Electron Configuration and Stability in Transition Metals
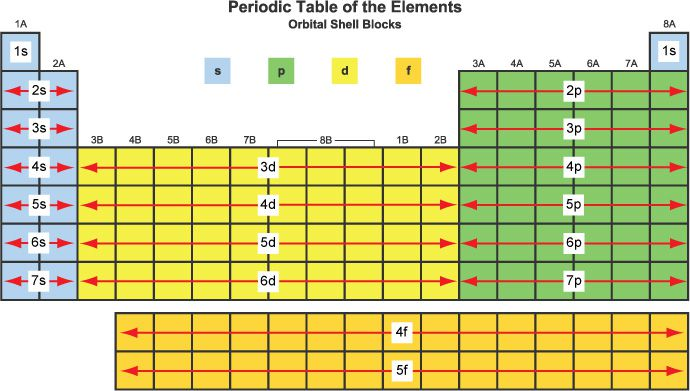
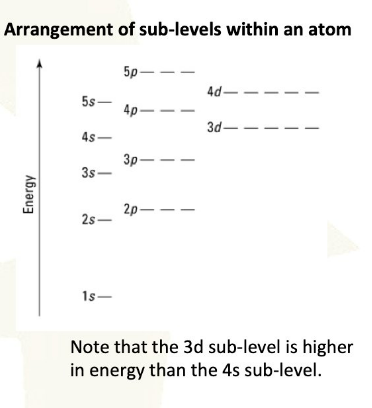
A fundamental principle governing the electron configurations of atoms is that a filled sublevel represents the most stable state, followed by a half-filled sublevel, which is more stable than a partially filled sublevel. To achieve maximum stability, which corresponds to the lowest energy state, electrons may sometimes be promoted from the s sublevel to the d sublevel. This phenomenon is particularly evident in elements belonging to Group 6B and Group 11B of the periodic table. For instance, chromium (Cr), which is in Group 6B, exhibits an electron configuration of [Ar] 4s
13d
5 instead of the expected [Ar] 4s
23d
4. Similarly, copper (Cu), in Group 11B, has a configuration of [Ar] 4s
13d
10 rather than [Ar] 4s
23d
9. This electron promotion allows for a half-filled 3d sublevel in chromium and a fully filled 3d sublevel in copper, both of which are more stable arrangements. When transition metals form ions, the electrons from the s sublevel are removed first because they require less energy to ionize compared to the electrons in the d sublevel.
Coloration in Transition Metal Complexes
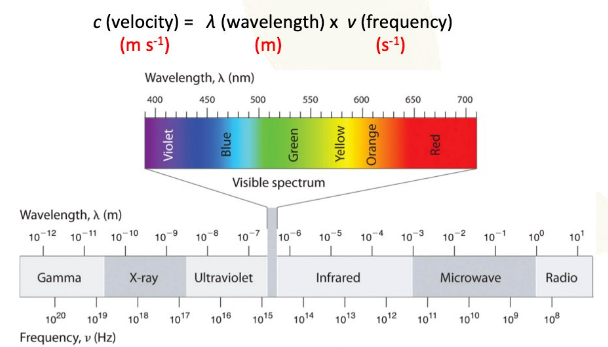
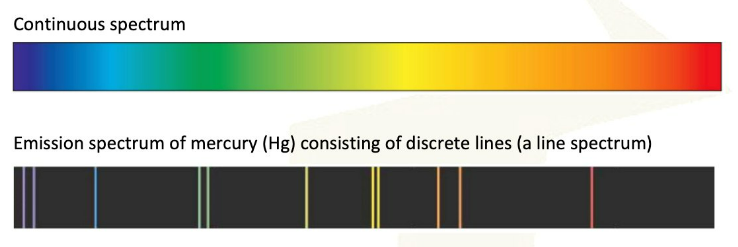
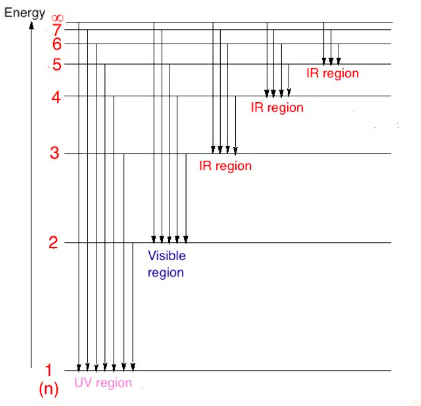
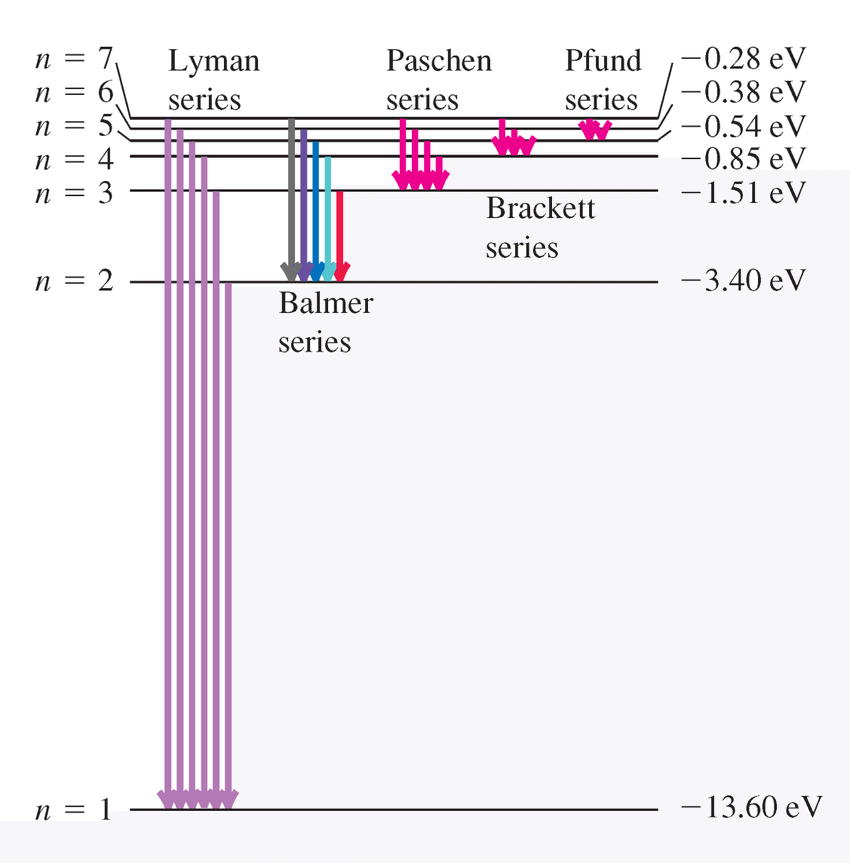
