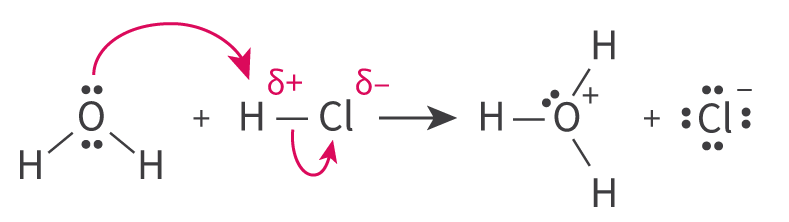
Defining Lewis Acids and Bases
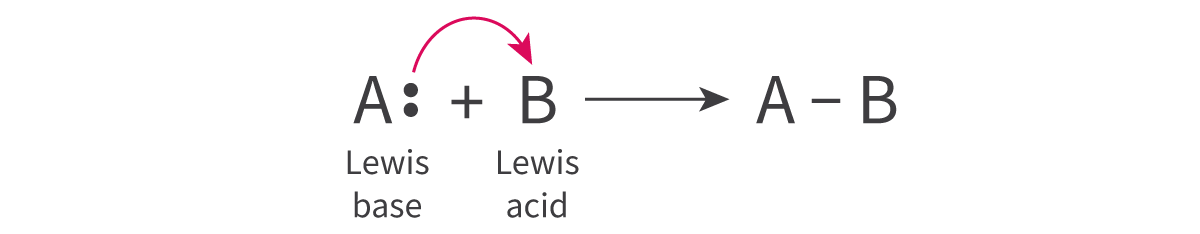
In the context of Lewis acid-base theory, a Lewis acid is characterized as a species that can accept a lone pair of electrons, while a Lewis base is a species capable of donating a lone pair of electrons. It is crucial to remember that these definitions refer to the movement of an entire electron pair, not just a single electron. The interaction between a Lewis acid and a Lewis base results in the formation of a coordinate bond, where both electrons in the bond are contributed by the Lewis base.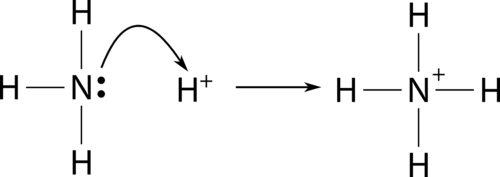
Characteristics of Lewis Acids
Molecules possessing incomplete octets are frequently strong Lewis acids due to their electron deficiency. Furthermore, transition metals often act as Lewis acids because their ions typically have incomplete d orbitals, allowing them to accept electron pairs. These