Electrolytic Cells: Principles and Applications
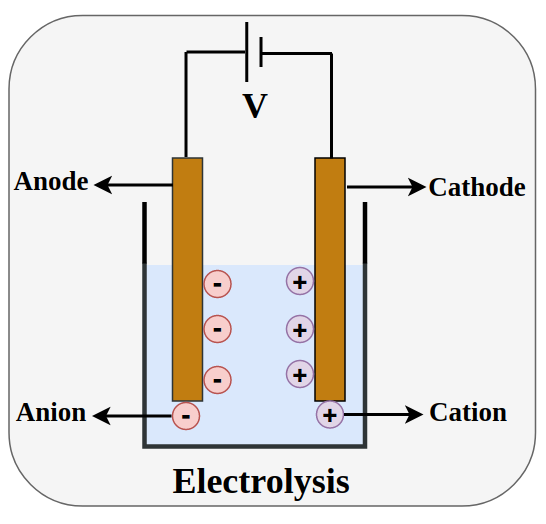
Electrolytic cells utilize an external power source to drive non-spontaneous redox reactions. This power source generates an electric current that flows through an electrolyte, which is a substance containing free ions. As the current passes, ions migrate to electrodes (typically made of metals or graphite) where they undergo oxidation or reduction, forming electrically neutral products. Electrolysis is a crucial industrial process, notably employed for extracting metals from their ores, such as aluminum from aluminum oxide (Al
2O
3) and sodium from sodium chloride (NaCl). These extractions are often necessary because the metal ions have very low standard electrode potentials (E
⚬), meaning there are no sufficiently strong reducing agents available to reduce them chemically.
Understanding the Components of an Electrolytic Cell
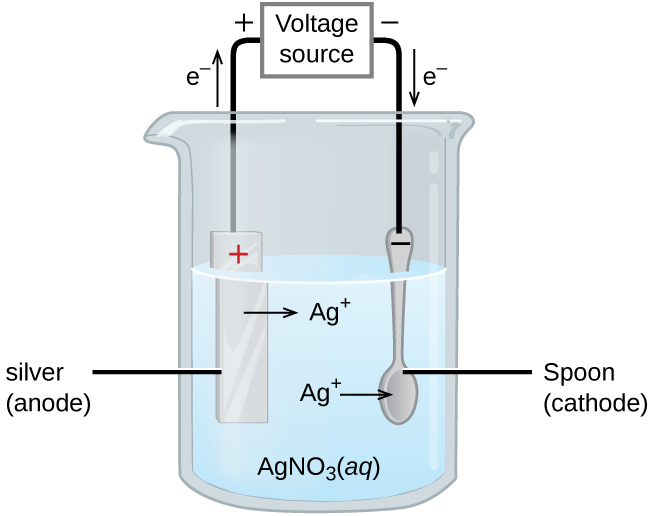
In an electrolytic cell, the power source is represented diagrammatically with a longer line indicating the positive terminal and a shorter line representing the negative terminal. The battery or power source actively pushes electrons towards the negative electrode, which is known as the cathode. Conversely, current is carried through the electrolyte by the movement of ions. At the positive electrode, called the anode, electrons are released from anions and then returned to the power source, completing the circuit.
Electrode Reactions in Electrolytic Cells
Despite the anode being positively charged and the cathode negatively charged in an electrolytic cell, the fundamental definitions of oxidation and reduction remain consistent: oxidation always occurs at the anode, and reduction always occurs at the cathode. This occurs because cations, being positively charged, are attracted to the negative cathode, where they gain electrons and undergo reduction (e.g., M
+ + e
- → M). Conversely, anions, being negatively charged, are attracted to the positive anode, where they lose electrons and undergo oxidation (e.g., A
- → A + e
-).
Example: Electrolysis of Molten Lead(II) Bromide
Consider the electrolysis of molten lead(II) bromide, PbBr
2(l). When molten, lead(II) bromide dissociates into its constituent ions: Pb
2+(l) and 2Br
-(l). During electrolysis, the positively charged lead(II) ions (Pb
2+) migrate towards the cathode, while the negatively charged bromide ions (Br
-) move towards the anode.
