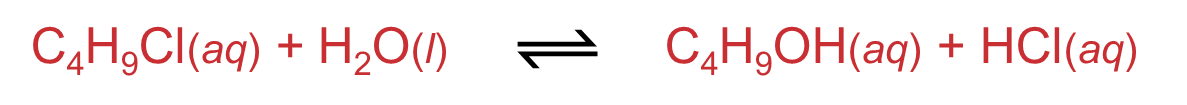Experimental Determination of Reaction Rates
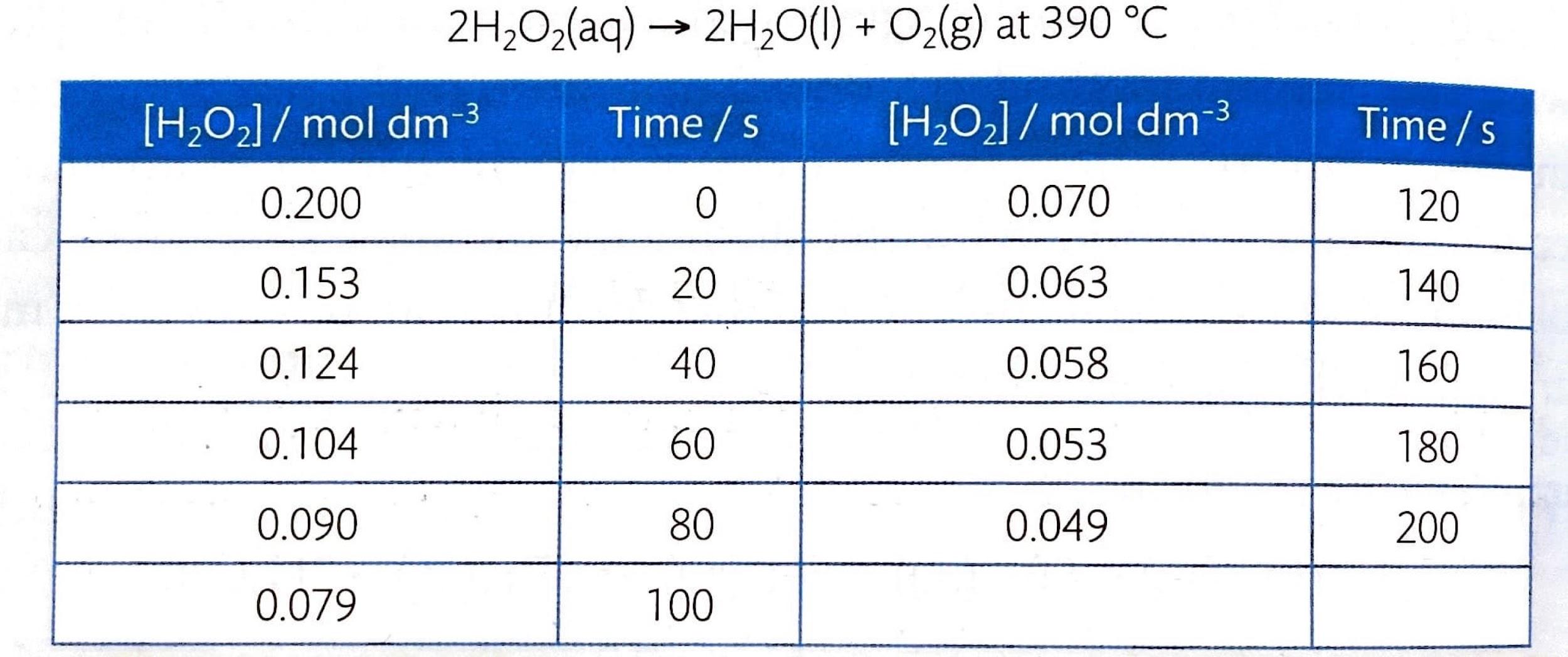
The rate of a chemical reaction can be experimentally determined by monitoring the change in concentration of either reactants or products over time. While direct measurement of concentration in mol dm
-3 is ideal, many experimental techniques rely on observing changes in other physical parameters that are directly proportional to concentration changes. These parameters can include the volume of gas produced, the mass lost from the reaction system, or changes in the color intensity of the solution. Consequently, the raw data collected may be in various units other than mol dm
-3. Modern data-logging devices are frequently employed to automate and improve the precision of these measurements.
Diverse Methods for Measuring Reaction Rates
A variety of experimental methods are available to measure reaction rates, each suited to different types of chemical reactions. These methods include monitoring the change in volume of gas produced, observing the change in mass of the reaction system, measuring the transmission of light using techniques like colorimetry or spectrophotometry, determining concentration changes through titration, assessing conductivity changes, and utilizing "clock reactions" which measure the time to a specific endpoint.