transition metal ions readily bond with ligands to form complex ions. A classic example is the reaction of aqueous copper(II) ions with water molecules: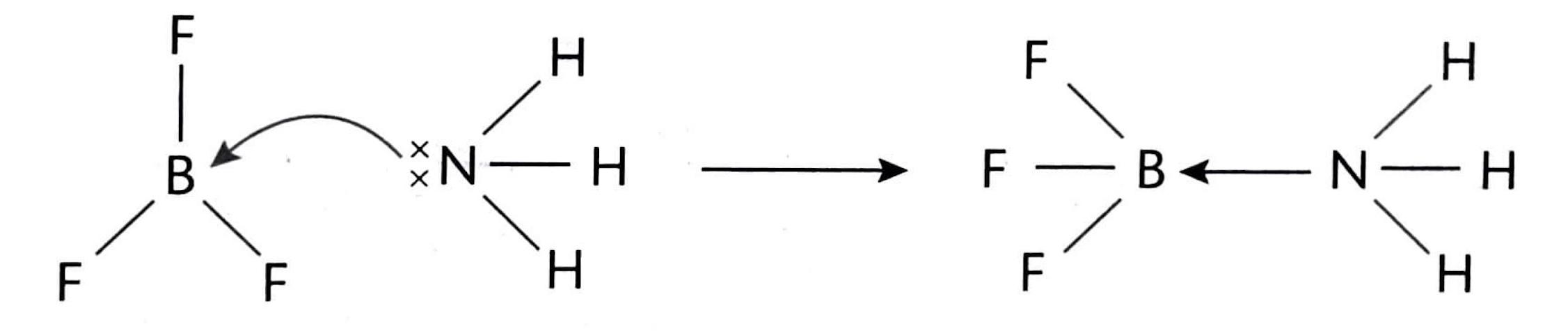
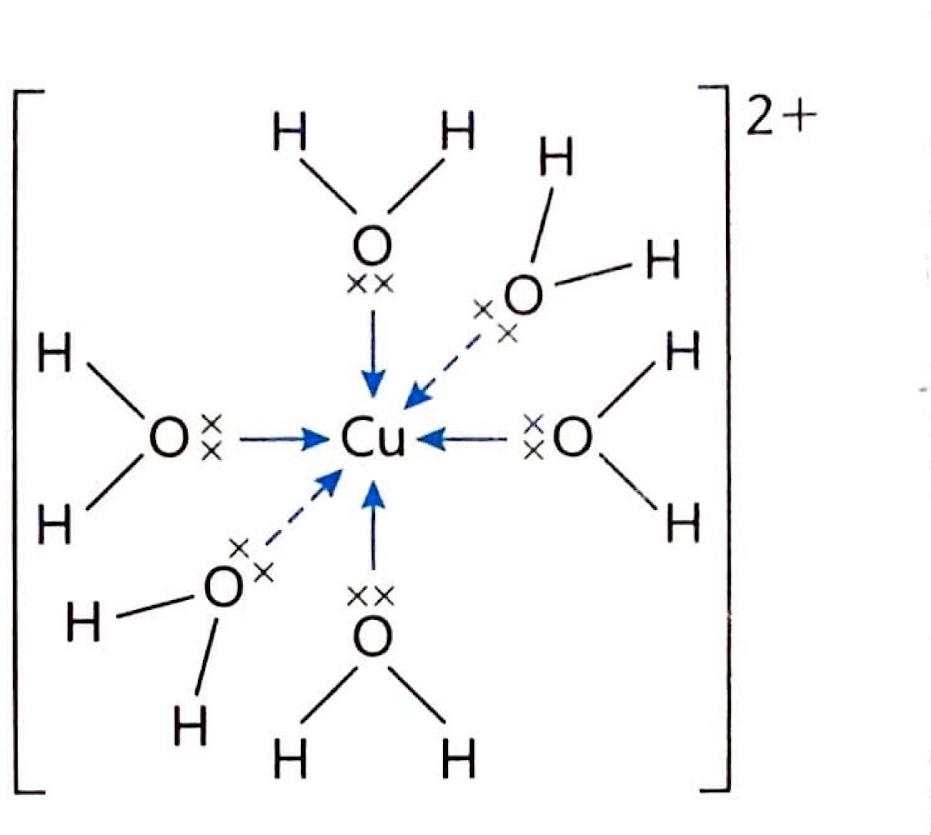
Cu2+(aq) + 6H2O(l) → [Cu(H2O)6]2+(aq). The formation of different complex ions, particularly with copper, is often associated with distinct and vibrant colors.

Ligands, Complexes, and Coordination Number
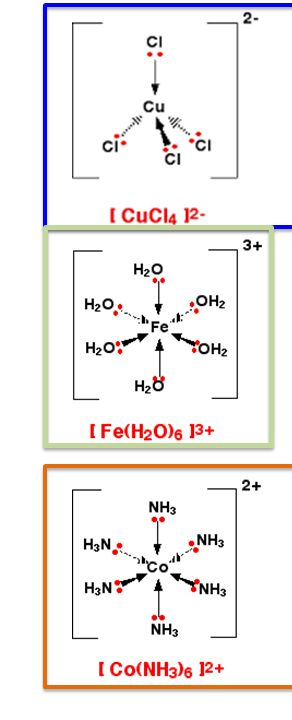
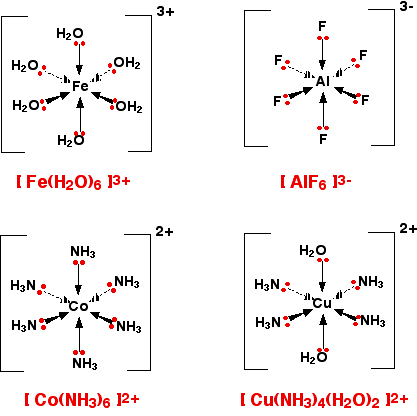
A ligand is defined as a molecule or ion that forms a coordinate bond with a central transition metal atom or ion by donating a pair of electrons. The resulting structure, where a transition metal is centrally located and surrounded by these ligands, is known as a complex. The coordination number of a complex refers to the total number of coordinate bonds formed between the ligands and the central transition metal.