At the cathode, reduction occurs:
Pb
2+(l) + 2e
- → Pb(l)
At the anode, oxidation occurs:
2Br
-(l) → Br
2(l) + 2e
-
The overall reaction for the electrolysis of molten lead(II) bromide is:
Pb
2+(l) + 2Br
-(l) → Pb(l) + Br
2(l)
As a result of these reactions, observable changes would include the formation of a brown liquid with a strong, pungent smell, characteristic of bromine (Br
2), at the anode. Simultaneously, a grey, metallic substance, which is lead (Pb), would be deposited at the cathode.
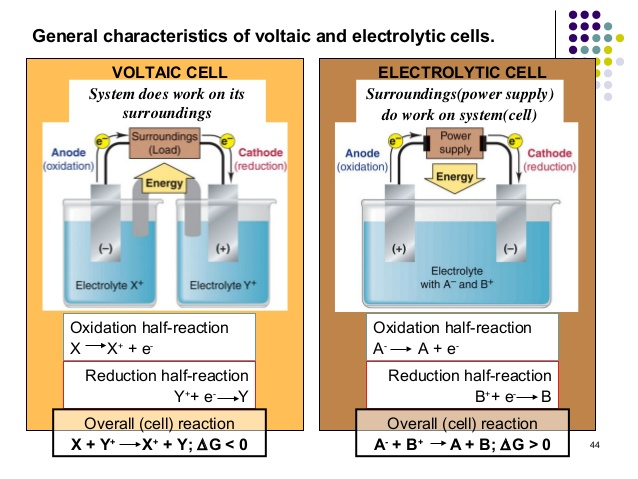
Distinguishing Voltaic and Electrolytic Cells
Voltaic (or galvanic) cells and electrolytic cells represent two fundamental types of electrochemical cells, differing primarily in their spontaneity and energy conversion.
The key differences between these two cell types can be summarized as follows:
|
Voltaic Cell |
|
Electrolytic Cell |
| Anode |
Oxidation occurs here |
negative |
Oxidation occurs here |
positive |
| Cathode |
Reduction occurs here |
positive |
Reduction occurs here |
negative |
Economical Considerations in Electrolysis
Many ionic compounds, particularly metal oxides and halides, possess very high melting points. To make their electrolysis more economically viable, it is often necessary to lower the melting point of the electrolyte. This is achieved by adding other compounds, provided they do not interfere with the desired discharge of the metal ion. For instance, calcium chloride (CaCl
2) can be added to sodium chloride (NaCl) to reduce its melting point.
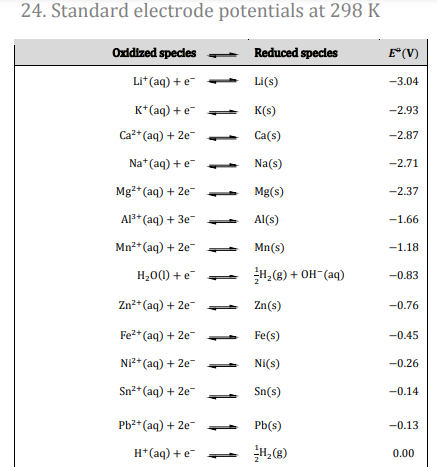
To ensure that the added compound does not interfere, the standard electrode potentials (E
⚬) of the ions involved are carefully considered. For example, when considering the electrolysis of NaCl with added CaCl
2:
Ca
2+(aq) + 2e
- → Ca(s) E
⚬ = -2.87 V
Na
+(aq) + e
- → Na(s) E
⚬ = -2.71 V
Since Na
+ has a higher (less negative) electrode potential than Ca
2+, Na
+ ions will be preferentially reduced at the cathode over Ca
2+ ions. This means that the addition of CaCl
2 will not interfere with the production of sodium metal.
Electrolysis of Aqueous Solutions
Predicting the products of electrolysis becomes more complex when aqueous solutions are involved, as water itself can undergo redox reactions at the electrodes. Water can be oxidized at the anode to produce oxygen gas or reduced at the cathode to produce hydrogen gas.