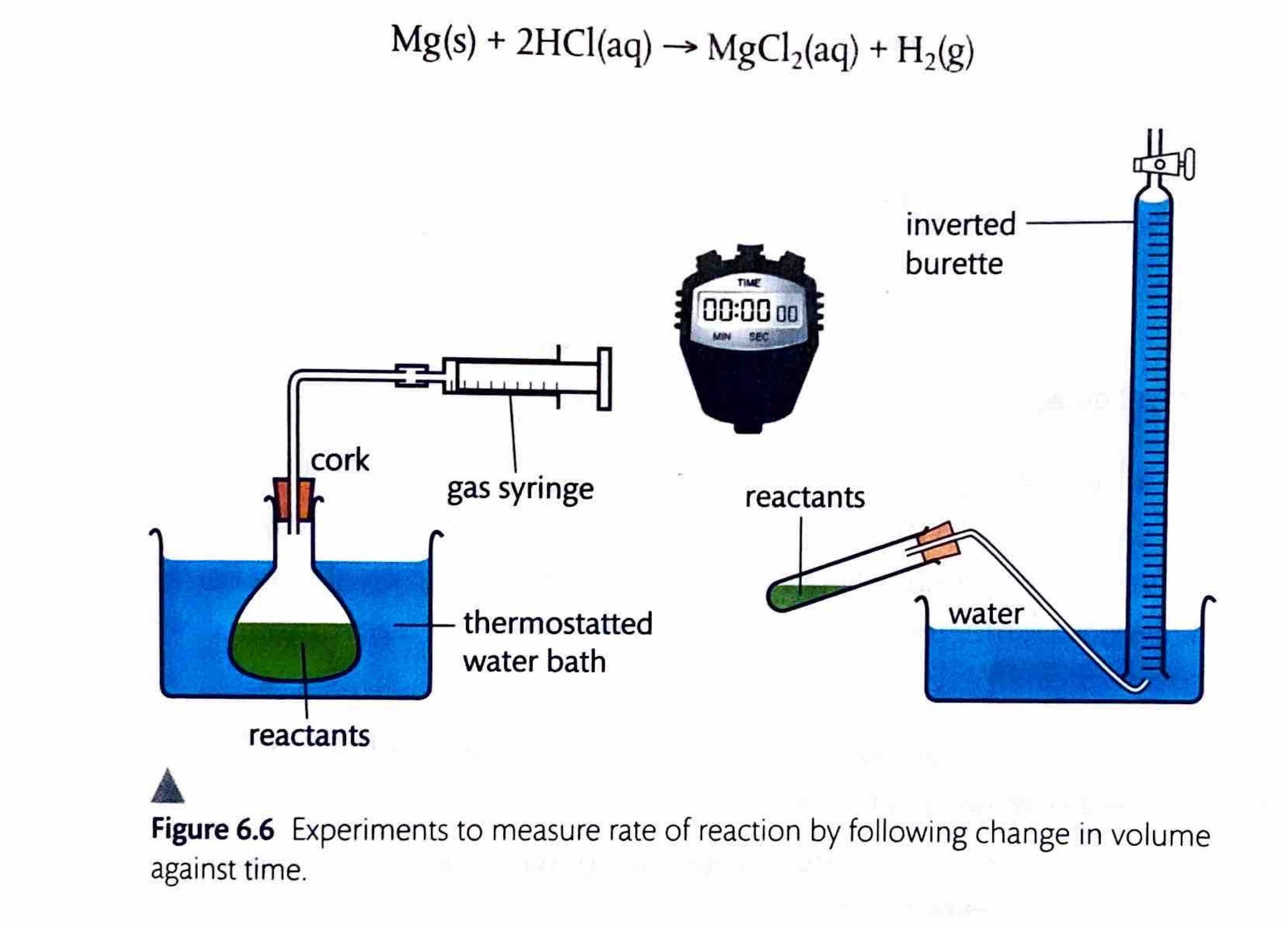
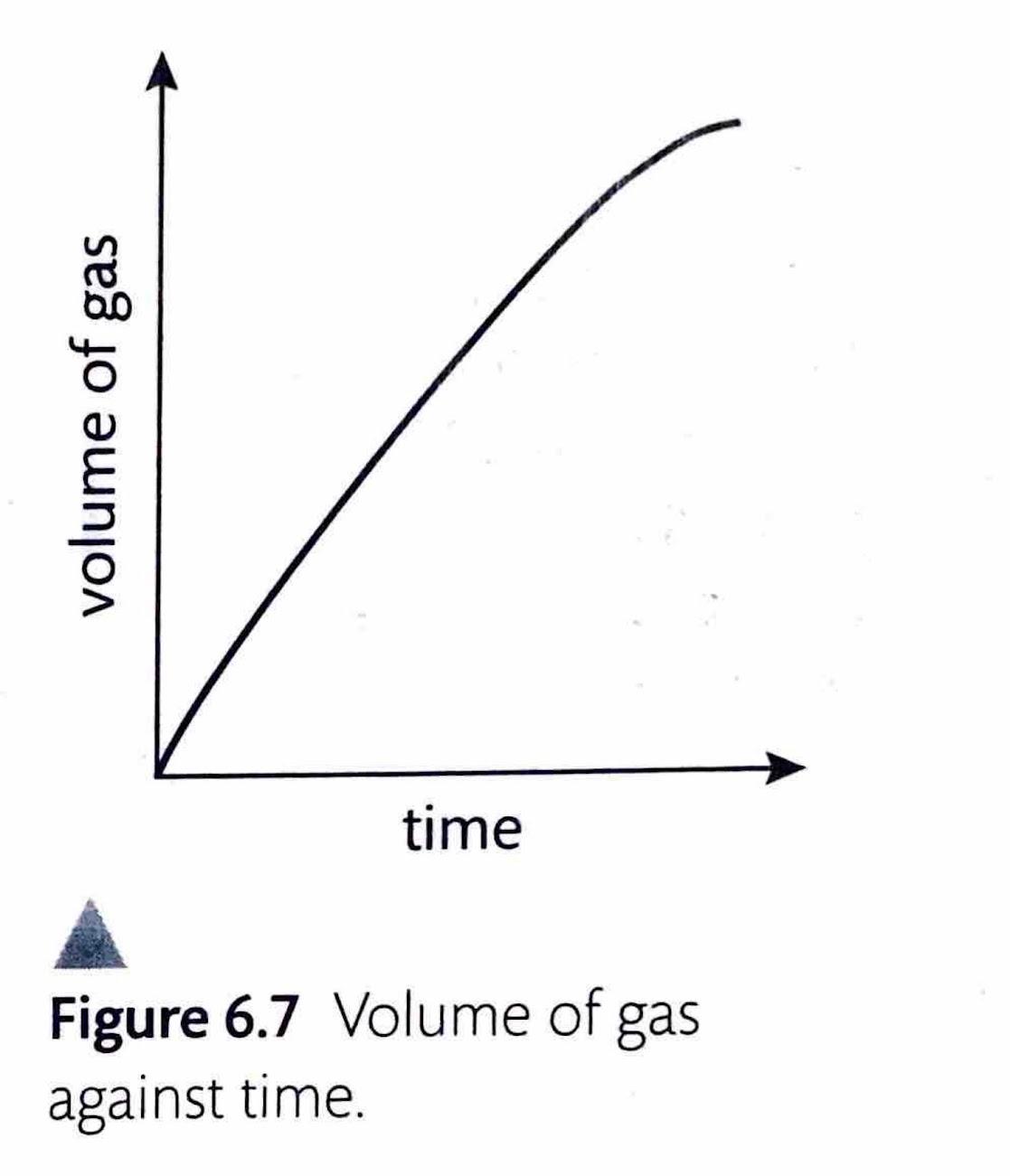
Measuring Rate by Change in Gas Volume
When one of the products of a reaction is a gas, measuring the change in its volume provides a convenient way to determine the reaction rate. A gas syringe is commonly used to collect the gas produced, allowing for direct measurement of