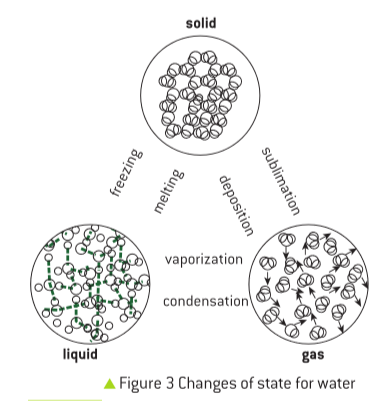
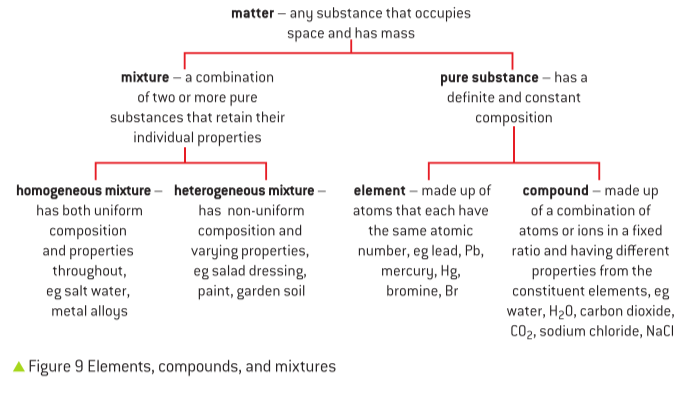
Understanding Isotopes: Variations of an Element
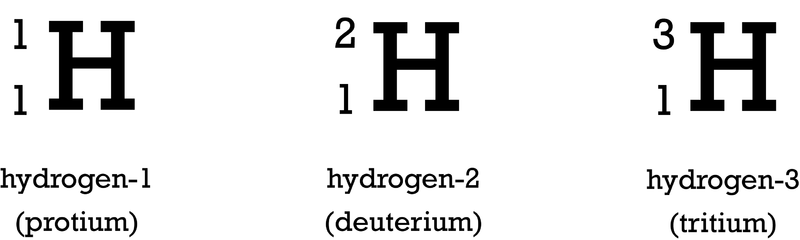
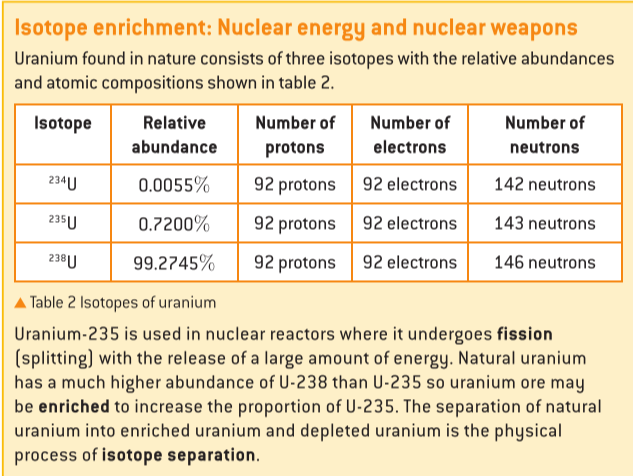
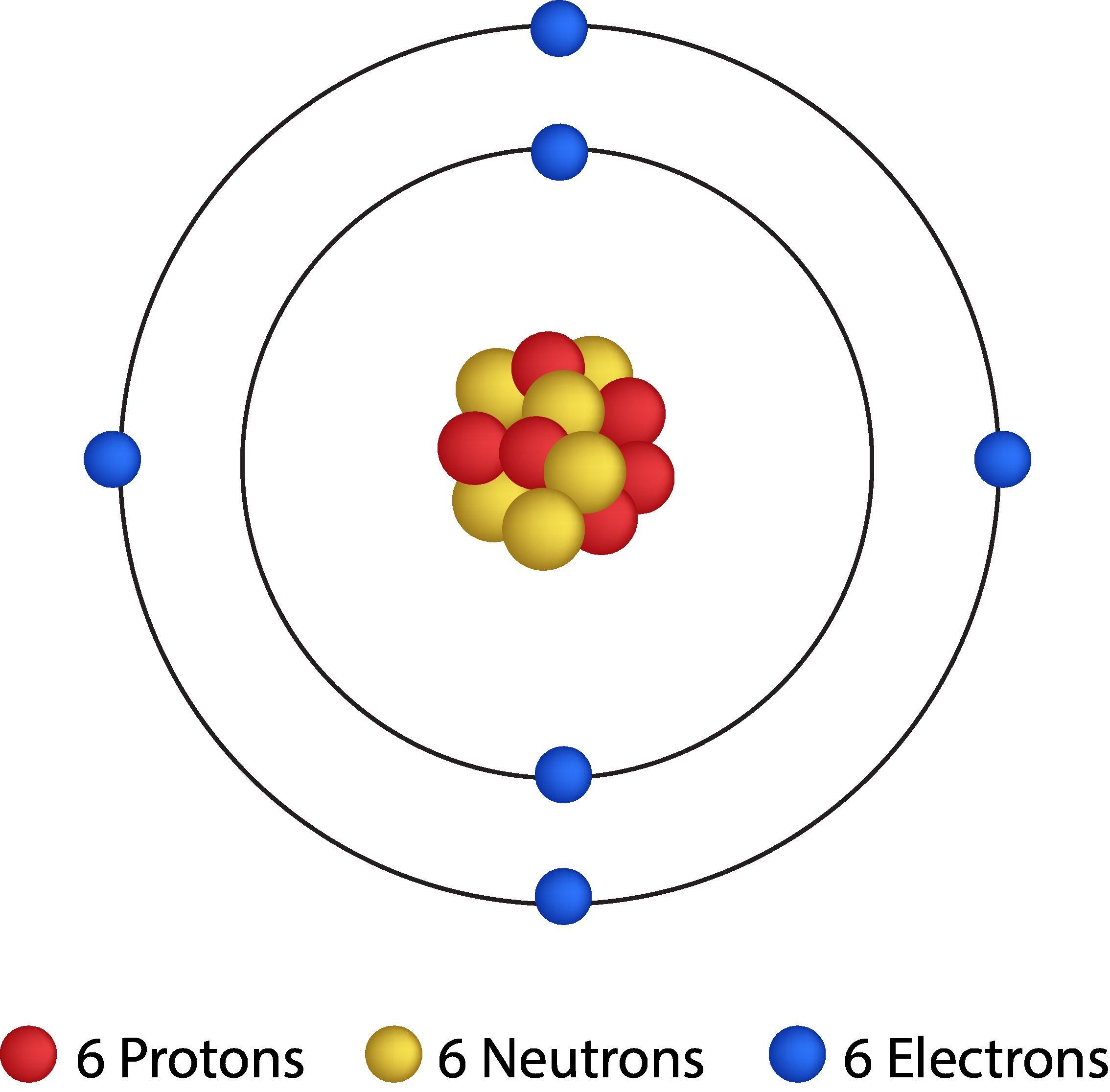
Isotopes are distinct forms of the same element, characterized by having an identical number of protons (atomic number, Z) but differing in their mass numbers (A) due to variations in their neutron count. Most elements naturally occur as a mixture of several isotopes. While isotopes of an element share identical chemical properties because their electron configurations (determined by the number of protons) are the same, they exhibit different physical properties, such as density and melting point, owing to their differing masses. For instance, uranium-235 (
235U) is a crucial isotope used in nuclear reactors and weapons; however, natural uranium ore predominantly contains uranium-238 (
238U), necessitating an enrichment process to increase the concentration of
235U through isotope separation techniques.
