Understanding Groups: Columns of the Periodic Table

The periodic table organizes elements based on their atomic number and recurring chemical properties. Three particularly important groups for study in IB Chemistry are Group 1 (the alkali metals), Group 17 (the halogens), and Group 18 (the noble gases). These groups exhibit distinct trends in their physical and chemical characteristics due to their electron configurations.
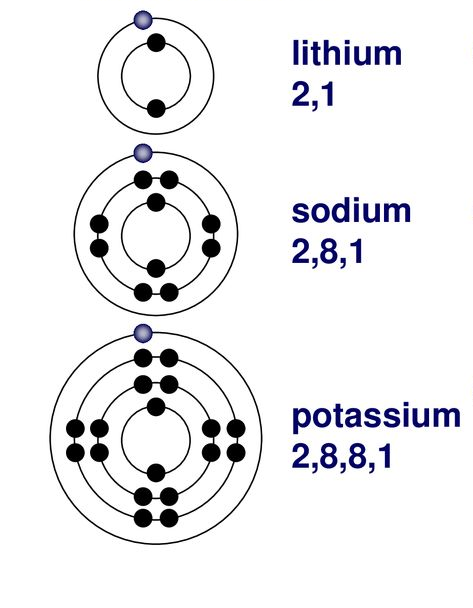
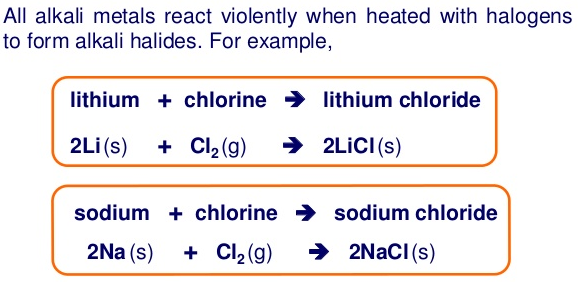
Group 1 elements, known as the alkali metals, are highly reactive metals characterized by having a single valence electron in their outermost shell. This electron is easily lost, leading to the formation of a +1 ion. The elements in this group are:
| Li |
| Na |
| K |
| Rb |
| Cs |
| Fr |
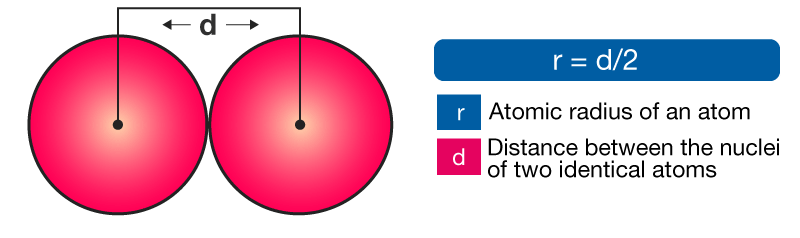
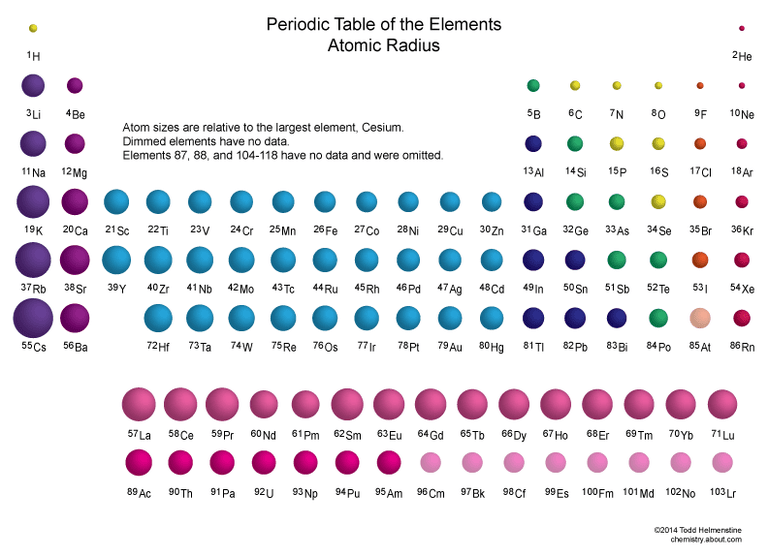
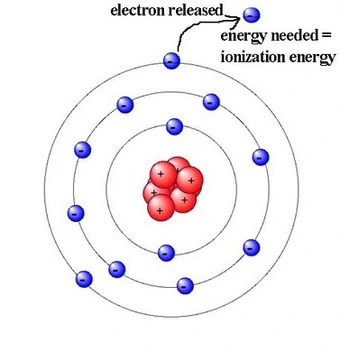
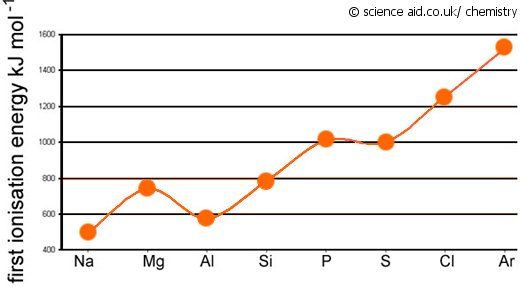
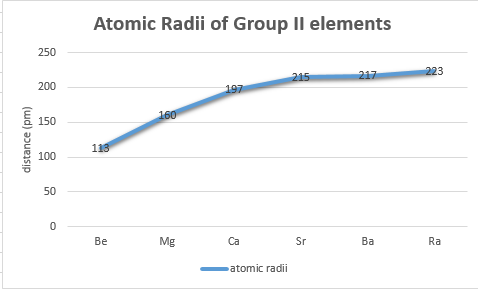
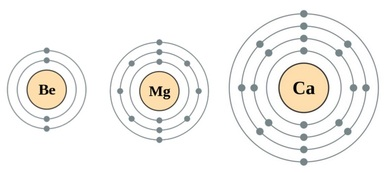
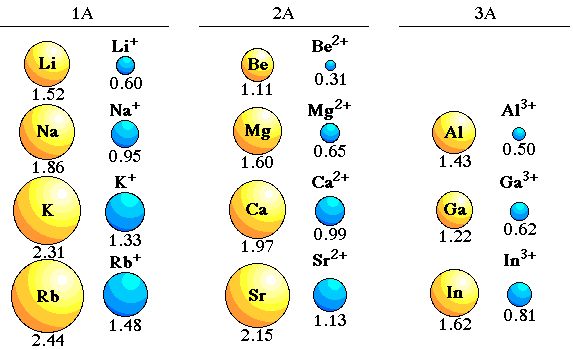
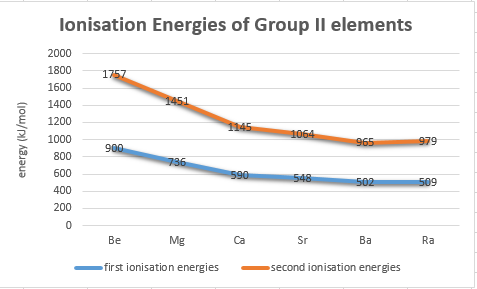
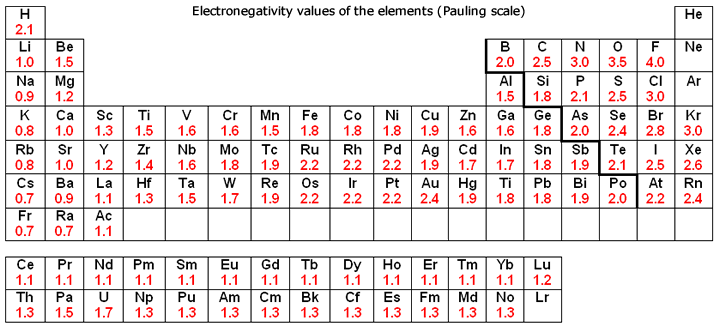
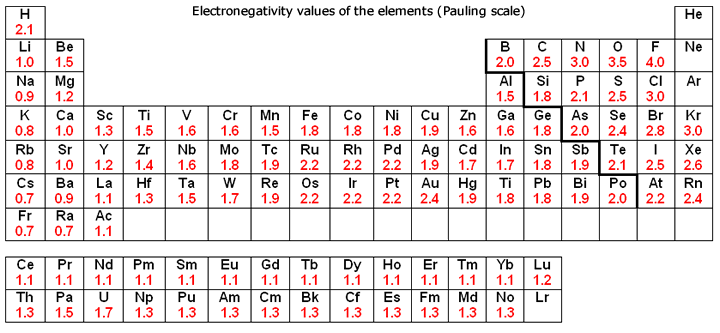
As one moves down Group 1, the atomic radius increases, the first ionization energy decreases, and the electronegativity decreases. These trends contribute to the increasing reactivity of the alkali metals down the group, as the outermost electron is further from the nucleus and more easily removed.
