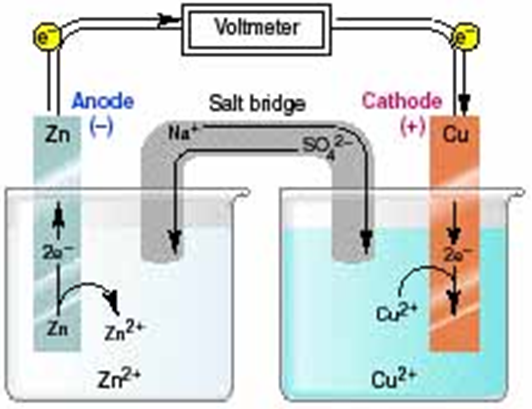
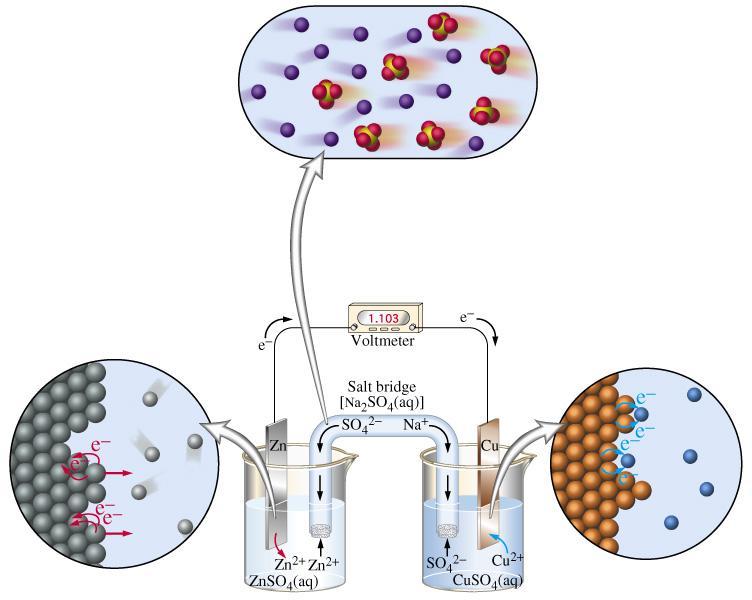
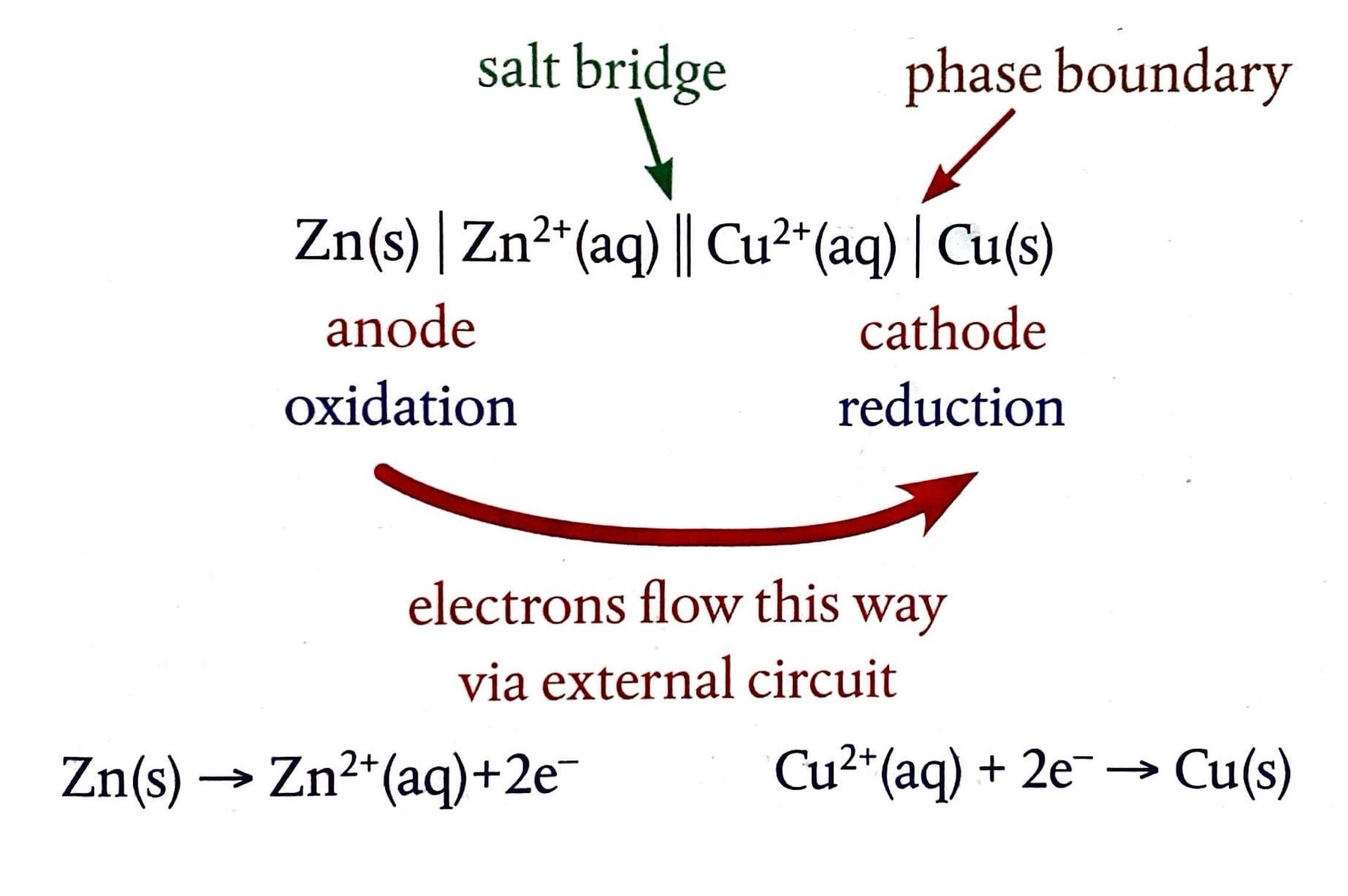
Distinguishing Redox Titrations from Acid-Base Titrations
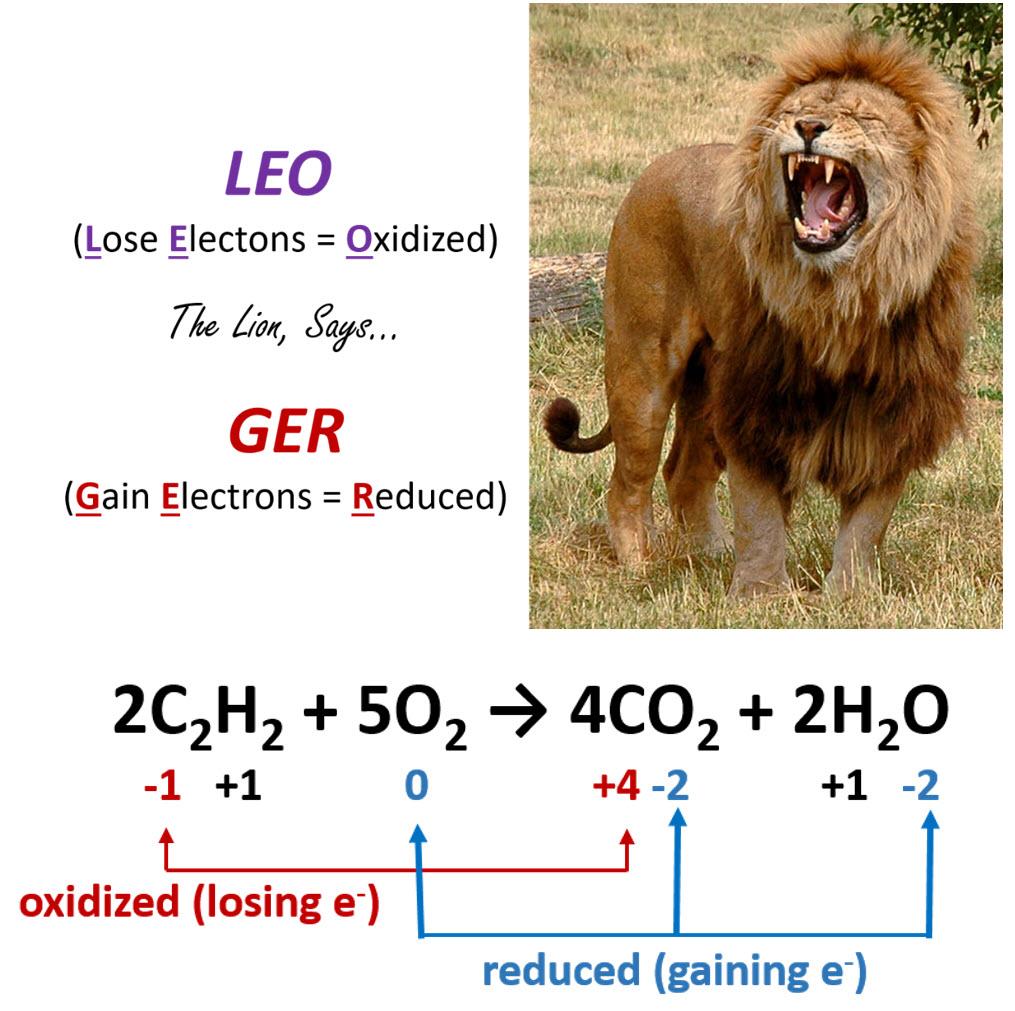
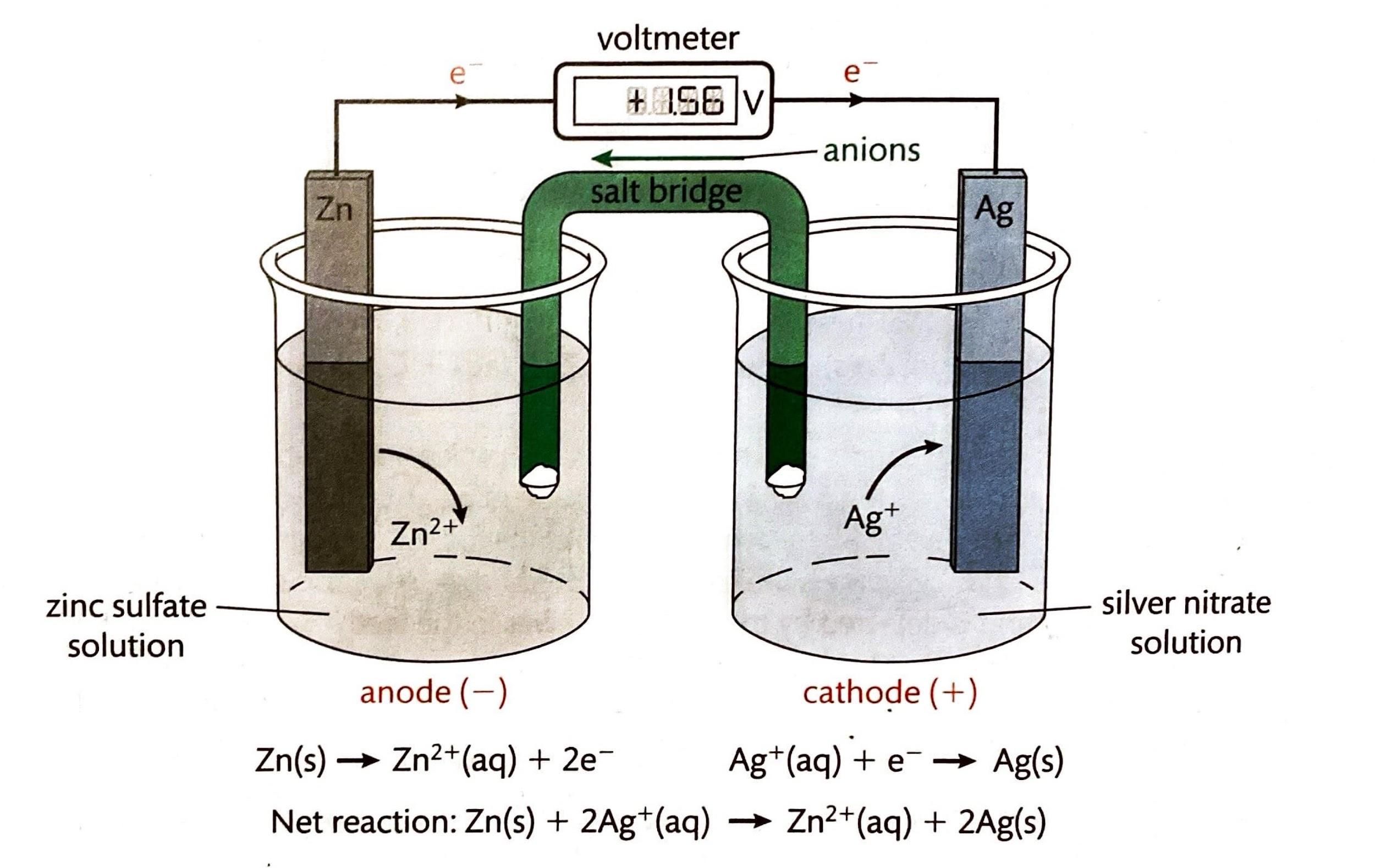
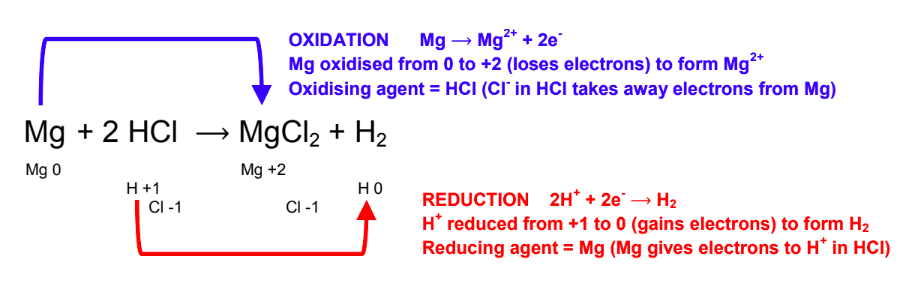
Redox titrations differ fundamentally from acid-base titrations in the nature of the chemical reaction being monitored. Instead of an acid reacting with a base, a redox titration involves an oxidizing agent reacting with a substance that is oxidized, or vice versa. The primary goal is to determine the amount of a substance that has undergone either reduction or oxidation. Unlike acid-base titrations, where the simple M