its volume over time. Alternatively, the gas can be collected by the displacement of water, where the volume of displaced water corresponds to the volume of gas produced.
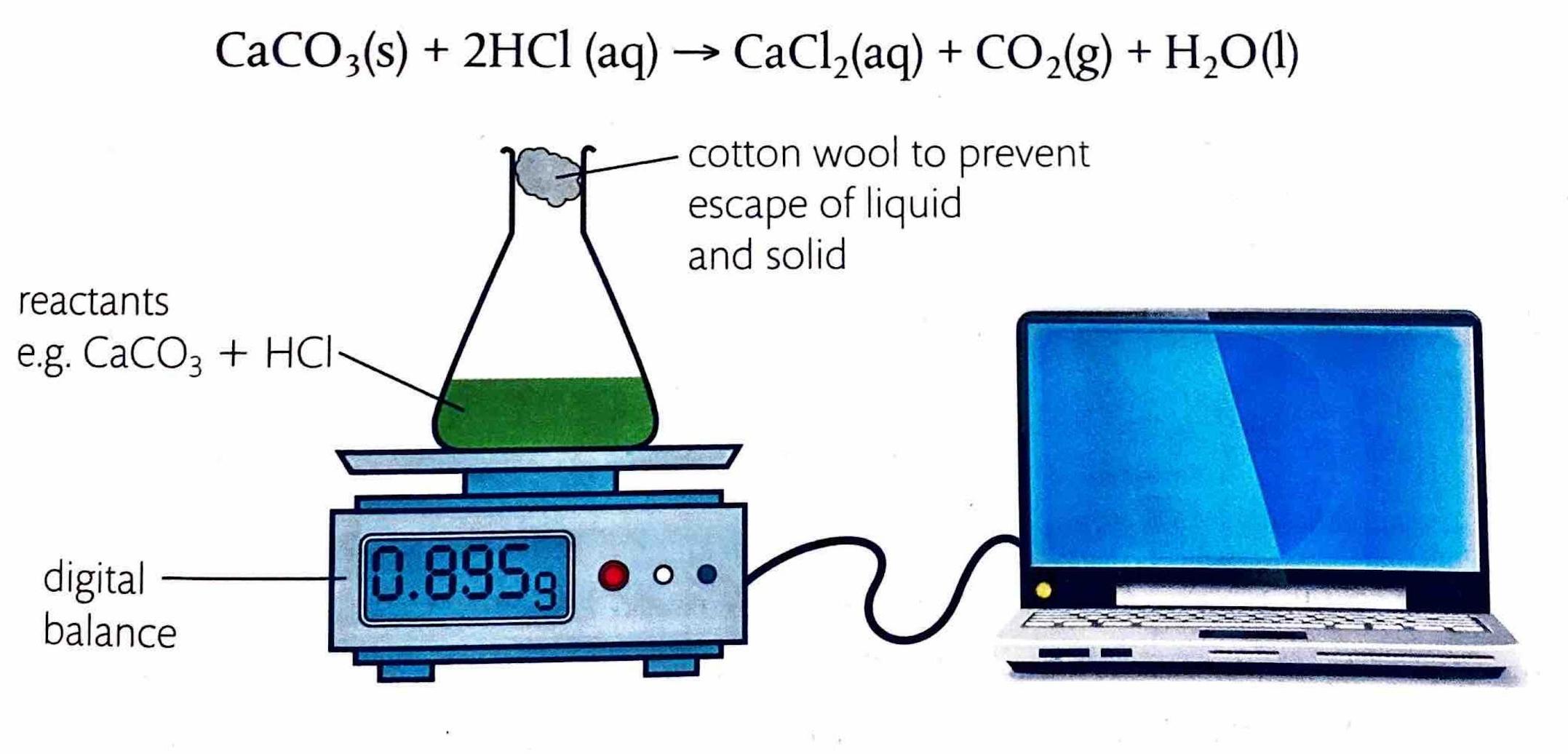
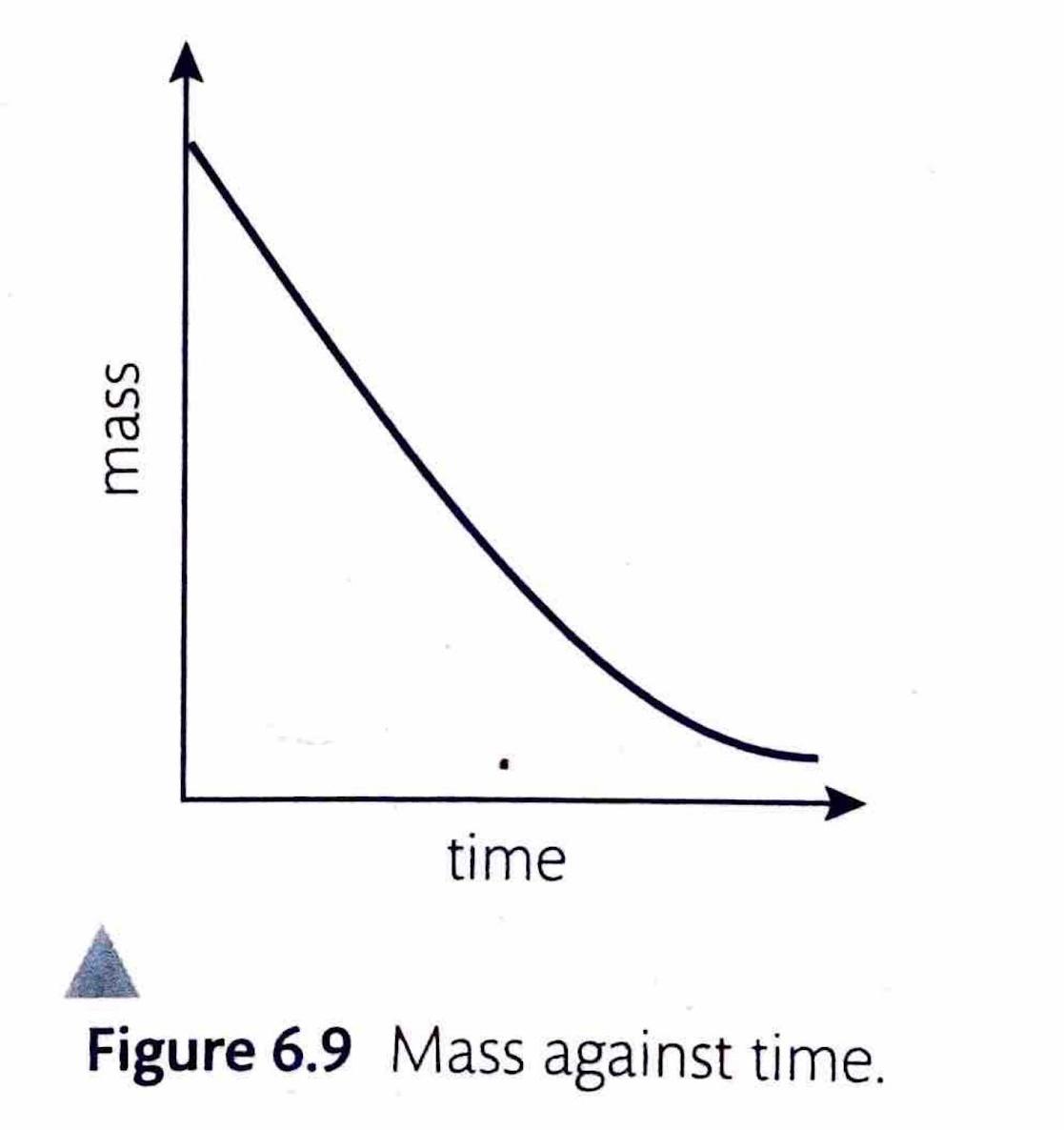
Measuring Rate by Change in Mass
If a reaction produces a gaseous product, the rate can be determined by measuring the decrease in the total mass of the reaction mixture over time. This is typically achieved by placing the reaction vessel on a balance. This method is particularly effective when small molar-mass gases, such as hydrogen (H
2) or carbon dioxide (CO
2), are evolved, as their escape leads to a more significant and easily measurable mass change.