Defining Relative Atomic Mass
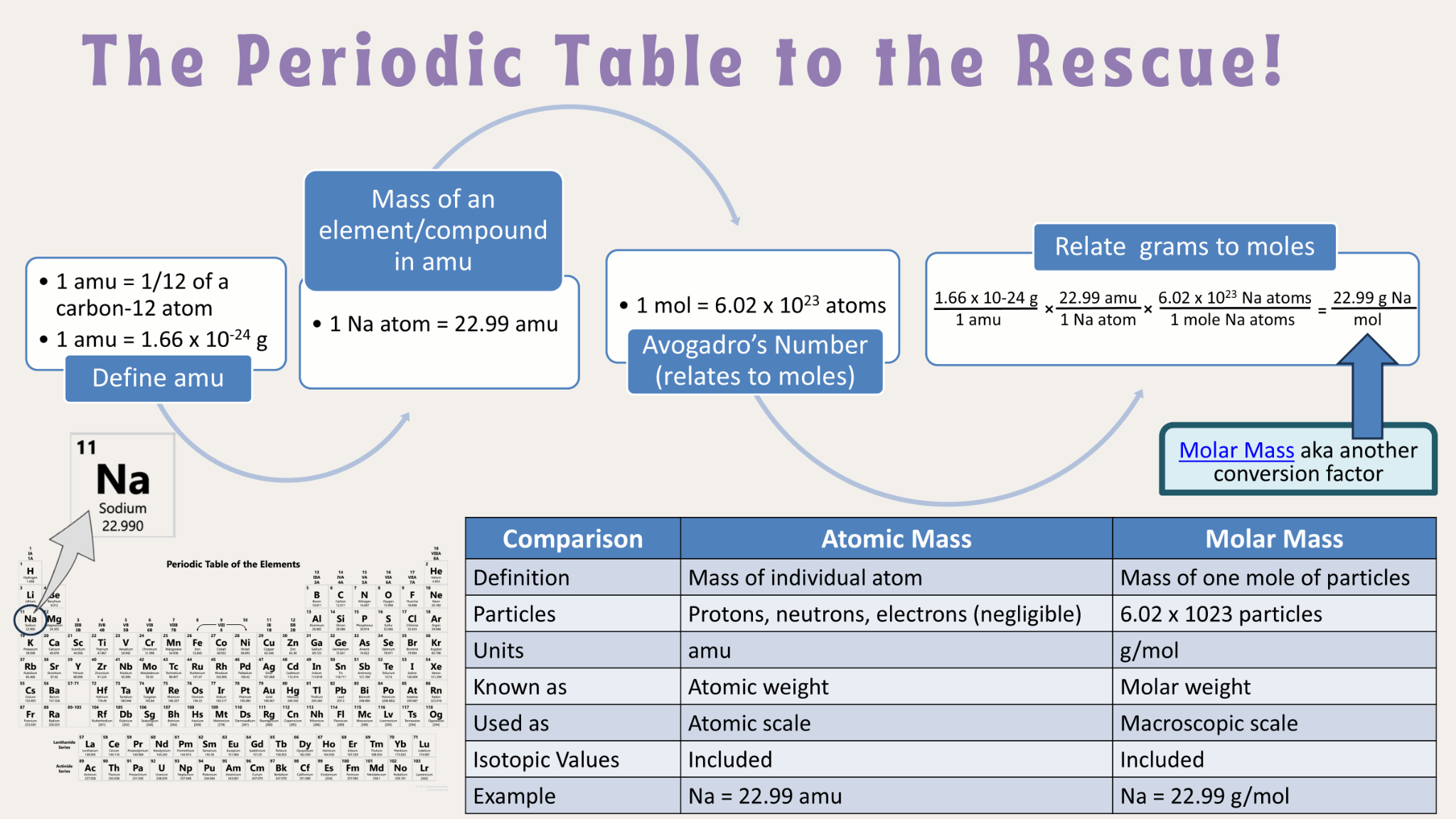
The relative atomic mass (A
r) of an element is a dimensionless quantity representing the ratio of the average mass of an atom of that element to the unified atomic mass unit. The mass of an atom is primarily determined by the combined mass of its protons and neutrons, as the mass of electrons is negligible in comparison. Given that the actual masses of individual atoms are exceedingly small, using relative atomic masses provides a more convenient and practical scale for chemical calculations. The unified atomic mass unit (amu or u) is defined as exactly 1/12th the mass of a single carbon-12 atom, with 1 amu being equivalent to 1.66005402 x 10
-27 kg. Since relative atomic masses are ratios, they are unitless. When referencing relative atomic masses from the IB Data Booklet, it is standard practice to express these values to two decimal places.