1V
1 = M
2V
2 formula is often applicable, redox titrations require a more nuanced approach due to the stoichiometry of electron transfer.
Applications of Redox Titrations and Indicator Considerations
In many redox titrations, external indicators are not required because the redox changes themselves are accompanied by a visible color change. For instance, the presence of iodine in water or the reaction of methylene blue with glucose can serve as self-indicators. Redox titrations are widely employed across various industries. In the food and beverage sector, they are used to analyze wines for sulfur dioxide content or to determine the vitamin C content in foods. The pharmaceutical industry utilizes them for purity and content analysis of drugs. Furthermore, environmental analysis, such as the determination of dissolved oxygen in water, heavily relies on redox titrations.
Systematic Approach to Redox Titration Calculations
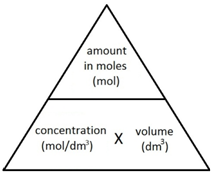
To determine the mass or concentration of an unknown substance in a redox titration, a systematic approach is followed. The process begins by writing out the two half-ionic equations for the oxidation and reduction reactions. These half-equations are then combined to form a balanced full ionic equation, ensuring that the number of electrons lost in oxidation equals the number of electrons gained in reduction. Next, the moles of the substance for which concentration and volume are known (typically the titrant in the burette) are calculated using the volume required to reach the equivalence point. Based on the stoichiometric ratio from the balanced full ionic equation, the moles of the reacting substance (usually in the flask) are determined. If the sample was diluted or taken from a larger volume, the moles in the original solution must be calculated by scaling up. Finally, the mass or concentration of the original sample can be calculated using the determined moles and the molar mass or original volume, respectively. The term "titre" refers specifically to the volume of solution added from the burette to reach the equivalence point of the titration.
