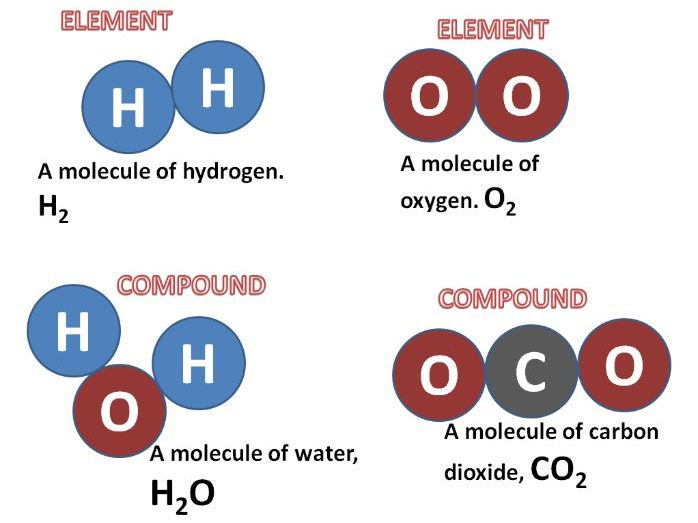
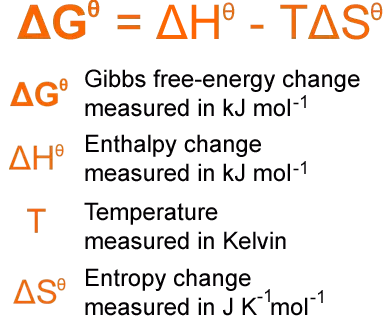Understanding Entropy
Entropy (S
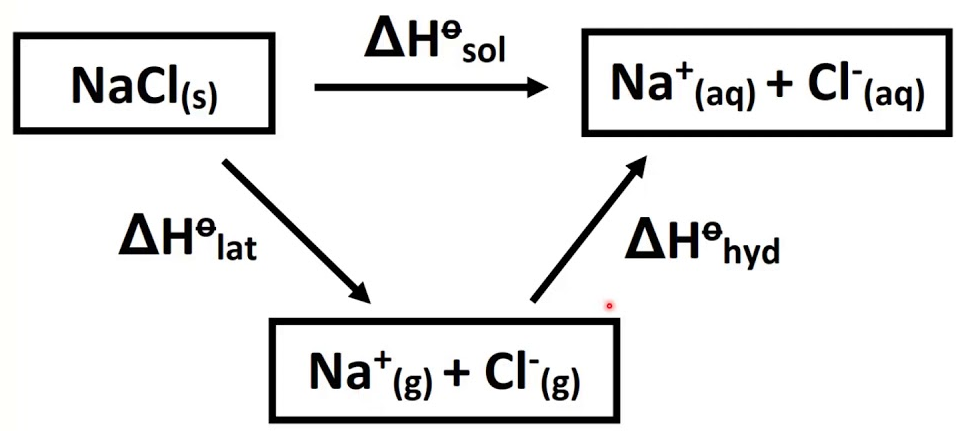
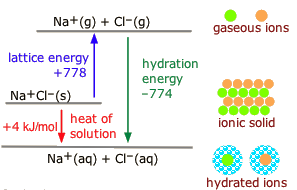
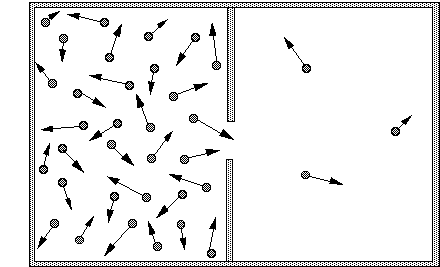
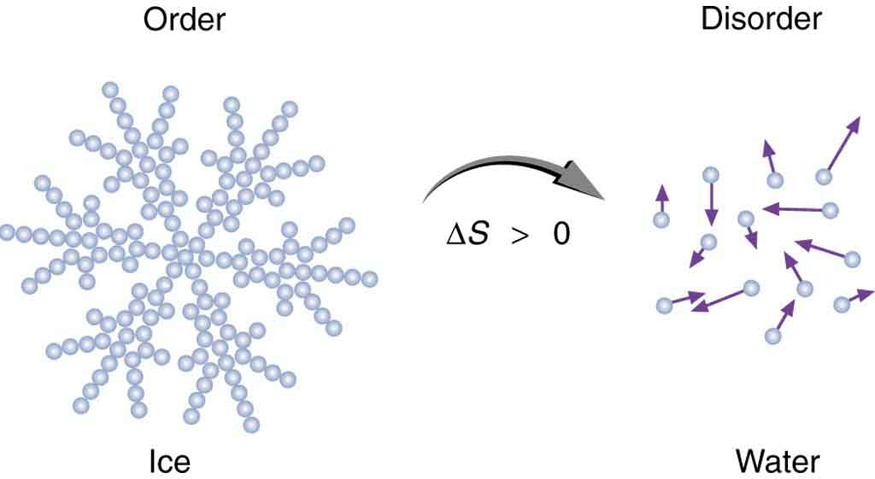
ϴ) is a fundamental thermodynamic property that quantifies the degree of disorder or randomness within a system. It can also be conceptualized as the number of possible arrangements or microstates that particles within a system can adopt. A higher number of accessible microstates corresponds to greater entropy. The more energy (quanta) particles possess, the more ways they can distribute that energy, leading to higher entropy. Similarly, an increase in the number of particles (moles) within a system generally results in higher entropy because there are more individual entities to arrange, as seen when salts dissolve.
Factors Affecting Entropy
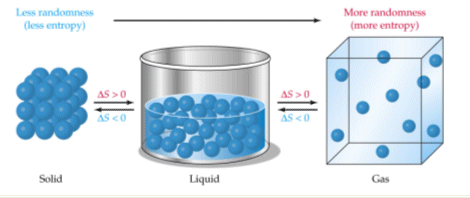
Several factors influence the entropy of a substance or system. For instance, the physical state of a substance significantly impacts its entropy; gases exhibit the highest entropy due to their particles having the greatest freedom of movement and arrangement, followed by liquids, and then solids, which have the most ordered structure. Therefore, NaCl(aq) has higher entropy than NaCl(s) because the dissolved ions are more disordered than in the solid lattice. Similarly, a more complex molecule like CO
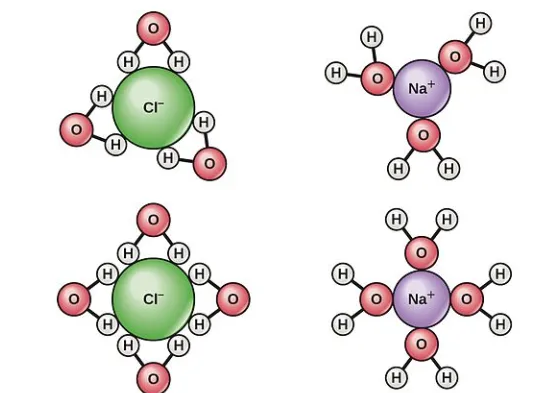