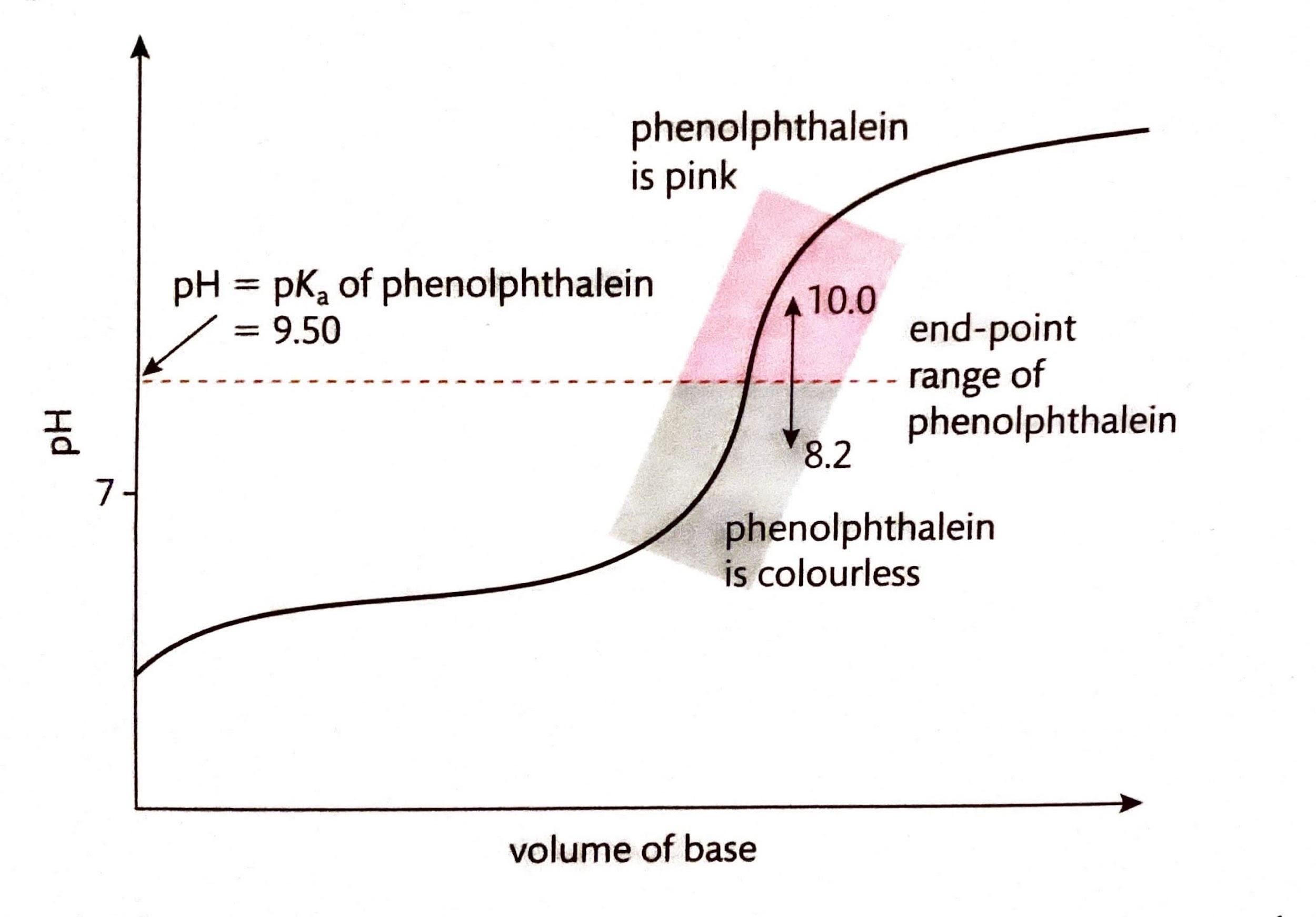
Coloration of Copper Ions
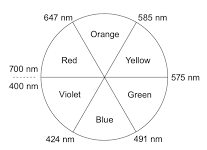
The hexaaquacopper(II) ion, [Cu(H2O)6]2+, typically presents a blue color in aqueous solution. This observed blue color arises because the solution absorbs light in the orange (red and yellow) region of the electromagnetic spectrum, allowing the complementary blue light to be transmitted or reflected. Information regarding the properties and colors of various ions can often be found in Section 17 of chemistry data booklets.

