Evidence for Sublevels from Ionization Energies
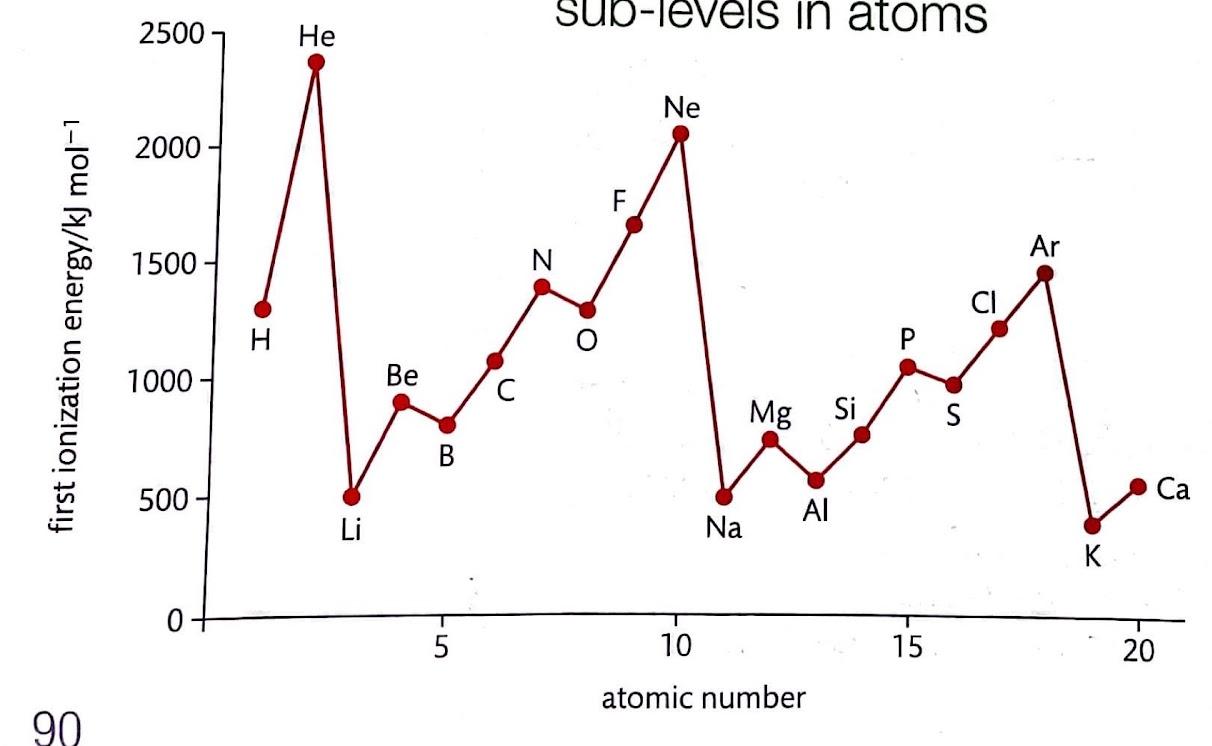
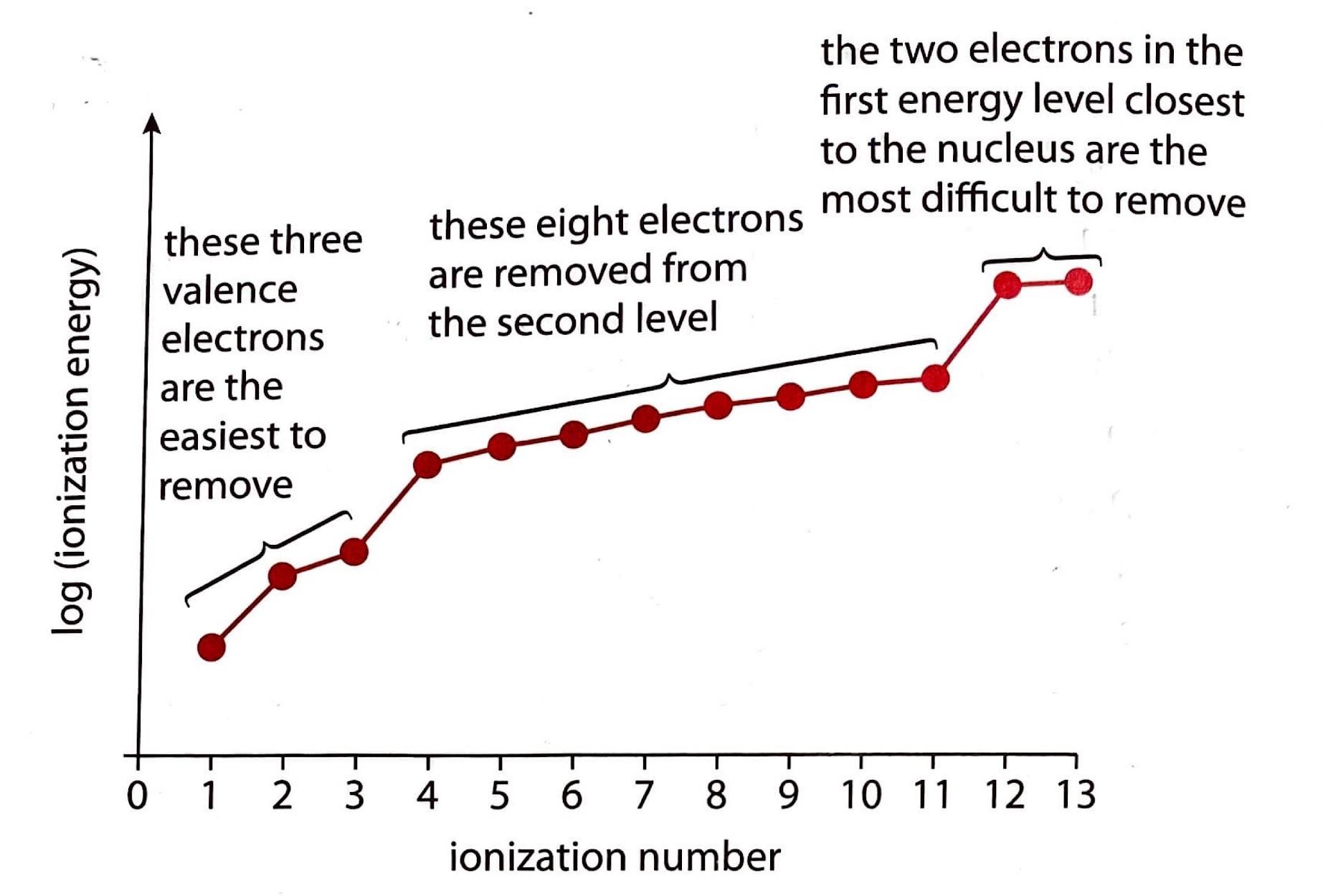
Ionization energies provide compelling evidence for the existence of electron sublevels within an atom's electron shell structure. A significant "jump" in the energy required to remove successive electrons indicates that the electron being removed is from a different, more stable energy level or sublevel. For instance, if we observe a substantial increase between the 9th and 10th ionization energies, it suggests that the 10th electron is considerably more difficult to remove than the preceding nine. This difficulty implies that the 10th electron resides in a more deeply bound sublevel, requiring a greater input of energy to overcome the increased nuclear attraction.
Ionization Energies of Aluminum
The ionization energies for aluminum (Al) clearly illustrate this principle. As electrons are progressively removed from an aluminum atom, the energy required for each subsequent removal generally increases due to the increasing effective nuclear charge on the remaining electrons. However, a particularly large increase, or "jump," in ionization energy signifies the removal of an electron from a new, more stable sublevel.