2 generally has higher entropy than a simpler one like CO because it has more atoms and thus more ways to vibrate and rotate. When a solute like NaCl dissolves in water to form NaCl(aq), the resulting solution has more arrangements and greater particle motion compared to the separate solute and solvent, leading to higher entropy. Comparing the entropy of N
2(g) + O
2(g) with 2NO(g) is difficult without specific values, as the number of moles of gas remains the same, suggesting a similar degree of disorder. Finally, increasing the temperature of a substance, such as H
2O from room temperature to 50°C, increases its entropy because the particles gain more kinetic energy and move more vigorously, leading to a greater number of possible arrangements.
Entropy and Temperature Relationship
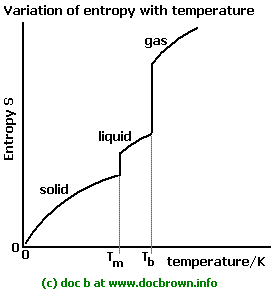
As temperature increases, the entropy of a substance generally increases. This is because particles gain more kinetic energy at higher temperatures, allowing them to occupy a wider range of energy states and spatial arrangements. This leads to a greater number of microstates and thus higher entropy. Significant and sudden increases in entropy are observed at phase transition points, specifically at the melting and boiling points. The increase in entropy is particularly pronounced at the boiling point, where a substance transitions from a relatively ordered liquid state to a highly disordered gaseous state. Gas particles possess significantly more translational and rotational freedom compared to liquid or solid particles, resulting in a much larger increase in disorder and entropy during vaporization.