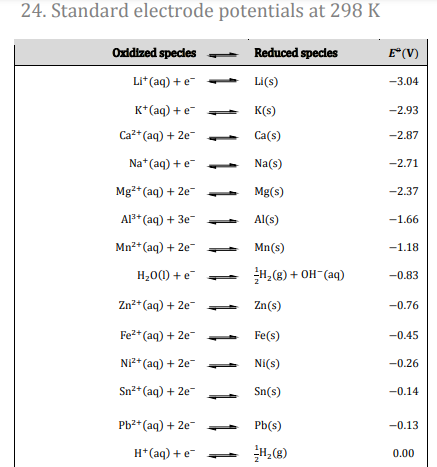
Common Reagents and Their Redox Half-Equations
Several common reagents are frequently used in redox titrations, each with characteristic half-equations. Manganate(VII) ions (MnO₄⁻), also known as permanganate, act as a strong oxidizing agent and are reduced to manganese(II) ions (Mn²⁺) in acidic solution:
MnO₄⁻ + 8H⁺ + 5e⁻ → Mn²⁺ + 4H₂O
Ethanedioate ions (C₂O₄²⁻) serve as a reducing agent, being oxidized to carbon dioxide:
C₂O₄²⁻(aq) → 2CO₂(g) + 2e⁻
Iron(II) ions (Fe²⁺) can act as a reducing agent, losing electrons to become iron(III) ions (Fe³⁺):
Fe²⁺(s) → Fe³⁺(s) + e⁻
Dichromate(VI) ions (Cr₂O₇²⁻) are another powerful oxidizing agent, reduced to chromium(III) ions (Cr³⁺) in acidic solution:
Cr₂O₇²⁻(aq) + 14H⁺(aq) + 6e⁻ → 2Cr³⁺(aq) + 7H₂O(l)
Example: Analysis of Iron with Manganate(VII)
Consider an example where 24.30 cm³ of 0.010 mol dm⁻³ aqueous potassium manganate(VII) reacted with 25.0 cm³ of acidified FeSO₄(aq) to form Mn²⁺. This 25.0 cm³ sample was originally taken from a 250 cm³ flask. To determine the original mass of Fe²⁺ used, we first write the balanced half-equations and the overall ionic equation.
The reduction half-equation for manganate(VII) is:
MnO₄⁻ + 8H⁺ + 5e⁻ → Mn²⁺ + 4H₂O
The oxidation half-equation for iron(II) is:
Fe²⁺ → Fe³⁺ + e⁻
To balance the electrons, the iron(II) half-equation is multiplied by 5:
5Fe²⁺ → 5Fe³⁺ + 5e⁻
Combining these gives the overall ionic equation:
MnO₄⁻ + 8H⁺ + 5Fe²⁺ → Mn²⁺ + 4H₂O + 5Fe³⁺
From this, the stoichiometric ratio of MnO₄⁻ to Fe²⁺ is 1:5.
Next, calculate the moles of MnO₄⁻ used:
Moles MnO₄⁻ = concentration × volume = 0.010 mol dm⁻³ × 0.0243 dm³ = 0.000243 mol
Using the 1:5 ratio, the moles of Fe²⁺ in the 25 cm³ flask are:
Moles Fe²⁺ = 0.000243 mol × 5 = 0.001215 mol
Since this 25 cm³ sample was taken from a 250 cm³ flask, the total moles of Fe²⁺ in the original 250 cm³ solution are:
Moles Fe²⁺ in 250 cm³ = 0.001215 mol × (250 cm³ / 25 cm³) = 0.001215 mol × 10 = 0.01215 mol
Finally, calculate the original mass of Fe²⁺ (molar mass of Fe is approximately 55.845 g/mol, often rounded to 56 g/mol for simpler calculations):
Mass Fe²⁺ = molar mass × moles = 56 g/mol × 0.01215 mol = 0.68 g
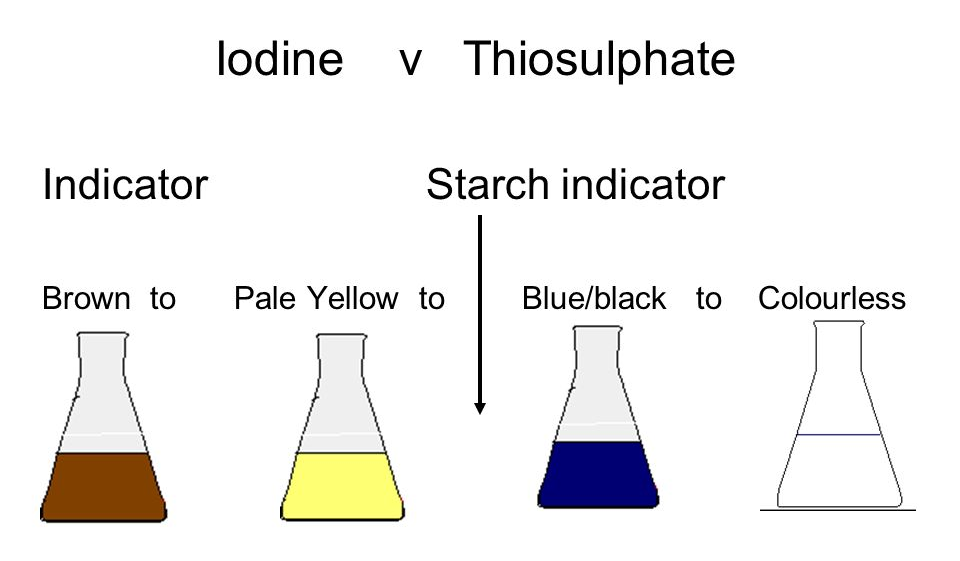
The Iodine-Thiosulfate Reaction
The iodine-thiosulfate reaction is a common redox titration method, particularly useful when an oxidizing agent reacts with excess iodide ions to produce iodine. The general reaction for the formation of iodine is:
2I⁻(aq) + oxidizing agent → I₂(aq) + reduced product