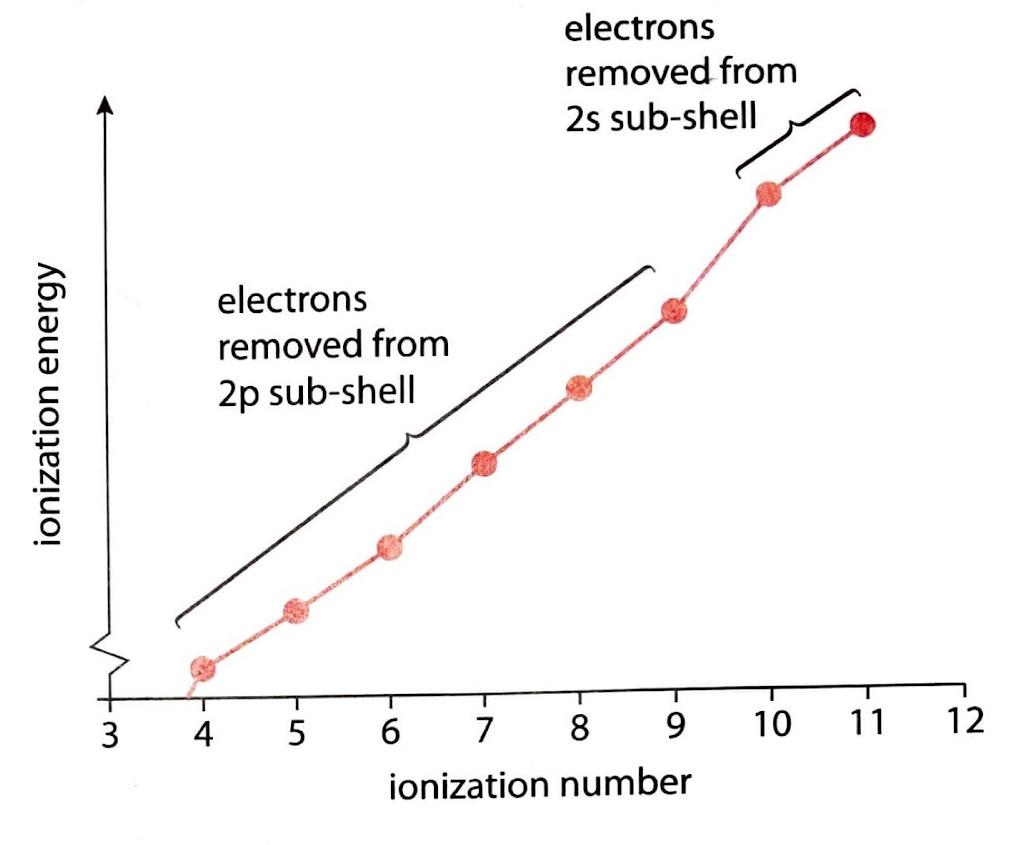
Sublevel Contributions to Ionization Energy Trends
When examining the ionization energies of an element like aluminum, the specific values for each successive electron removal reveal the underlying electron configuration. For example, consider the ionization energies associated with removing electrons from the 2p sublevel. The first three electrons removed from this sublevel (corresponding to the 4th, 5th, and 6th ionization energies overall for an atom with a 2p sublevel) generally require less energy than removing the fourth electron (the 7th ionization energy). This phenomenon is attributed to electron-pair repulsion.
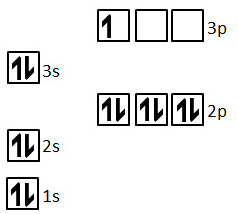
In the 2p sublevel, electrons are paired up after the first three electrons have been removed (assuming a typical filling order). When electrons are paired within an orbital, the electrostatic repulsion between them makes it slightly easier to remove one of these paired electrons compared to an unpaired electron in a half-filled orbital or a new, more stable sublevel. Therefore, the 4th, 5th, and 6th ionization energies, which correspond to removing electrons that are experiencing electron-pair repulsion, are lower than the 7th ionization energy, which might involve removing an electron from a more stable, inner sublevel or an unpaired electron from a different orbital. This pattern provides direct evidence for the distinct energy levels and sublevels within an atom.