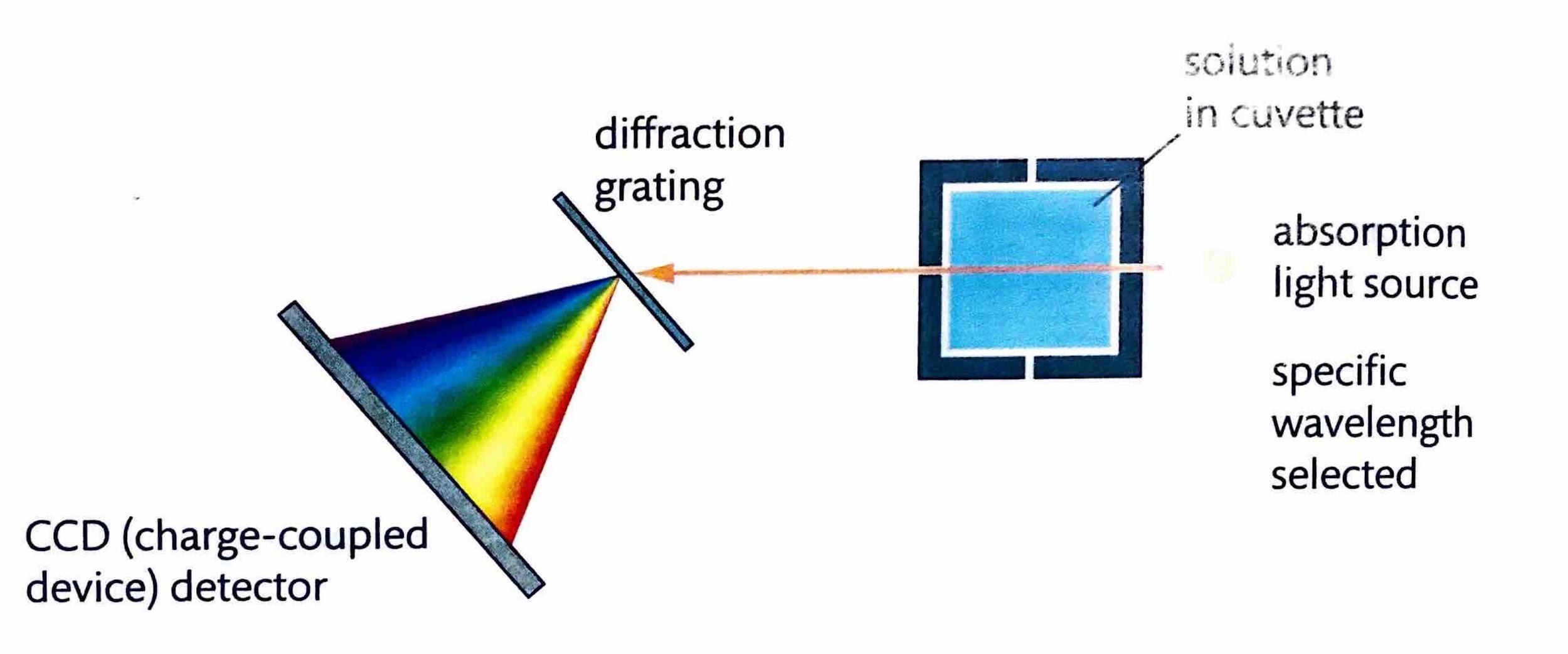
Measuring Rate by Change in Light Transmission
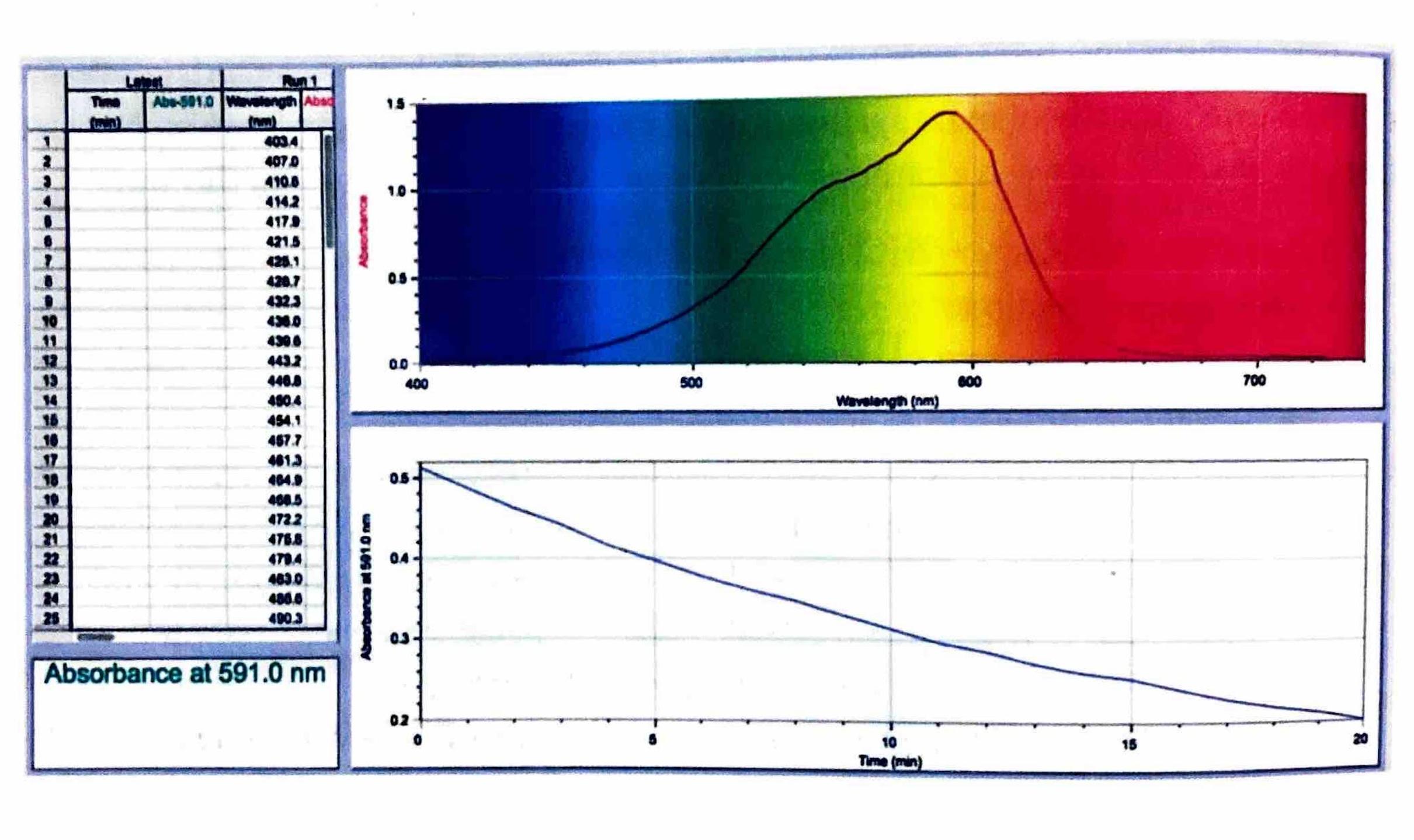
The rate of a reaction can be monitored by observing changes in the transmission of light through the reaction mixture, provided that one of the reactants or products is colored, or if a color indicator can be used. Colored compounds absorb light in the visible region of the electromagnetic spectrum. A colorimeter or spectrophotometer works by passing specific wavelengths of light through the solution and measuring the amount of light that is transmitted. As the concentration of a colored compound increases, its light absorbance proportionally increases, leading to a decrease in the amount of transmitted light. This technique allows for continuous readings, enabling the creation of a graph of absorbance (or transmittance) versus time, from which the reaction rate can be determined.
