1
Condensation Polymerisation
▼
2
Polyesters
▼
Introduction to Polyesters
Polyesters are a significant class of synthetic polymers characterized by the presence of ester linkages (-COO-) along their main chain. These polymers are formed through a condensation polymerization reaction, where monomers combine, and a small molecule, typically water, is eliminated. The general structure of a polyester involves repeating ester units.Monomer Requirements for Polyester Synthesis
The formation of polyesters requires specific types of monomers that can react to form ester bonds. There are two primary approaches to synthesizing polyesters based on the monomer types:- One method involves the reaction between a diol (a molecule with two hydroxyl -OH groups) and a dicarboxylic acid (a molecule with two carboxyl -COOH groups). In this reaction, one hydroxyl group from the diol reacts with one carboxyl group from the dicarboxylic acid to form an ester linkage, with water being eliminated. This process repeats to form a long polymer chain.
- Alternatively, polyesters can be formed from a single type of monomer, a hydroxycarboxylic acid. This monomer contains both a hydroxyl group and a carboxyl group within the same molecule. The hydroxyl group of one monomer reacts with the carboxyl group of another monomer, leading to the formation of an ester linkage and the elimination of water.
Terylene: A Common Polyester Example
Terylene, also known as polyethylene terephthalate (PET), is a widely used polyester with diverse applications. Its synthesis involves specific monomers and results in a strong, versatile polymer. The monomers required for the production of Terylene are:- Ethane-1,2-diol (also known as ethylene glycol), which provides the two hydroxyl groups.
- Benzene-1,4-dicarboxylic acid (also known as terephthalic acid), which provides the two carboxyl groups.
- Manufacturing of synthetic fibers for clothing, carpets, and upholstery due to their durability, wrinkle resistance, and ability to hold shape.
- Production of plastic bottles and food containers because of their strength, transparency, and barrier properties against gases and liquids.
Monomers and Repeat Unit of Terylene
As previously stated, the monomers for Terylene are ethane-1,2-diol and benzene-1,4-dicarboxylic acid. The polymerization of these monomers results in a repeating unit that contains the characteristic ester linkage. The ester linkage is the functional group -COO- that connects the monomer units within the polymer chain.
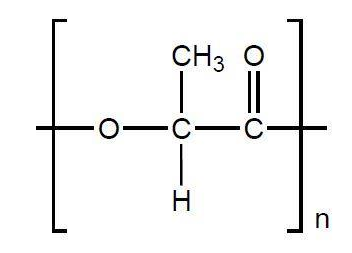
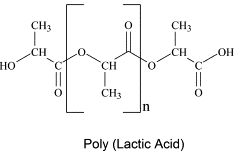
Poly(lactic acid): A Biodegradable Biopolymer
Poly(lactic acid), often abbreviated as PLA,is an important biopolymer that stands out
due to its biodegradable nature. It is typically
produced from renewable resources, such as
maize (corn starch), making it an
environmentally friendly alternative to
traditional petroleum-based plastics. PLA's biodegradability means it can decompose into natural substances under specific conditions, reducing its environmental impact compared to non-biodegradable plastics. This property makes it suitable for various applications, including the production of food and drinks cartons, where its biodegradability is a significant advantage for waste management. The single monomer from which poly(lactic acid) is synthesized is 2-hydroxypropanoic acid, commonly known as lactic acid. This molecule contains both a hydroxyl group and a carboxyl group, allowing it to undergo self-condensation polymerization to form the polyester chain.

3
Polyamides
▼
Formation of Polyamides from Monomers
Polyamides are a class of polymers characterized by the presence of amide linkages (–CONH–) along the polymer chain. These polymers are typically formed through condensation polymerization. To synthesize a polyamide, two main types of monomer combinations can be employed: either a diamine (a molecule with two amine functional groups, –NH2) and a dicarboxylic acid (a molecule with two carboxylic acid functional groups, –COOH), or a single amino acid monomer (a molecule containing both an amine and a carboxylic acid functional group). The reaction between these functional groups results in the elimination of a small molecule, usually water, and the formation of the amide bond.Synthesis and Structure of Nylon-6,6
Nylon-6,6 is a well-known synthetic polyamide. Its name reflects the number of carbon atoms in each of its constituent monomers: six carbons in the diamine and six carbons in the dicarboxylic acid. The specific monomers required for the production of Nylon-6,6 are 1,6-diaminohexane (H2N–(CH2)6–NHincorporates the amide link, which is the defining functional
groupof polyamides.