The relevant half-reactions for water are:
Reduction at cathode: 2H
2O(l) + 2e
- → H
2(g) + 2OH
-(aq)
Oxidation at anode: 2H
2O(l) → 4H
+(aq) + O
2(g) + 4e
-
The selective discharge of ions and water at the electrodes depends on several factors:
- The relative standard electrode potentials (E⚬) of all species present.
- The relative concentrations of the ions in the electrolyte.
- The nature of the electrodes (e.g., inert or reactive).
Guidelines for Predicting Electrode Reactions
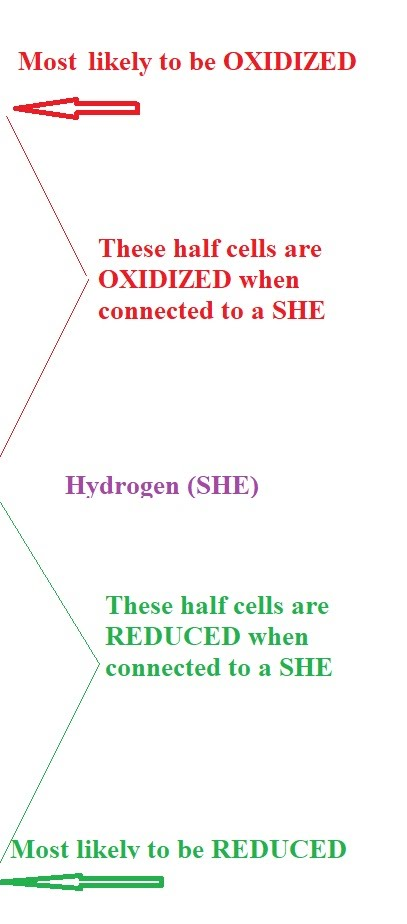
When multiple reactions are possible at an electrode, the following general rules apply based on standard electrode potentials:
- At the cathode (reduction): The species with the larger (more positive) E⚬ value will be preferentially reduced.
- At the anode (oxidation): The species with the smaller (more negative) E⚬ value (or the one that is more easily oxidized) will be preferentially oxidized.
It is crucial to remember that when comparing electrode potentials to determine which species is oxidized, one should not reverse the sign of the standard electrode potential, even if writing the reaction as an oxidation. The E
⚬ values are always given as reduction potentials, and their relative magnitudes directly indicate the tendency for reduction.
Standard Half-Reactions for Water
The IB Data Booklet provides specific half-reactions for the reduction of water, which are important for predicting outcomes in aqueous electrolysis:
- 2H+(aq) + ½ O2(g) + 2e- → H2O(l) E⚬ = +1.23 V (This represents the reverse of water oxidation, and can be considered for oxidation at the anode).
- H2O(l) + e- → ½ H2(g) + OH-(aq) E⚬ = -0.83 V (This is the direct reduction of water at the cathode).
It is important to recall that electrolytic cells are non-spontaneous, meaning an external energy input is required to force the reactions to occur. Therefore, at the cathode, the half-reaction with the smaller (more negative) reduction potential will be forced to be reduced if it is the only option or if it is kinetically favored.
Refined Rules for Selective Discharge
To reiterate and clarify the selection process for electrode reactions:
If there are two possible reactions at the cathode, the species with the larger (more positive) standard reduction potential will be preferentially reduced.
If there are two possible reactions at the anode, the species with the smaller (more negative) standard reduction potential (meaning it is more easily oxidized) will be preferentially oxidized.
Example: Electrolysis of Water with Sodium Hydroxide
Consider the electrolysis of water with added sodium hydroxide (NaOH), which acts as an electrolyte to increase conductivity.
Ions present in the solution are Na
+(aq) and OH
-(aq), along with H
2O(l).
Possible reactions at the cathode (reduction):
Na
+(aq) + e
- → Na(s) E
⚬ = -2.71 V
2H
2O(l) + 2e
- → H
2(g) + 2OH
-(aq) E
⚬ = -0.83 V
Comparing the reduction potentials, -0.83 V is larger (less negative) than -2.71 V.
Therefore, water will be preferentially reduced at the cathode.