Example: Crystal Violet and Sodium Hydroxide Reaction
An excellent example of measuring reaction rate via light transmission is the reaction between crystal violet (CV
+) and sodium hydroxide (OH
-):
CV
+ + OH
- → CVOH
In this reaction, the crystal violet reactant is intensely colored, while the product, CVOH, is colorless. Therefore, the reaction rate can be conveniently measured by monitoring the decrease in absorbance (or increase in transmittance) of the solution over time as the colored crystal violet is consumed.
Measuring Rate by Titration
Titration is a method used to determine the concentration of a reactant or product by reacting it with a solution of known concentration (a standard). To monitor reaction rates, samples are periodically removed from the reaction mixture at specific time intervals. These samples are then analyzed by titration. To prevent the reaction from continuing in the sample after it has been drawn, a process called "quenching" is often employed, where a substance is added to effectively stop the reaction. For instance, the concentration of iodine in a reaction can be determined by titrating it with sodium thiosulfate. At regular time intervals, samples are taken and quenched with sodium carbonate before titration.
Measuring Rate by Change in Conductivity
The total electrical conductivity of a solution is dependent on the total concentration of ions present and their respective charges. Therefore, changes in conductivity can be used to monitor reaction rates if there is a net change in the number or type of ions as reactants are converted to products. A conductivity meter can be used to continuously record these changes. To relate conductivity measurements to actual concentrations, calibration is often performed. This involves measuring the conductivities of solutions with known concentrations and then graphing this data to create a calibration curve, which can then be used to determine the concentrations of unknown solutions from their measured conductivities. For example, a reaction that consumes ions or produces neutral molecules from ions would result in a decrease in conductivity.
Non-Continuous Method: Clock Reactions
Clock reactions are a non-continuous method for measuring reaction rates. They involve determining the time it takes for a reaction to reach a specific, observable "end point." By comparing the time taken to reach this