Standard Entropy (Sϴ)
Standard entropy (S
ϴ) refers to the entropy of one mole of a substance under standard atmospheric conditions (SATP), which are defined as 100 kPa pressure and 298 K (25°C) temperature. These values are typically found in data booklets. The units for standard entropy are Joules per Kelvin per mole (J K
-1 mol
-1). It is crucial to ensure correct unit usage in calculations.
Standard entropy values can vary based on several factors. For example, molecular complexity plays a role; NO
2 has a standard entropy of 240 J K
-1 mol
-1, which is higher than NO's 211 J K
-1 mol
-1. This difference arises because the more complex NO
2 molecule has more atoms and therefore more possible vibrational and rotational modes, leading to a greater number of microstates. The physical state of a substance also significantly impacts its standard entropy. For instance, H
2O(g) has a standard entropy of 699 J K
-1 mol
-1, which is considerably higher than H
2O(l)'s 189 J K
-1 mol
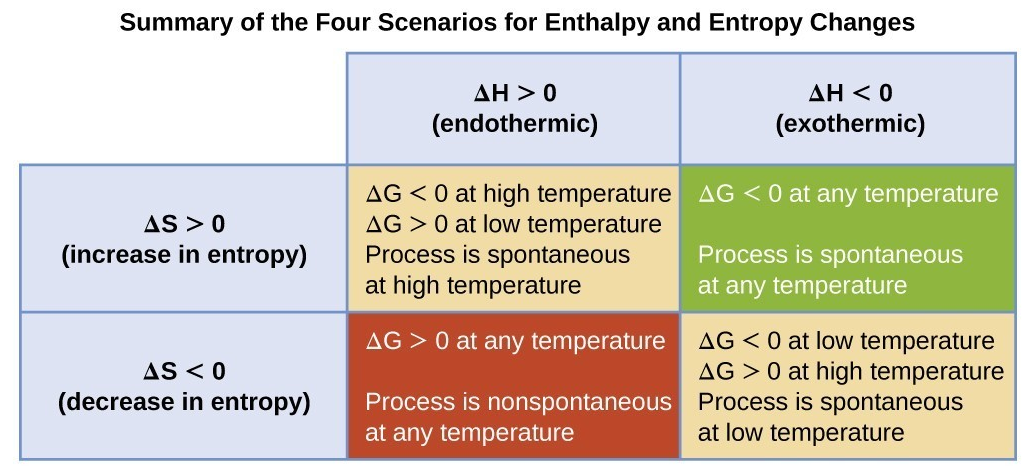
-1, reflecting the greater disorder in the gaseous state. It is important to note that spontaneous endothermic reactions, which absorb heat from their surroundings, can occur if the increase in entropy of the system and/or surroundings is sufficiently favorable.
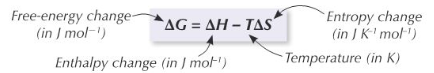
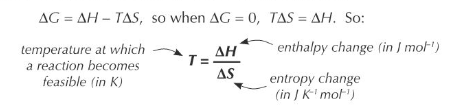
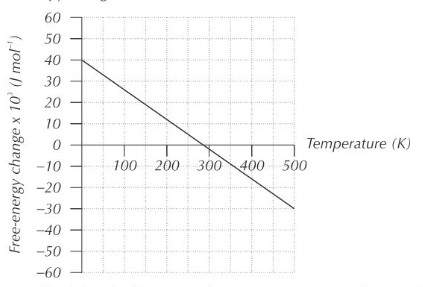
Interpreting Entropy Change
An increase in entropy for a reaction is indicated by a positive change in entropy (ΔS > 0), suggesting that the products are more disordered than the reactants. Conversely, a decrease in entropy is indicated by a negative change in entropy (ΔS < 0), meaning the products are more ordered. For a reaction to be considered feasible or spontaneous under specific conditions, the total entropy change (ΔS
total) must be positive.