Possible reactions at the anode (oxidation):
4OH
-(aq) → 2H
2O(l) + O
2(g) + 4e
- E
⚬ = +0.40 V (derived from the reduction of O
2 to OH
-)
2H
2O(l) → 4H
+(aq) + O
2(g) + 4e
- E
⚬ = +1.23 V
Comparing the oxidation potentials (or the reverse of reduction potentials), +0.40 V is smaller (less positive) than +1.23 V. Therefore, hydroxide ions will be preferentially oxidized at the anode.
The overall balanced equation for the electrolysis of water is:
2H
2O(l) → 2H
2(g) + O
2(g)
Observed changes at the electrodes would include the release of oxygen gas at the anode and hydrogen gas at the cathode. Notably, 2 moles of hydrogen gas are released for every 1 mole of oxygen gas. The pH at the anode would decrease due to the production of H
+ (or consumption of OH
-), while the pH at the cathode would increase due to the production of OH
-.
Example: Electrolysis of Aqueous Sodium Chloride (Brine)
Let's examine the electrolysis of aqueous sodium chloride, commonly known as brine.
Ions present: Na
+(aq), Cl
-(aq), and H
2O(l).
Possible reactions at the cathode (reduction):
Na
+(aq) + e
- → Na(s) E
⚬ = -2.71 V
2H
2O(l) + 2e
- → H
2(g) + 2OH
-(aq) E
⚬ = -0.83 V
Water is preferentially reduced at the cathode because its reduction potential (-0.83 V) is higher than that of Na
+ (-2.71 V).
Possible reactions at the anode (oxidation):
2Cl
-(aq) → Cl
2(g) + 2e
- E
⚬ = +1.36 V
2H
2O(l) → 4H
+(aq) + O
2(g) + 4e
- E
⚬ = +1.23 V
Although the standard oxidation potential for water (+1.23 V) is slightly lower than that for chloride ions (+1.36 V), chloride ions are preferentially oxidized, especially when the concentration of NaCl is high (typically above 25%). This is due to overpotential effects, which make the oxidation of water kinetically less favorable than predicted by standard potentials alone.
The overall balanced equation for the electrolysis of concentrated aqueous sodium chloride is:
2NaCl(aq) + 2H
2O(l) → H
2(g) + Cl
2(g) + 2Na
+(aq) + 2OH
-(aq)
Observed changes include the release of chlorine gas (Cl
2) at the anode, which has a strong smell and bleaching effect, and hydrogen gas (H
2) at the cathode. The pH of the solution increases due to the production of hydroxide ions (OH
-).
Example: Electrolysis of Aqueous Copper(II) Sulfate with Inert Electrodes
Let's consider the electrolysis of aqueous copper(II) sulfate (CuSO
4) using inert electrodes, such as graphite.
Ions present: Cu
2+(aq), SO
42-(aq), and H
2O(l).
Possible reactions at the cathode (reduction):
Cu
2+(aq) + 2e
- → Cu(s) E
⚬ = +0.34 V
2H
2O(l) + 2e
- → H
2(g) + 2OH
-(aq) E
⚬ = -0.83 V
Since +0.34 V is significantly larger (more positive) than -0.83 V, copper(II) ions will be preferentially reduced at the cathode.
Possible reactions at the anode (oxidation):
2H
2O(l) → 4H
+(aq) + O
2(g) + 4e
- E
⚬ = +1.23 V
Sulfate ions (SO
42-) cannot be oxidized further because sulfur is already in its maximum oxidation state (+6). Therefore, water will be oxidized at the anode.
The overall balanced equation for this electrolysis is:
2CuSO
4(aq) + 2H
2O(l) → 2Cu(s) + O
2(g) + 4H
+(aq) + 2SO
42-(aq)
Observed changes include the deposition of pink-brown copper metal at the cathode and the release of oxygen gas at the anode. The pH of the solution will decrease due to the production of H
+ ions, and the blue color of the copper(II) sulfate solution will gradually fade as Cu
2+ ions are consumed.