Calculating Relative Atomic Mass from Isotopic Abundance
The relative atomic mass of an element is a weighted average of the masses of its naturally occurring isotopes, taking into account their relative abundances.
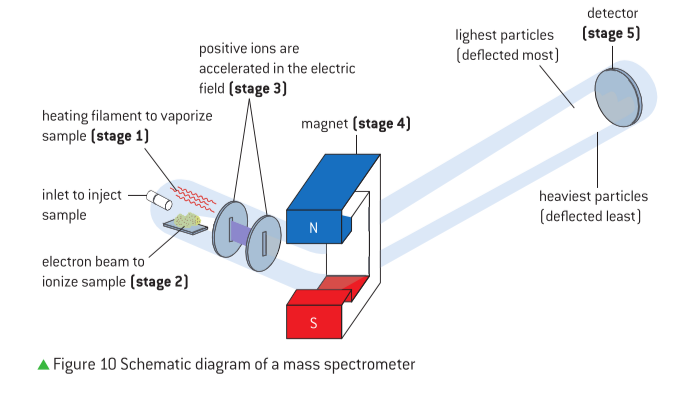
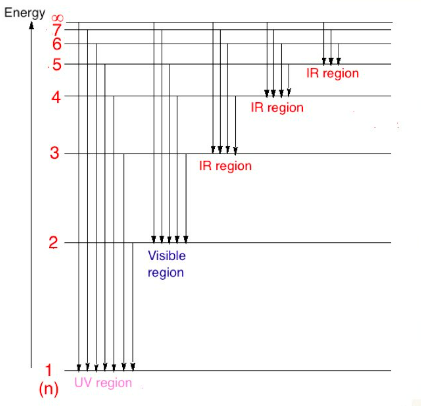
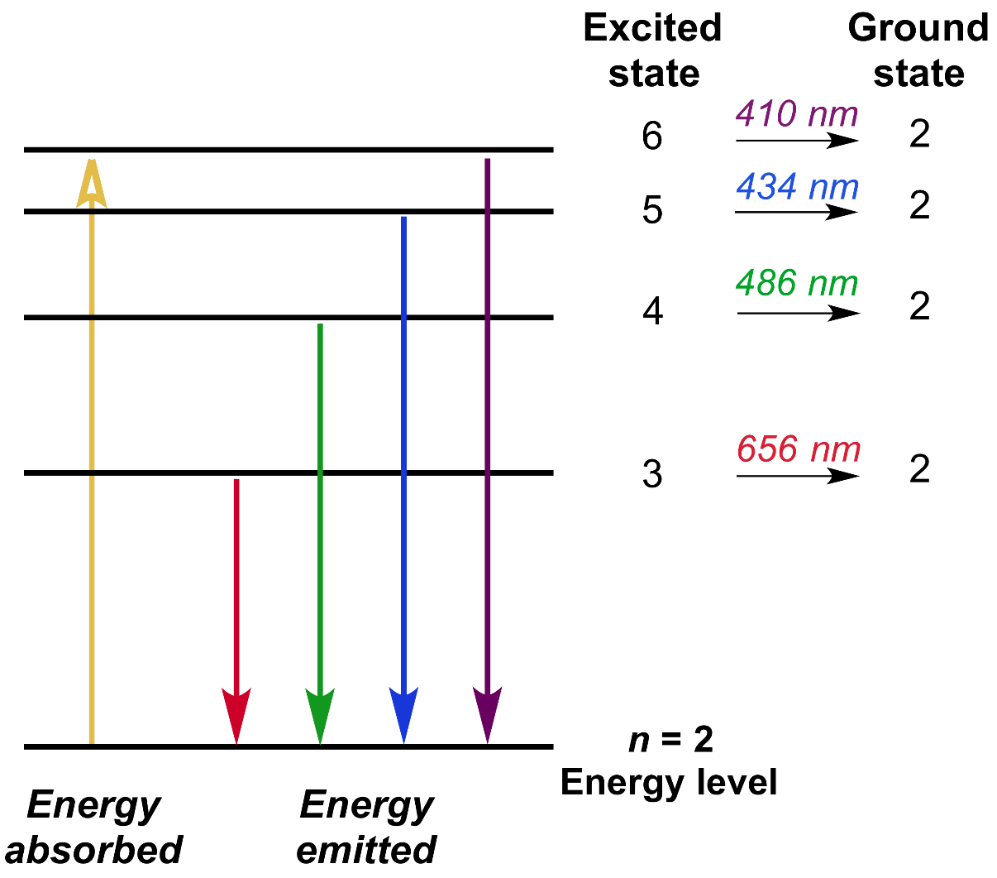
Measuring Atomic Mass and Isotopic Composition with Mass Spectrometry
Mass spectrometry is a powerful analytical technique employed to measure the masses of atoms and molecules and determine the isotopic composition of elements. This method works by ionizing particles and then separating them based on their mass-to-charge ratio (m/z). The output of a mass spectrometer is a mass spectrum, where each peak corresponds to a specific isotope, and the height of each peak is directly proportional to the relative abundance of that isotope.
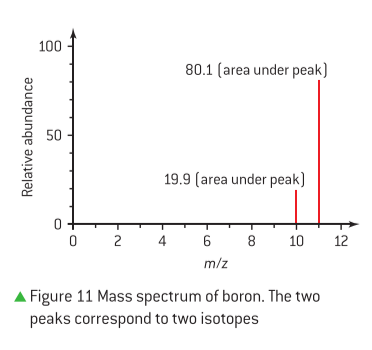
Determining Relative Atomic Mass from a Mass Spectrum
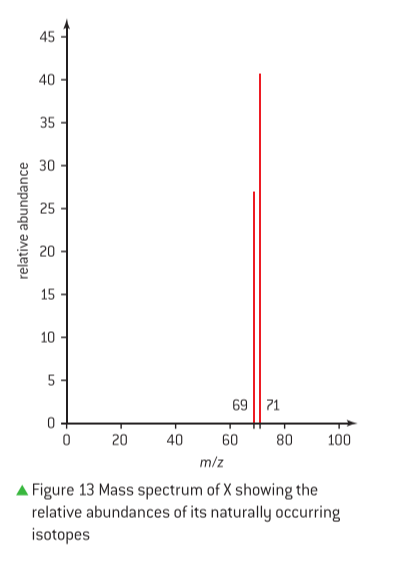
To deduce the relative atomic mass of an element from its mass spectrum, one must first determine the relative abundance of each isotope. For example, consider an element X with two isotopes, X-69 and X-71, as shown in a mass spectrum. If the peak height for X-69 is 27 and for X-71 is 41, the total area of the peak heights is 27 + 41 = 68.
The relative abundance of each isotope can then be calculated:
- For X-69: (27 / 68) x 100 = 40%
- For X-71: (41 / 68) x 100 = 60%
The relative atomic mass is calculated by multiplying the mass of each isotope by its fractional abundance and summing these values:
Relative atomic mass = (0.40 x 69) + (0.60 x 71) = 27.6 + 42.6 = 70.2.
Rounding to an appropriate number of significant figures, typically determined by the precision of the given data, this value is approximately 70. By comparing this relative atomic mass to the periodic table, element X can be identified as Gallium (Ga). It is crucial to apply the correct number of significant figures when performing these mathematical calculations, as dictated by the problem's context.