Understanding Crystal Field Theory
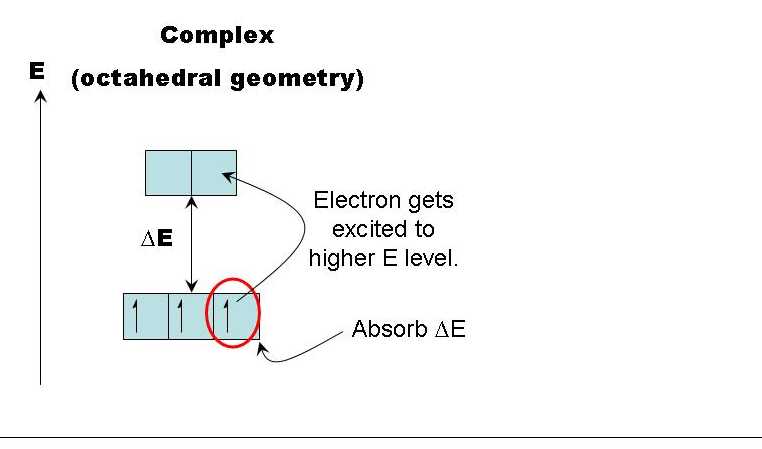
Crystal field theory provides an explanation for the characteristic properties of complex ions, particularly their color and magnetic behavior. This theory posits that when ligands approach a central metal ion, the repulsion between the lone pairs of electrons on the ligands and the electrons in the d orbitals of the central metal ion causes a splitting of the d orbitals into two sets of different energy levels. This uneven repulsion results in two d orbitals having higher energy and three d orbitals having lower energy.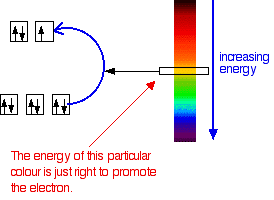
Incomplete d Subshells in Transition Metals
Transition metals are characterized by having incomplete d subshells, which is a key factor contributing to their ability to form complex ions and exhibit variable oxidation states.Transition Metals as Lewis Acids in Complex Formation
As previously mentioned, transition metals frequently act as Lewis acids due to their incomplete d orbitals, enabling them to accept electron pairs from ligands. This interaction leads to the formation of complex ions, such as the reaction between aqueous copper(II) ions and water to form the hexaaquacopper(II) complex: Cu2+(aq) + 6H2O(l) → [Cu(H2O)6]2+(aq). The specific ligands and the central metal ion determine the distinct colors observed for different complex ions of copper.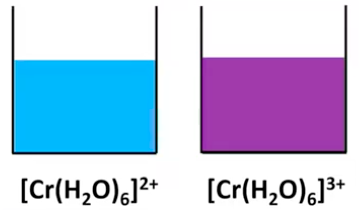
Factors Influencing Complex Ion Colors
The color of a complex ion is directly related to the energy difference between the split d orbitals. Any factor that alters this energy difference will consequently change the wavelengths of light absorbed when an electron transitions from a lower-energy d orbital to a higher-energy d orbital. Key factors influencing complex ion colors include: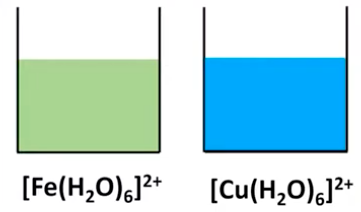
• The oxidation state of the central metal ion.
• The geometry of the complex ion.
• The identity of the ligands, which can be ordered according to the spectrochemical series based on their ability to cause d-orbital splitting.