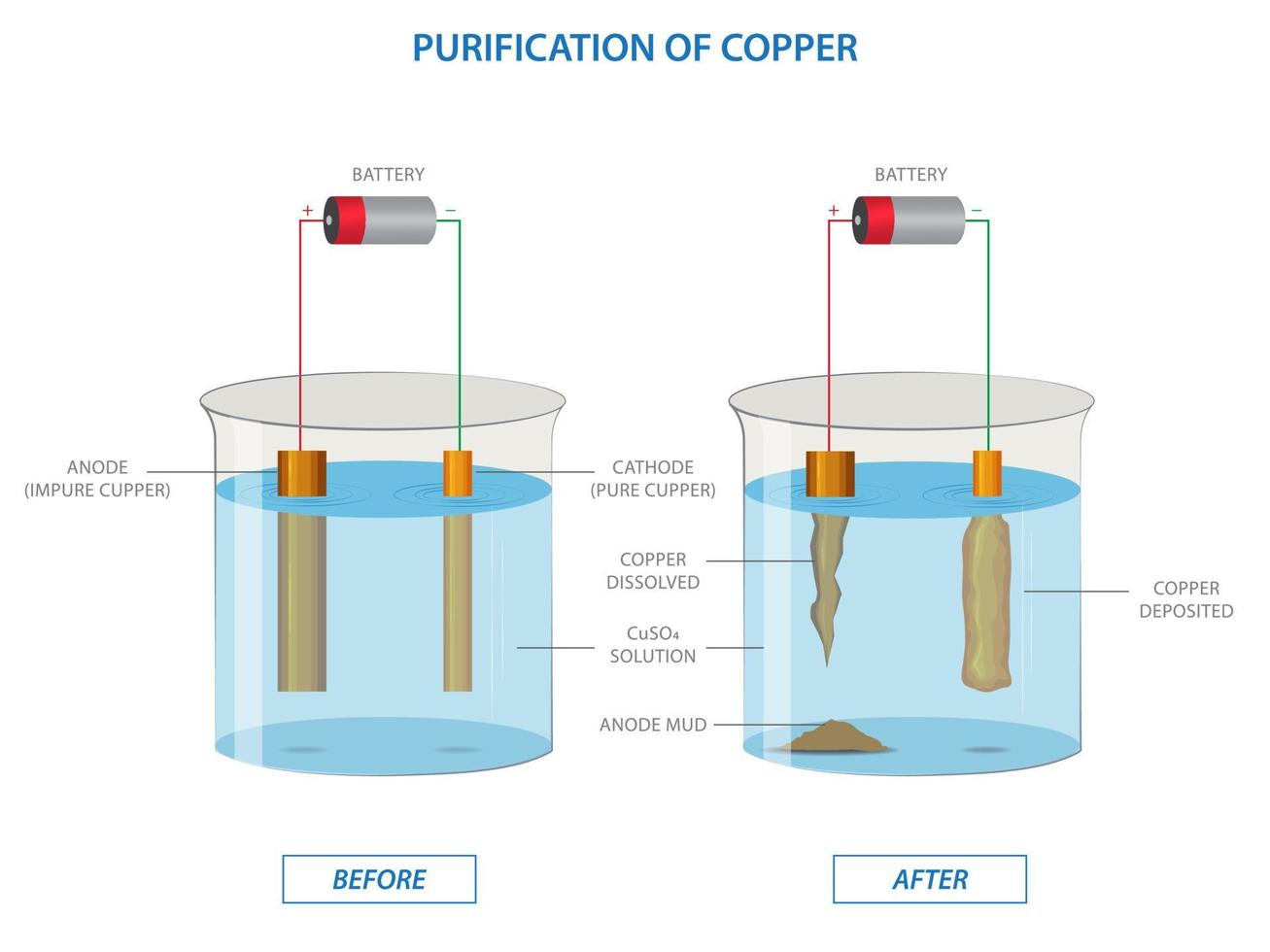
Example: Electrolysis of Aqueous Copper(II) Sulfate with Copper Electrodes
When the electrolysis of aqueous copper(II) sulfate is carried out using active copper electrodes instead of inert ones, the anode itself participates in the reaction.
Ions present: Cu
2+(aq), SO
42-(aq), and H
2O(l).
