Lesson Slideshow Section 01
Unit Summary Section 02
Key Concepts
Brønsted-Lowry Theory, pH Scale, Strong vs Weak Acids, Buffer Solutions, Ka/Kb/Kw
Learning Objectives
Explore Brønsted-Lowry theory, the pH scale, and the properties of strong and weak acid/base systems.
Prior Knowledge Needed
Equilibrium, stoichiometry, hydrogen ion structure
IB Syllabus Reference
R3.1
Interactive Study Guide
Click here to view the full, detailed topic summary for this unit.
29
Cu
63.546
Video Lesson Section 03
Video Instruction
5 Lessons Available
Arrhenius Acids_Bases and Acid Base Reactions
Bronsted Lowry Acids_Bases and Strength of Acids_Bases
Environmental Effects of Acid Rain
Lewis Acids_Ka
Salt Hydrolysis_Buffers
Practice Quiz Section 04 · Interactive
10
Ne
20.180
Vocabulary Review Section 05
acidic buffer
A buffer with a pH of less than 7 containing a mixture of a weak acid with one of its salts (or a weak acid partially neutralised with a strong base).

basic buffer
A buffer with a pH of more than 7 containing a mixture of a weak base with one of its salts.

buffer
A solution that minimizes changes in pH when extraneous acids or bases are added to the solution.

buffering capacity
the ability of a buffered solution to absorb protons or hydroxide ions without a significant change in pH; determined by the magnitudes of [HA] and [A-] in the solution; affected by the dilution of the buffer

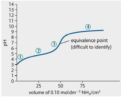
equivalence point
stoichiometrically equal amounts of acid and base have neutralized each other; occurs halfway up the jump in inflection in a titration
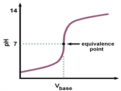
anion hydrolysis
when the anion of a weak acid reacts with water to make a weakly basic solution

cation hydrolysis
when the cation of a weak base reacts with water to make a weakly acidic solution

inflection point
the point in a titration where there is a large jump in pH
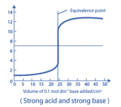
strong acid/strong base titration
Equivalence point will be at pH 7
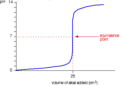
strong acid/weak base titration
Equivalence point < 7. Curve ends at the pH of the weak base.
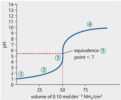
Equivalence point > 7. Curve ends at the pH of the strong base.

weak acid/weak base titration
No clearly defined equivalence point
indicator
Weak acids/bases that can reversibly change color when the pKa=pH of the solution.

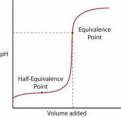
half-equivalence point
The point in a titration of a weak acid/strong base at which exactly half the molar equivalence of reactant is consumed by the titrant being added. At this point in an acid-base titration, the pKa = pH
ocean buffer system
” ” CO2 (g)+ H2O(l) H2CO3 (aq) H+ (aq)+HCO3- (aq) Helps maintain the ocean pH by absorbing excess hydrogen ions produced by carbonic acid.

Lewis acid
electron pair acceptor
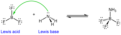
Lewis base
electron pair donor
nucleophile
An atom (or group of atoms) that is attracted to an electron- deficient centre or atom, where it donates a pair of electrons to form a new covalent bond.
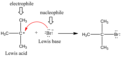
electrophile
An atom (or group of atoms) that is attracted to an electron-rich centre or atom, where it accepts a pair of electrons to form a new covalent bond.
coordinate bond
A shared pair of electrons which has been provided by one of the bonding atoms only; also called a dative covalent bond.

Acid dissociation constant
the ratio of the concentration of the dissociated form of an acid to the concentration of the undissociated form
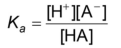
Base dissociation constant
the ratio of the concentration of the conjugate acid times the concentration of the hydroxide ion to the concentration of the base
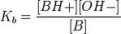
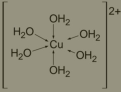
coordination complex
A compound in which a central metal atom or ion is bonded by coordinate covalent bonds to other atoms or groups.
polyprotic

pKa
-log(Ka)
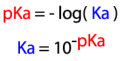
pKb
-log(Kb)
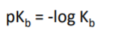
ligand
a molecule or ion that forms a co-ordinate bond with a transition metal by donating a pair of electrons
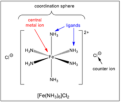
coordination number
the number of coordinate bonds that occur between ligands and a transition metal in a coordination complex

26
Fe
55.845
Revision Notes Section 06
79
Au
196.967
Practice Tests Section 07
Topic-Based Assessments (HL)
Paper 1A: MCQ Practice (HL)
PDF
Paper 1B: Data & Experiments (HL)
PDF
Paper 2: Short Response (HL)
PDF
Official Markscheme (HL)
PDF
17
Cl
35.45
Assignments & Labs Section 08