The iodine produced is then titrated with a standard solution of sodium thiosulfate (Na₂S₂O₃). Starch is used as an indicator in this titration. The half-equations for this reaction are:
Oxidation: 2S₂O₃²⁻ → S₄O₆²⁻ + 2e⁻
Reduction: I₂ + 2e⁻ → 2I⁻
The overall balanced equation for the titration is:
2S₂O₃²⁻(aq) + I₂(aq) → 2I⁻(aq) + S₄O₆²⁻(aq)
In the presence of starch, iodine forms a deep blue complex. As the thiosulfate is added, it reacts with the iodine, causing the blue color to disappear precisely at the equivalence point, signaling the end of the titration.
Introduction to the Winkler Method
The Winkler method is a crucial redox titration technique used to measure the dissolved oxygen content in water. Dissolved oxygen is a vital indicator of water quality; lower oxygen levels often correlate with higher levels of pollution. A related concept is Biological Oxygen Demand (BOD), which quantifies the amount of oxygen consumed by microorganisms to decompose organic matter in a water sample over a specific period (typically 5 days) at a given temperature. A high BOD indicates lower dissolved oxygen levels, suggesting greater organic pollution.
Detailed Steps of the Winkler Method
The Winkler method employs a series of redox reactions to determine dissolved oxygen. Initially, the dissolved oxygen (O₂) in the water sample is "fixed" by adding a manganese(II) salt, such as MnSO₄, in a basic solution. The oxygen oxidizes the Mn(II) to higher oxidation states, typically Mn(IV), forming a precipitate of manganese dioxide.
The first step involves the oxidation of Mn(II) by dissolved oxygen:
2Mn²⁺(aq) + O₂(g) + 4OH⁻(aq) → 2MnO₂(s) + 2H₂O(l)
Next, acidified iodide ions (I⁻) are added to the solution.
The Mn(IV) in the manganese dioxide then oxidizes the iodide ions to iodine (I₂):
MnO₂(s) + 2I⁻(aq) + 4H⁺(aq) → Mn²⁺(aq) + I₂(aq) + 2H₂O(l)
Finally, the liberated iodine is titrated with a standard solution of sodium thiosulfate (Na₂S₂O₃), using starch as an indicator:
2S₂O₃²⁻(aq) + I₂(aq) → 2I⁻(aq) + S₄O₆²⁻(aq)
Alternative Representation of the Winkler Method
Another way to represent the initial stages of the Winkler method involves the rapid oxidation of Mn²⁺ ions to Mn³⁺ by dissolved oxygen under alkaline conditions, forming a pale brown precipitate of Mn(OH)₃:
4Mn(OH)₂(aq) + O₂(g) + 2H₂O(l) → 4Mn(OH)₃(s)
Subsequently, a sample of river or stream water, after being shaken with excess alkaline Mn²⁺ ions, has its precipitate reacted with an excess of potassium iodide (KI).