Possible reactions at the cathode (reduction):
Cu
2+(aq) + 2e
- → Cu(s) E
⚬ = +0.34 V
2H
2O(l) + 2e
- → H
2(g) + 2OH
-(aq) E
⚬ = -0.83 V
As before, copper(II) ions are preferentially reduced at the cathode.
Possible reactions at the anode (oxidation):
Instead of water being oxidized, the copper anode itself will be oxidized because copper is more easily oxidized than water.
Cu(s) → Cu
2+(aq) + 2e
-
In this setup, copper(II) ions are produced at the anode and then migrate to the cathode, where they are discharged as solid copper. This process is commonly used in copper refining.
Observed changes include the deposition of pink-brown copper metal at the cathode and the gradual disintegration of the copper anode. Crucially, there will be no significant change in the pH of the solution and no change in the blue color of the copper(II) sulfate solution, as Cu
2+ ions are consumed at the cathode but replenished at the anode.
Factors Affecting Product Amounts in Electrolysis
The amount of product formed during electrolysis is directly proportional to:
- The magnitude of the current (I) flowing through the cell.
- The duration of the electrolysis (t).
- The charge on the ion (z), which determines the number of electrons required per mole of product.
Calculating Amounts Produced from Electrolysis
Faraday's laws of electrolysis provide the quantitative relationships between charge, current, time, and the amount of substance produced. The total charge (Q) passed through an electrolytic cell is given by:
Q = I × t
where Q is in coulombs (C), I is in amperes (A), and t is in seconds (s).
The relationship between charge and moles of electrons is given by Faraday's constant (F), which is approximately 96500 C mol
-1.
Moles of electrons = Q / F
Once the moles of electrons are known, the moles of product (n) can be determined using the stoichiometry of the balanced half-reaction. Finally, the mass of the product (m) can be calculated using its molar mass (M):
m = n × M
Example: Calculating Copper Deposited
Let's calculate the mass of copper deposited on the cathode of an electrolytic cell containing CuCl
2(aq) if a current of 2.00 A is run for 15.0 minutes.
First, calculate the total charge (Q):
Q = 2.00 A × (15.0 × 60 s) = 1800 C
Next, calculate the moles of electrons:
Moles of e
- = 1800 C / 96500 C mol
-1 = 0.01865 mol e
-
The half-reaction for copper deposition from CuCl
2(aq) is:
Cu
2+(aq) + 2e
- → Cu(s)
From this, 2 moles of electrons are required to produce 1 mole of copper.
Moles of Cu = 0.01865 mol e
- × (1 mol Cu / 2 mol e
-) = 0.00933 mol Cu
Finally, calculate the mass of copper:
Mass of Cu = 0.00933 mol × 63.55 g mol
-1 = 0.593 g Cu
Now, consider how the amount would differ if the same conditions were applied using CuCl(aq) instead.
The half-reaction for copper deposition from CuCl(aq) is:
Cu
+(aq) + e
- → Cu(s)
In this case, 1 mole of electrons is required to produce 1 mole of copper.
Moles of Cu = 0.01865 mol e
- × (1 mol Cu / 1 mol e
-) = 0.01865 mol Cu
Mass of Cu = 0.01865 mol × 63.55 g mol
-1 = 1.185 g Cu
As seen, the mass of copper deposited is approximately double when starting with CuCl(aq) compared to CuCl
2(aq) under the same conditions, due to the difference in the charge of the copper ion.
Electroplating: A Practical Application of Electrolysis
Electroplating is a widely used electrolytic process that involves depositing a thin layer of one metal onto the surface of another metal or a conductive object. This technique is employed for various purposes, including enhancing appearance, improving corrosion resistance, or increasing hardness.
In an electroplating setup:
- The electrolyte contains the metal ions that are to be deposited.
- The object to be plated serves as the cathode, attracting the metal ions for reduction.
- The anode is typically made of the same metal that is being coated. As the anode oxidizes, it replenishes the metal ions in the electrolyte, maintaining a consistent concentration.
A common application of electroplating is galvanization, where metals like iron are coated with a thin layer of zinc to protect them from corrosion.