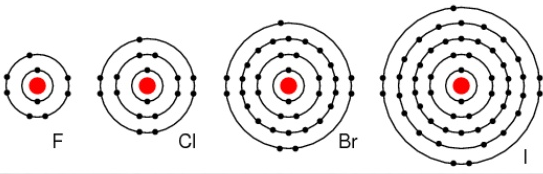
Group 17 elements are known as the halogens, a term derived from Greek meaning "salt-forming." These non-metals are highly reactive, possessing seven valence electrons, and readily gain one electron to achieve a stable octet, forming a -1 ion. The elements in this group are:
| F |
| Cl |
| Br |
| I |
| At |
Moving down Group 17, the atomic radius increases, the electronegativity decreases, and the reactivity generally decreases. This is because the increasing atomic size means the incoming electron is further from the nucleus, experiencing less attraction, making it harder to gain an electron. Halogens exist as diatomic molecules (e.g., F2, Cl2) in their elemental form.
Group 18 elements are the noble gases, characterized by their exceptional stability and very low reactivity. This inertness is due to their full outermost electron shells, which means they have a stable octet (or duet for helium) and no strong tendency to gain, lose, or share electrons. The elements in this group are:
| He |
| Ne |
| Ar |
| Kr |
| Xe |
| Rn |
Noble gases exist as monatomic gases at standard temperature and pressure. Their boiling points and melting points are very low, increasing slightly down the group due to increasing London dispersion forces as the atomic size and number of electrons increase.
Transition metals are elements that possess an incomplete d subshell in one or more of their common oxidation states. This definition excludes elements like scandium (Sc) and zinc (Zn). Scandium, while located in the d-block, typically forms only the Sc3+ ion, which has an empty d subshell. Zinc, on the other hand, forms the Zn2+ ion, which has a full d subshell (d10). Therefore, neither scandium nor zinc strictly meet the definition of a transition metal based on their common oxidation states.
Transition metals exhibit several characteristic properties due to their partially filled d orbitals. These include the formation of complex ions, the presence of multiple oxidation states, the formation of colored compounds, and their catalytic activity. These properties are directly linked to the electronic configuration of their d subshells, which allows for electron transitions and interactions with ligands.