Determining the Charge of Complex Ions
The overall charge of a complex ion is determined by the sum of the charges of the central metal ion and all the ligands attached to it. For instance, if a central metal ion M has a charge of +2 (e.g., Cu2+) and is coordinated with six neutral ligands (e.g., H2O), the overall charge of the complex will be +2. However, if the same Cu2+ ion were coordinated with two hydroxide ligands (OH-), the overall charge would be 2+ + 2(-1) = 0, resulting in a neutral complex.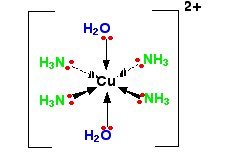
Nucleophiles and Electrophiles
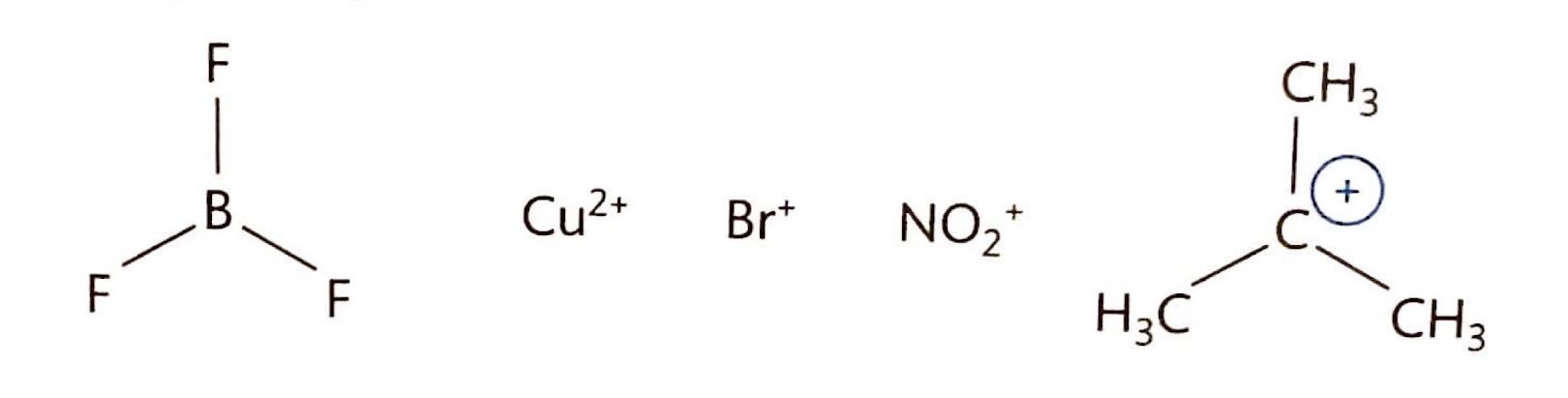
Nucleophiles are electron-rich species that donate a lone pair of electrons to form a new covalent bond in a chemical reaction; they are essentially Lewis bases. Conversely, electrophiles are electron-deficient species that accept a lone pair of electrons from another reactant to form a new covalent bond; thus, they function as Lewis acids.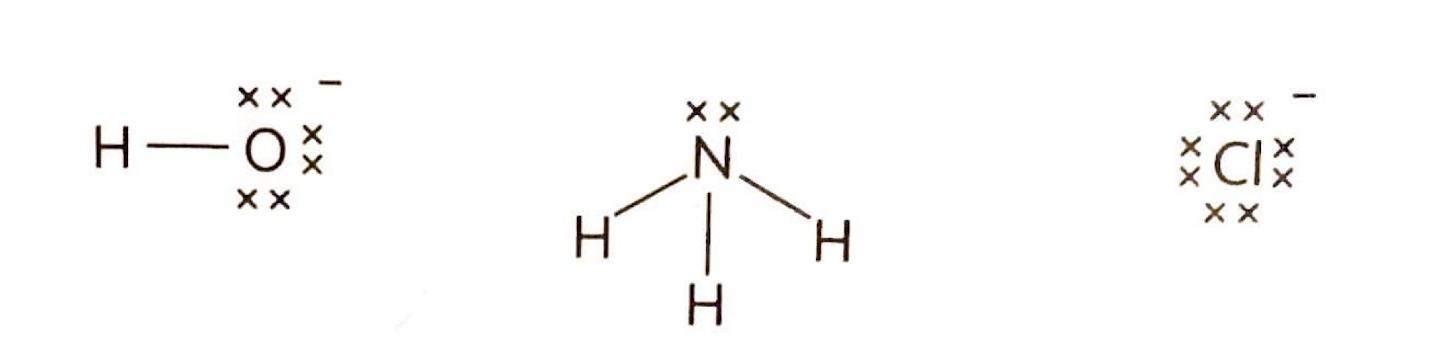
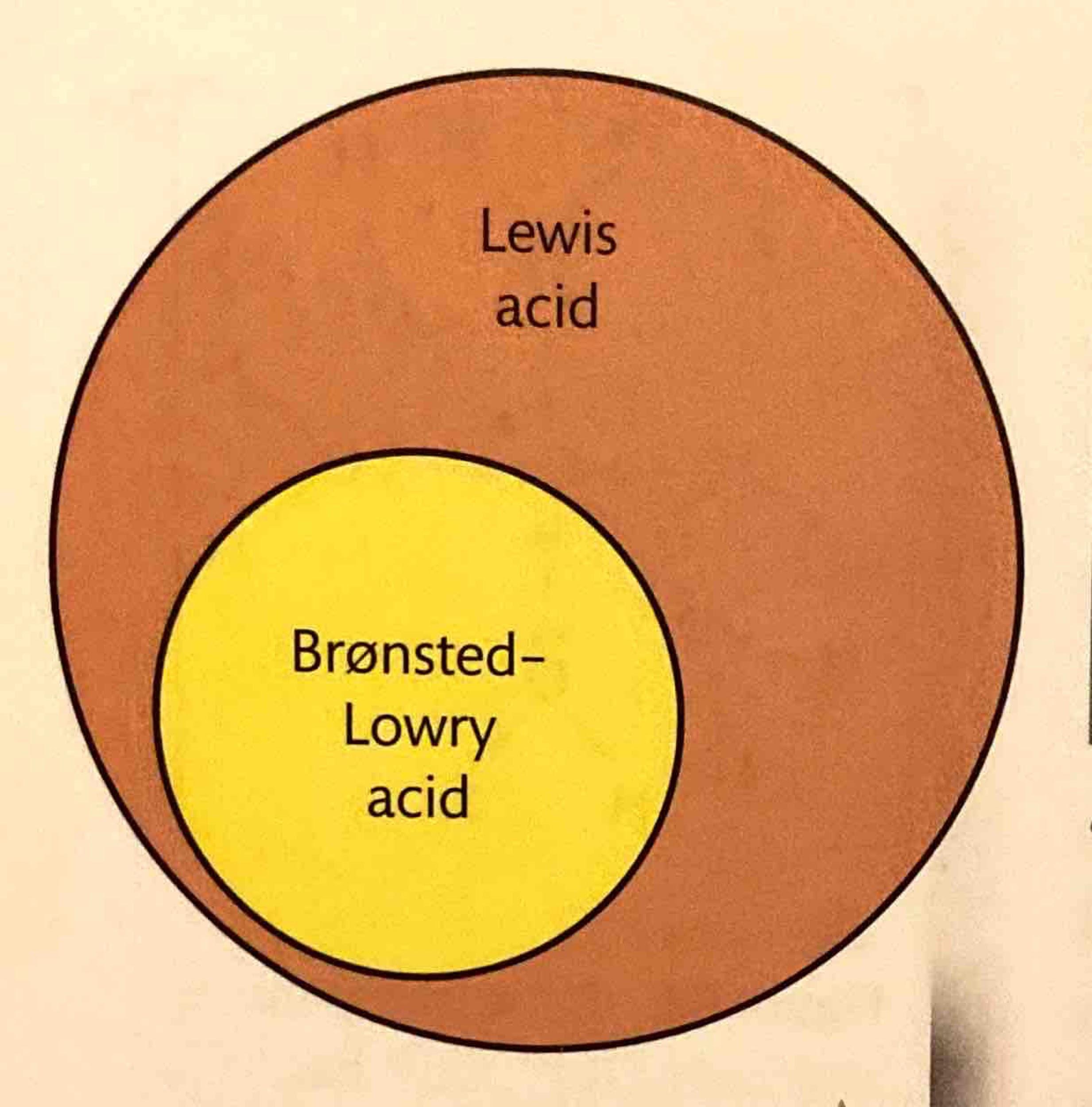
Relationship Between Brønsted-Lowry and Lewis Theories
It is important to note that all Brønsted-Lowry acids are also Lewis acids, as a proton donor (Brønsted-Lowry acid) can accept an electron pair. However, not all Lewis acids are Brønsted-Lowry acids, as Lewis acids encompass a broader range of electron-pair acceptors that may not necessarily donate a proton. Similarly, Lewis bases are electron-pair donors. Some substances, like aluminum oxide (Al2O3), can exhibit both acidic and basic properties depending on the reaction environment, making them amphoteric. For example, Al2O3(s) reacts with a strong base like NaOH(aq) to form NaAlO2(aq) and 2H2O(l). While some Lewis acids/bases are amphoteric, they are not necessarily amphiprotic (meaning they don't necessarily donate or accept protons). The following table summarizes the definitions of acids and bases according to Brønsted-Lowry and Lewis theories:| Theory | Definition of acid | Definition of base |
|---|---|---|
| Brønsted-Lowry | Proton donor | Proton acceptor |
| Lewis | Electron pair acceptor | Electron pair donor |