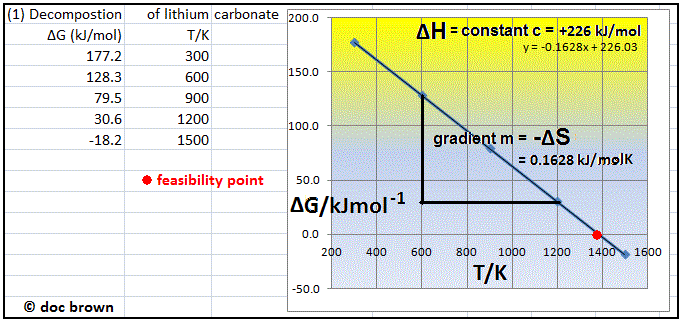
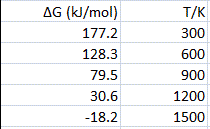
Calculating Entropy Change
The total entropy change for a reaction can be determined using three key equations, which are essential to learn and apply. These equations allow for the calculation of entropy changes within the system, the surroundings, and the overall process. Thermodynamic data, including standard entropy values, can be found in section 13 of IB Chemistry data booklets.
The entropy change of the system (ΔS
system) is calculated by subtracting the sum of the standard entropies of the reactants from the sum of the standard entropies of the products:
ΔS
system = ∑S
ϴproducts - ∑S
ϴreactants
The entropy change of the surroundings (ΔS
surroundings) is related to the enthalpy change of the reaction (ΔH) and the absolute temperature (T) in Kelvin:
ΔS
surroundings = -ΔH/T (in J mol
-1K
-1)
Finally, the total entropy change (ΔS
total) for a process is the sum of the entropy change of the system and the entropy change of the surroundings:
ΔS
total = ΔS
system + ΔS
surroundings
Entropy Change Example 1: Calcium Oxidation
Let's calculate the total entropy change for the reaction 2Ca(s) + O
2(g) → 2CaO(s) under standard conditions. We are given the standard enthalpy change (ΔH
ϴ = -1270 kJ mol
-1) and the standard entropy values (S
ϴ) for each substance: Ca = 41.6 J K
-1 mol
-1, O
2 = 205 J K
-1 mol
-1, and CaO = 40 J K
-1 mol
-1.
First, we calculate the entropy change of the system:
ΔS
system = (2 × S
ϴCaO) - [(2 × S
ϴCa) + S
ϴO2]
ΔS
system = (2 × 40) - [(2 × 41.6) + 205]
ΔS
system = 80 - [83.2 + 205]
ΔS
system = 80 - 288.2
ΔS
system = -208.2 J K
-1 mol
-1
Next, we calculate the entropy change of the surroundings. Note that ΔH
ϴ must be converted from kJ to J:
ΔS
surroundings = -ΔH
ϴ/T
ΔS
surroundings = -(-1270 × 1000 J mol
-1) / 298 K
ΔS
surroundings = 1270000 J mol
-1 / 298 K
ΔS
surroundings = +4261.7 J K
-1 mol
-1
Finally, we calculate the total entropy change:
ΔS
total = ΔS
system + ΔS
surroundings
ΔS
total = -208.2 J K
-1 mol
-1 + 4261.7 J K
-1 mol
-1
ΔS
total = +4053.5 J K
-1 mol
-1
Since the total entropy change is positive (+4053.5 J K
-1 mol
-1), this reaction is kinetically feasible (spontaneous) under standard conditions.
Entropy Change Example 2: Dissolution of Sodium Bromide
Let's determine if NaBr dissolves in solution at 298 K. We are given the entropy change of the system (ΔS
ϴsystem = +55.0 J K
-1 mol
-1) for the reaction NaBr(s) → Na
+(aq) + Br
-(aq), and the standard enthalpy change (ΔH
ϴ = -0.6 kJ mol
-1).
First, we calculate the entropy change of the surroundings, converting ΔH
ϴ from kJ to J:
ΔS
surroundings = -ΔH
ϴ/T
ΔS
surroundings = -(-0.6 × 1000 J mol
-1) / 298 K
ΔS
surroundings = 600 J mol
-1 / 298 K
ΔS
surroundings = +2.01 J K
-1 mol
-1 (rounded to two decimal places)
Next, we calculate the total entropy change:
ΔS
total = ΔS
system + ΔS
surroundings
ΔS
total = +55.0 J K
-1 mol
-1 + 2.01 J K
-1 mol
-1
ΔS
total = +57.01 J K
-1 mol
-1
Since the total entropy change is positive (+57.01 J K
-1 mol
-1), the dissolution of NaBr in water is kinetically feasible (spontaneous) at 298 K.