The vibrant colors often observed in transition metal compounds are directly linked to the presence of unpaired electrons in their d sublevels and their interaction with ligands. When white light passes through a solution containing a transition metal ion, certain wavelengths of light are absorbed by the d-orbital electrons. This absorption causes the electrons to jump to higher energy d-orbitals, a process known as d-orbital splitting. The remaining unabsorbed wavelengths are transmitted or reflected, and these are perceived as the color of the compound. The color observed is complementary to the color of the light absorbed.
For example, consider copper and its ions:
- Copper (Cu) in its elemental form has a configuration of [Ar] 4s13d10 and is colored.
- The Cu+ ion has a configuration of [Ar] 4s03d10. Since its d sublevel is completely filled, it typically does not exhibit color.
- The Cu2+ ion has a configuration of [Ar] 4s03d9. The presence of an unpaired electron in the partially filled d sublevel allows for d-d transitions, resulting in a characteristic color.
In contrast, elements like zinc (Zn) and its ion Zn
2+ do not typically display color.
- Zinc (Zn) has a configuration of [Ar] 4s23d10.
- The Zn2+ ion has a configuration of [Ar] 4s03d10.
Both Zn and Zn
2+ have fully filled d sublevels, meaning there are no empty d orbitals for electrons to transition into, and thus no d-d transitions occur, leading to a lack of color.
Defining Transition Elements
It is important to note that the definition of a transition element specifically refers to an element that forms at least one ion with a partially filled d sublevel. Based on this definition, zinc (Zn) is technically not considered a transition element because both its neutral atom and its common ion (Zn
2+) have a completely filled d sublevel.