This reaction oxidizes the iodide to iodine (I₂):
2KI(aq) + 2Mn(OH)₃(s) → I₂(aq) + 2Mn(OH)₂(aq) + 2KOH(aq)
Titration in the Winkler Method
The amount of iodine (I₂) produced in the preceding steps is then precisely determined by titration with a sodium thiosulfate solution of known concentration. Starch is added as an indicator, which forms a deep blue complex with iodine. The endpoint of the titration is marked by the disappearance of this blue color as the thiosulfate reacts with all the iodine.
The titration reaction is:
2S₂O₃²⁻(aq) + I₂(aq) → S₄O₆²⁻(aq) + 2I⁻(aq)
Example Calculation: Winkler Method for Dissolved Oxygen
Let's consider a 500 cm³ water sample analyzed for dissolved oxygen using the Winkler method. After the addition of MnSO₄ in basic solution and subsequent acidification with KI, 12.50 cm³ of 0.0500 mol dm⁻³ Na₂S₂O₃(aq) was required to react with the iodine produced. We aim to calculate the dissolved oxygen content of the water in g dm⁻³.
First, calculate the moles of S₂O₃²⁻ used:
Moles S₂O₃²⁻ = concentration × volume = 0.0500 mol dm⁻³ × 0.01250 dm³ = 0.000625 mol
From the titration equation (2S₂O₃²⁻ + I₂ → 2I⁻ + S₄O₆²⁻), the ratio of S₂O₃²⁻ to I₂ is 2:1.
Moles I₂ = 0.000625 mol S₂O₃²⁻ × (1 mol I₂ / 2 mol S₂O₃²⁻) = 0.0003125 mol I₂
Now, we need to relate moles of I₂ back to moles of O₂ using the earlier reactions:
MnO₂(s) + 2I⁻(aq) + 4H⁺(aq) → Mn²⁺(aq) + I₂(aq) + 2H₂O(l) (Ratio I₂ : MnO₂ is 1:1)
2Mn²⁺(aq) + O₂(g) + 4OH⁻(aq) → 2MnO₂(s) + 2H₂O(l) (Ratio MnO₂ : O₂ is 2:1)
Combining these, the overall stoichiometric relationship between O₂ and I₂ is 1 mol O₂ to 2 mol I₂.
Moles O₂ = 0.0003125 mol I₂ × (1 mol O₂ / 2 mol I₂) = 0.00015625 mol O₂
Next, calculate the mass of O₂ in the 500 cm³ sample (Molar mass of O₂ = 32.00 g/mol):
Mass O₂ = molar mass × moles = 32.00 g/mol × 0.00015625 mol = 0.00500 g
This mass is present in 500 cm³ (0.500 dm³) of water.
To express the dissolved oxygen content in g dm⁻³:
Dissolved oxygen = 0.00500 g / 0.500 dm³ = 0.0100 g dm⁻³
Alternative Example Calculation: Winkler Method
Let's consider another example where the moles of S₂O₃²⁻ are 0.0000348 mol.
Moles S₂O₃²⁻ = 0.00200 mol dm⁻³ × 0.01740 dm³ = 0.0000348 mol
Using the 2:1 ratio of S₂O₃²⁻ to I₂:
Moles I₂ = 0.0000348 mol S₂O₃²⁻ × 0.5 = 0.0000174 mol I₂
Using the overall 1:2 ratio of O₂ to I₂ (derived from the reactions:
2Mn²⁺ + O₂ → 2MnO₂ and MnO₂ + 2I⁻ → I₂):
Moles O₂ = 0.0000174 mol I₂ × 0.5 = 8.70 × 10⁻⁶ mol O₂
If this amount of oxygen was present in a 50 cm³ (0.050 dm³) sample, the dissolved oxygen concentration would be:
Dissolved oxygen = 8.70 × 10⁻⁶ mol / 0.050 dm⁻³ = 1.74 × 10⁻⁴ mol dm⁻³
The key reactions involved are:
2Mn²⁺(aq) + O₂(g) + 4OH⁻(aq) → 2MnO₂(s) + 2H₂O(l)
MnO₂(s) + 2I⁻(aq) + 4H⁺(aq) → Mn²⁺(aq) + I₂(aq) + 2H₂O(l)
2S₂O₃²⁻(aq) + I₂(aq) → 2I⁻(aq) + S₄O₆²⁻(aq)