end point under different experimental conditions, insights into how reaction rates change can be gained. However, it is important to note that this method only provides an average rate of reaction over the measured time interval, rather than instantaneous rates.
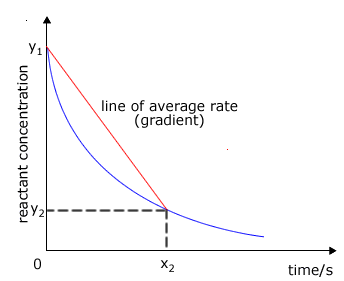
Calculating Instantaneous Rates
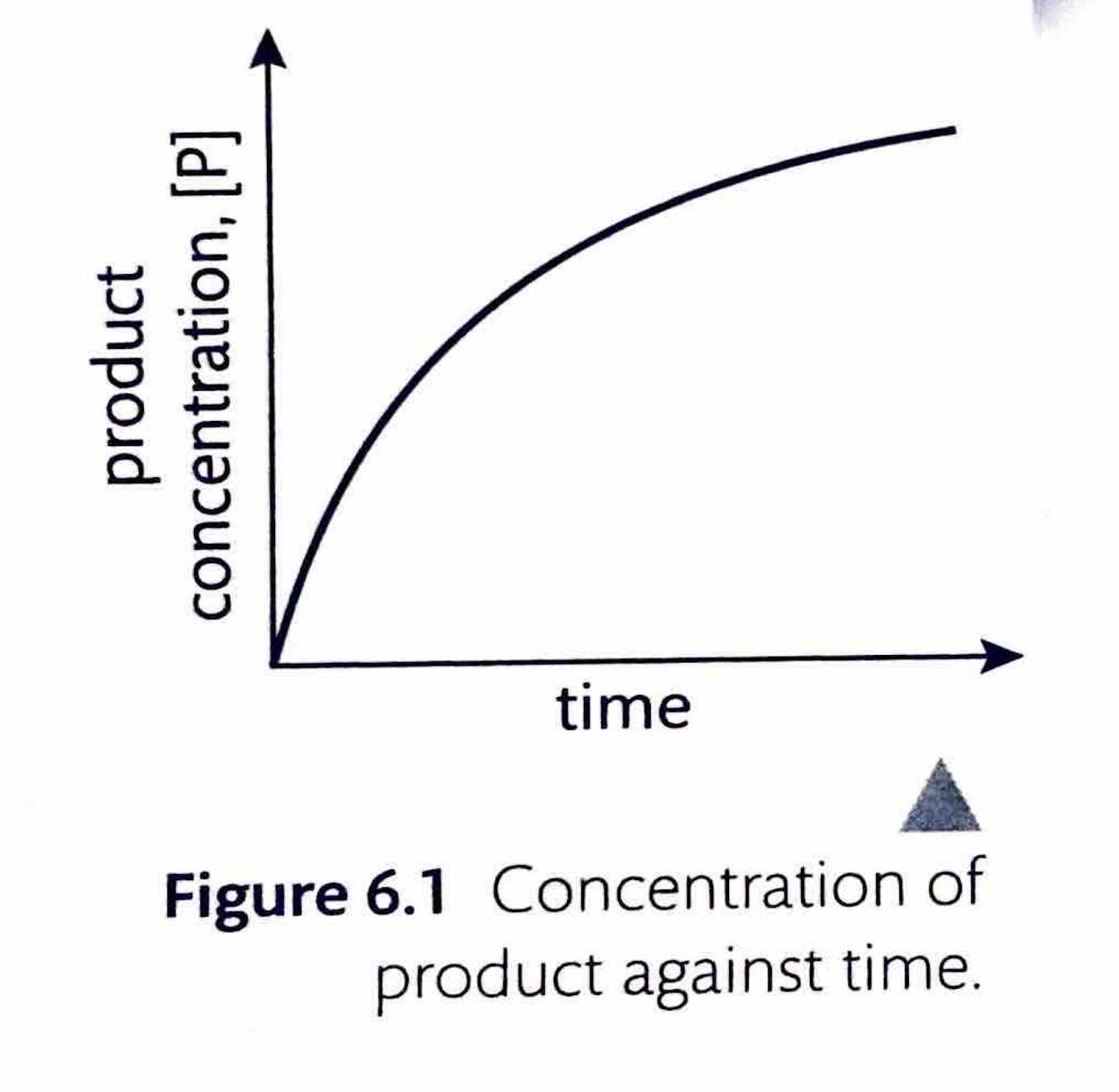
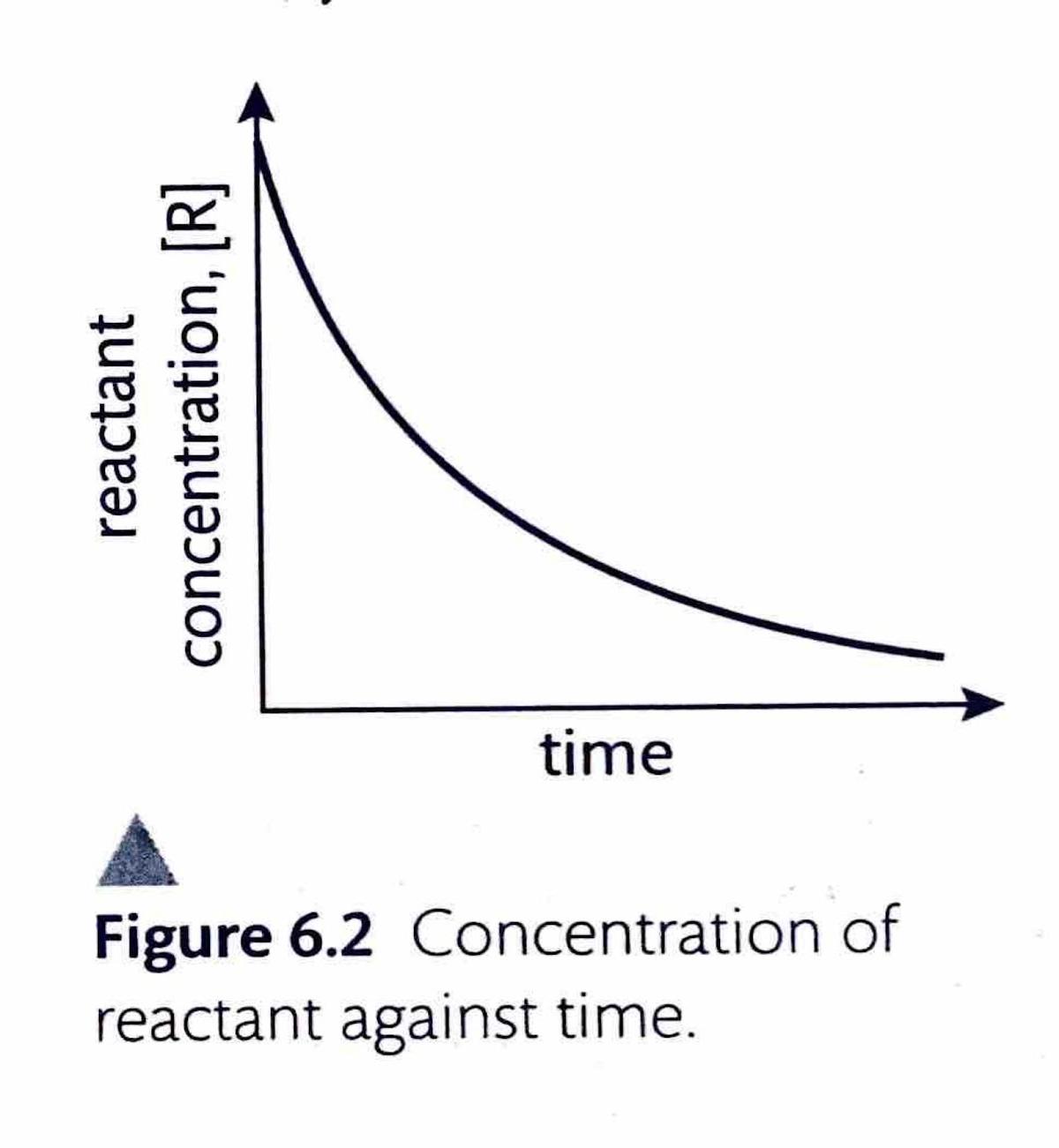
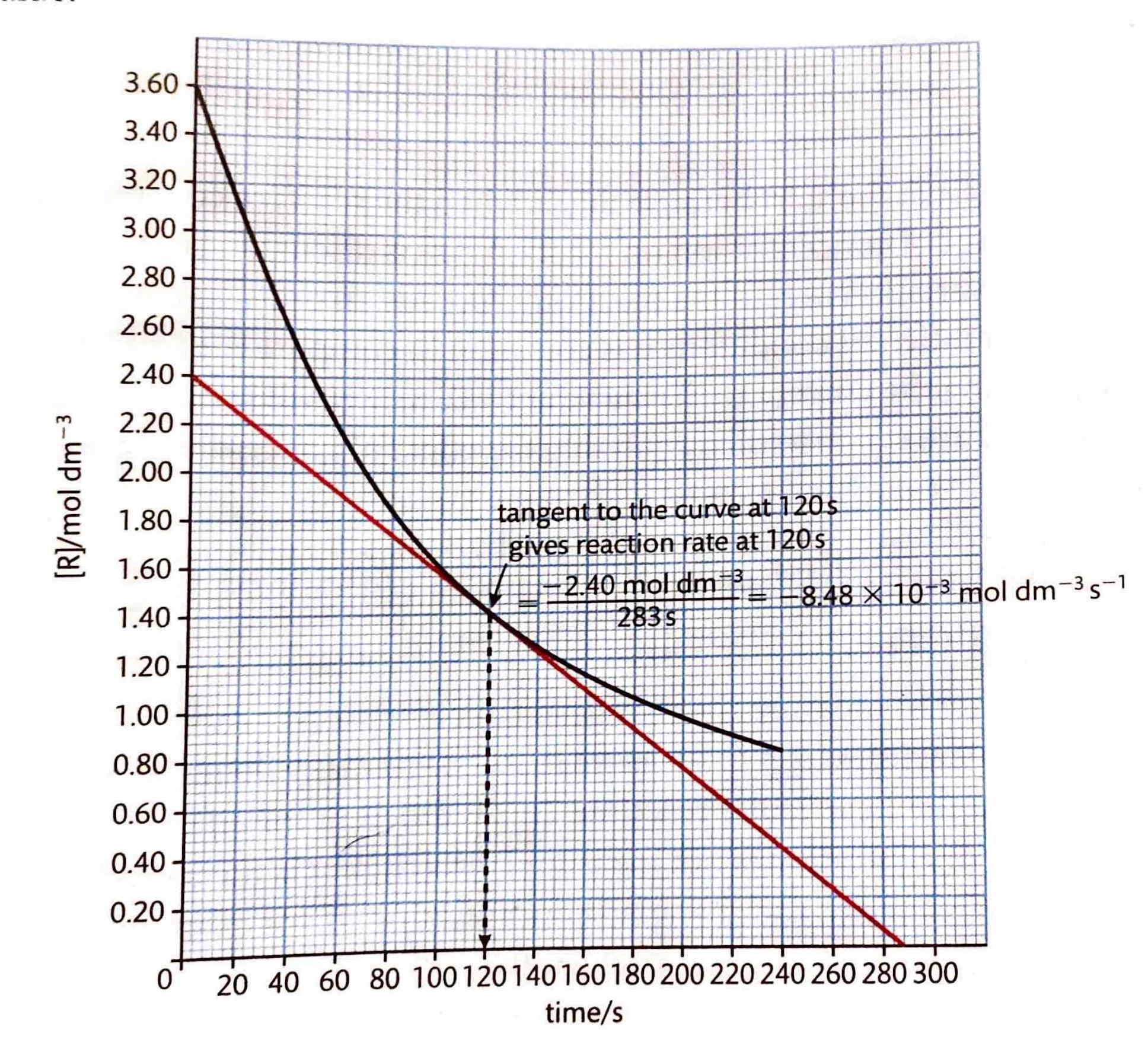
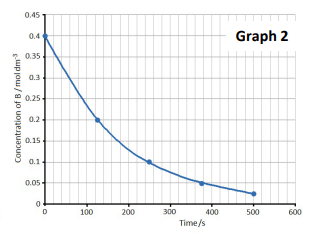
The instantaneous rate of a reaction at any given moment can be determined from a concentration-time graph. The steepness, or gradient, of the curve at a particular point represents the change in concentration per unit time, which is the reaction rate. Since the gradient of a curve is not constant, the rate can only be specified for a particular time point or a very short time frame. This instantaneous rate is measured by drawing a tangent to the curve at the desired time point. To ensure greater accuracy, the tangent should be extended as long as possible. By convention, even though the concentration of reactants ([R]) decreases over time, reaction rates are always expressed as positive values.