Lanthanoids, also known as the lanthanide series, are a group of 15 metallic chemical elements with atomic numbers from 57 (lanthanum, La) to 71 (lutetium, Lu). These elements are characterized by the filling of their 4f electron shells. They are often referred to as rare earth elements, although many are not particularly rare in abundance. Their chemical properties are very similar due to the shielding effect of the 5s and 5p orbitals, which makes the 4f electrons less involved in bonding.
Actinoids, or the actinide series, comprise 15 metallic chemical elements with atomic numbers from 89 (actinium, Ac) to 103 (lawrencium, Lr). These elements are characterized by the filling of their 5f electron shells. All actinoids are radioactive, and many are synthetic, meaning they are not found naturally on Earth. Their chemistry is more complex than that of the lanthanoids due to the greater involvement of the 5f electrons in bonding and the relativistic effects on their electron orbitals.
The periodic table provides a systematic organization of elements, where transition metals occupy the d-block, typically groups 3-12. The lanthanoids and actinoids, often referred to as inner transition metals, are placed in the f-block, usually shown as two separate rows below the main body of the periodic table. This arrangement reflects their electron configurations, with the d-block elements filling their d orbitals and the f-block elements filling their f orbitals. The table below illustrates the positions of some key elements within the broader periodic table structure.
| Species | Protons | Electrons | Electron Shells |
|---|---|---|---|
| Cl | 17 | 17 | 3 |
| Cl- | 17 | 18 | 3 |

| Property | Across periods | Down groups |
|---|---|---|
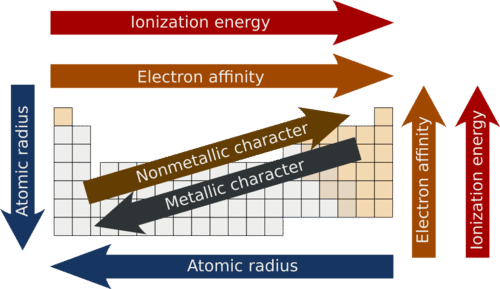
| Atomic Radius | Decreases | Increases |
| Ionic Radius | Decreases (for cations), then increases (for anions), then decreases | Increases |
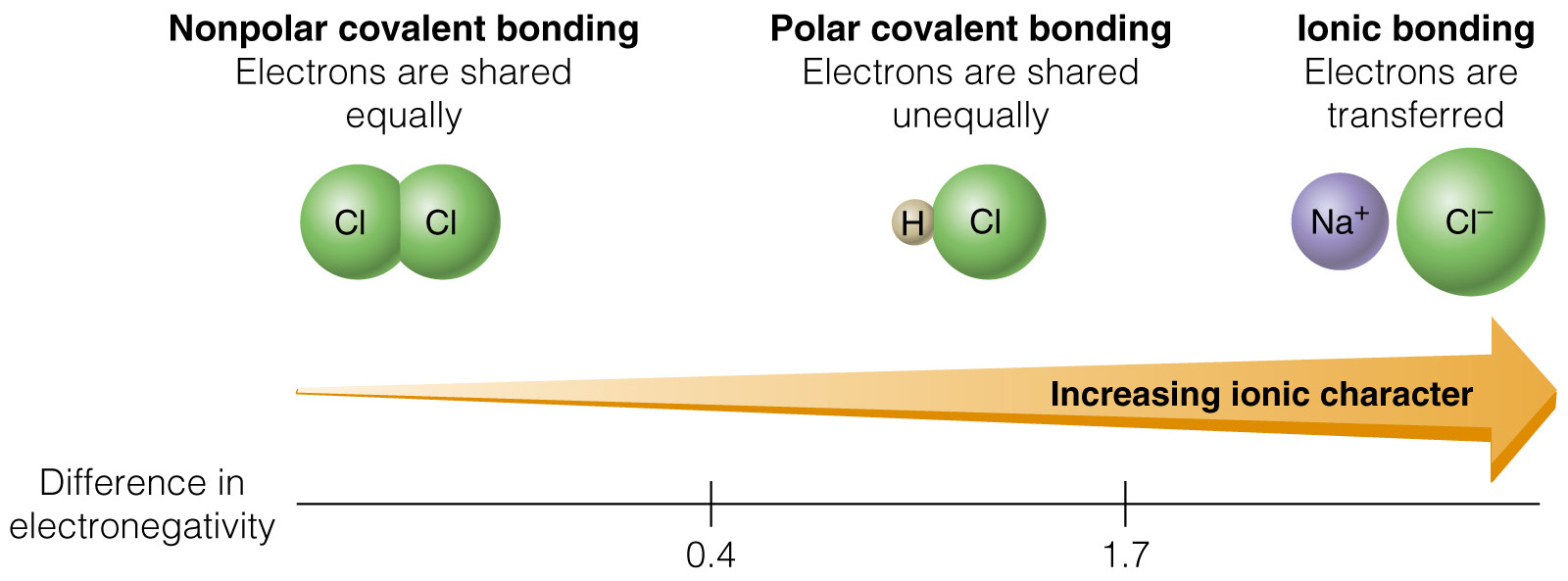
| Electronegativity | Increases | Decreases |
| Ionisation Energy | Increases | Decreases |
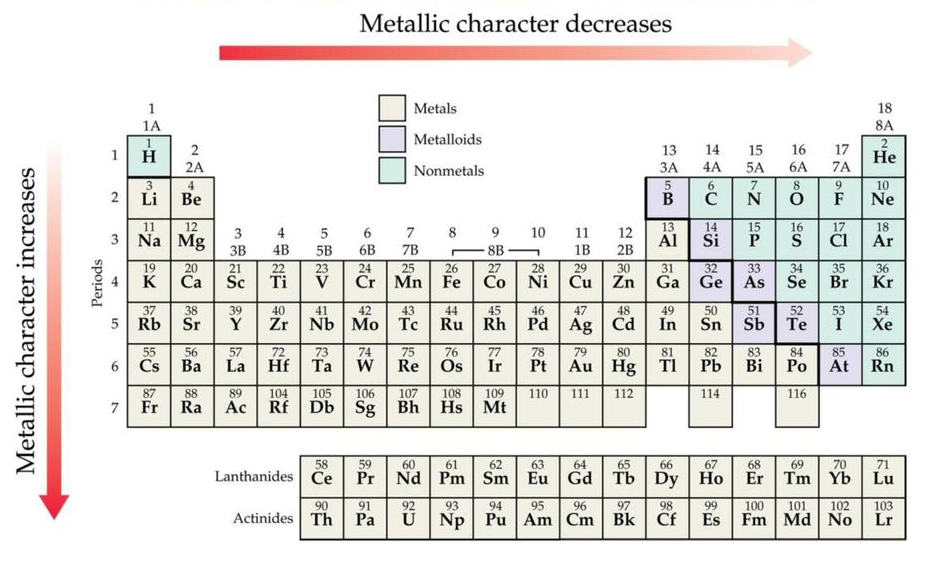
| Metallic character | Decreases | Increases |


| Halogen | Physical properties at room temperature |
|---|---|
| Fluorine | Pale yellow, gas at room temperature, pungent odor, density: 1.696 g/L |
| Chlorine | Yellow-green, gas at room temperature, pungent odor, density: 3.214 g/L |
| Bromine | Reddish-brown, liquid at room temperature, irritating odor, Density: 7.14 g/L |
| Iodine | Violet-black, non-metallic solid at room temperature, Density: 4930 g/L |