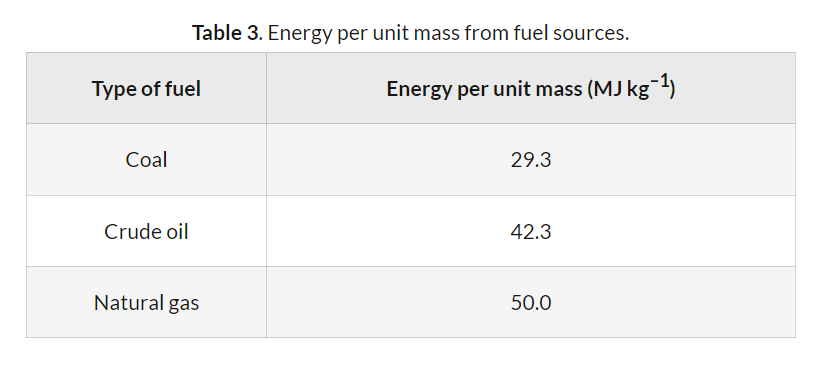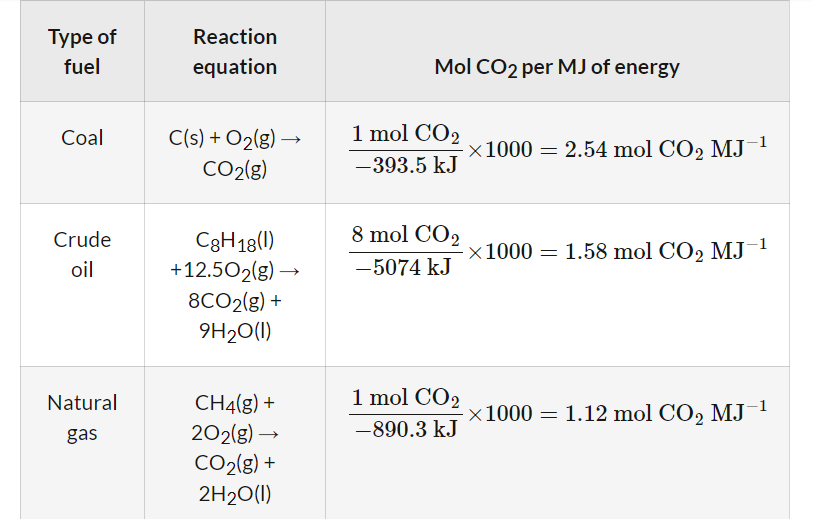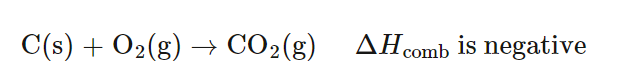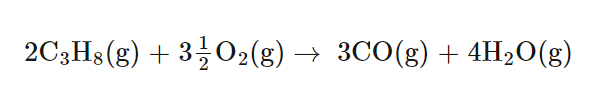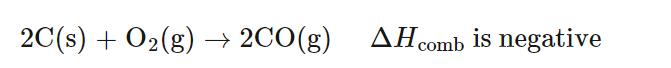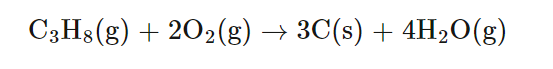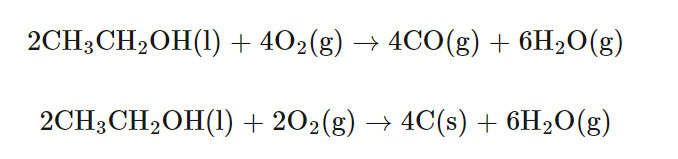The Law of Conservation of Energy and Its Implications for Energy Sources
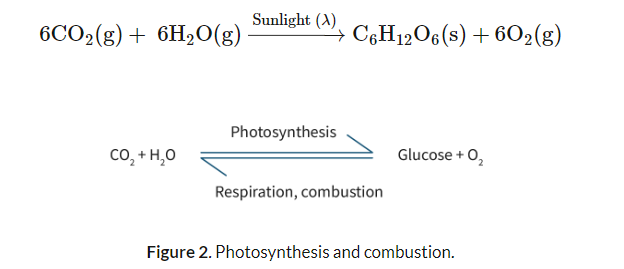
The fundamental principle governing energy is the Law of Conservation of Energy, which states that energy cannot be created or destroyed; it can only be converted from one form to another. However, during these conversions, the "quality" of energy often degrades, meaning that some energy is lost to the environment, typically as heat, and is no longer available to perform useful work. For instance, when fossil fuels are burned, they release high-quality chemical energy, but a significant portion of this energy dissipates as heat into the surroundings, becoming unusable. Therefore, when evaluating energy sources, several criteria are crucial: they should be inexpensive, abundant, easily accessible, capable of providing high-quality energy at a suitable rate, and have a minimal environmental impact. Energy sources that aim to meet these criteria include fossil fuels, nuclear fission, electrochemical cells, solar energy, biomass, and various alternative energy sources like wind and hydroelectric power.
Understanding Fuels and Their Energy Release
A fuel is defined as any substance capable of releasing energy through changes in its chemical or nuclear structure. This inherent ability to release energy makes fuels vital for various applications, from powering vehicles to generating electricity.
Combustion Reactions of Common Fossil Fuels
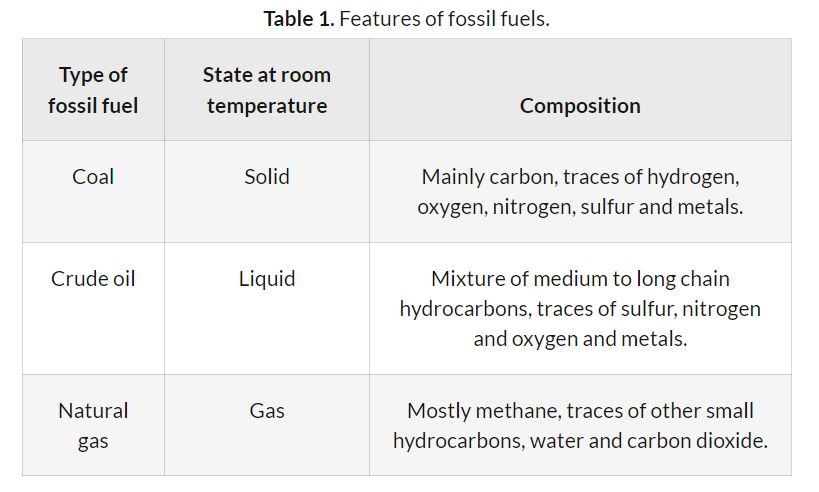
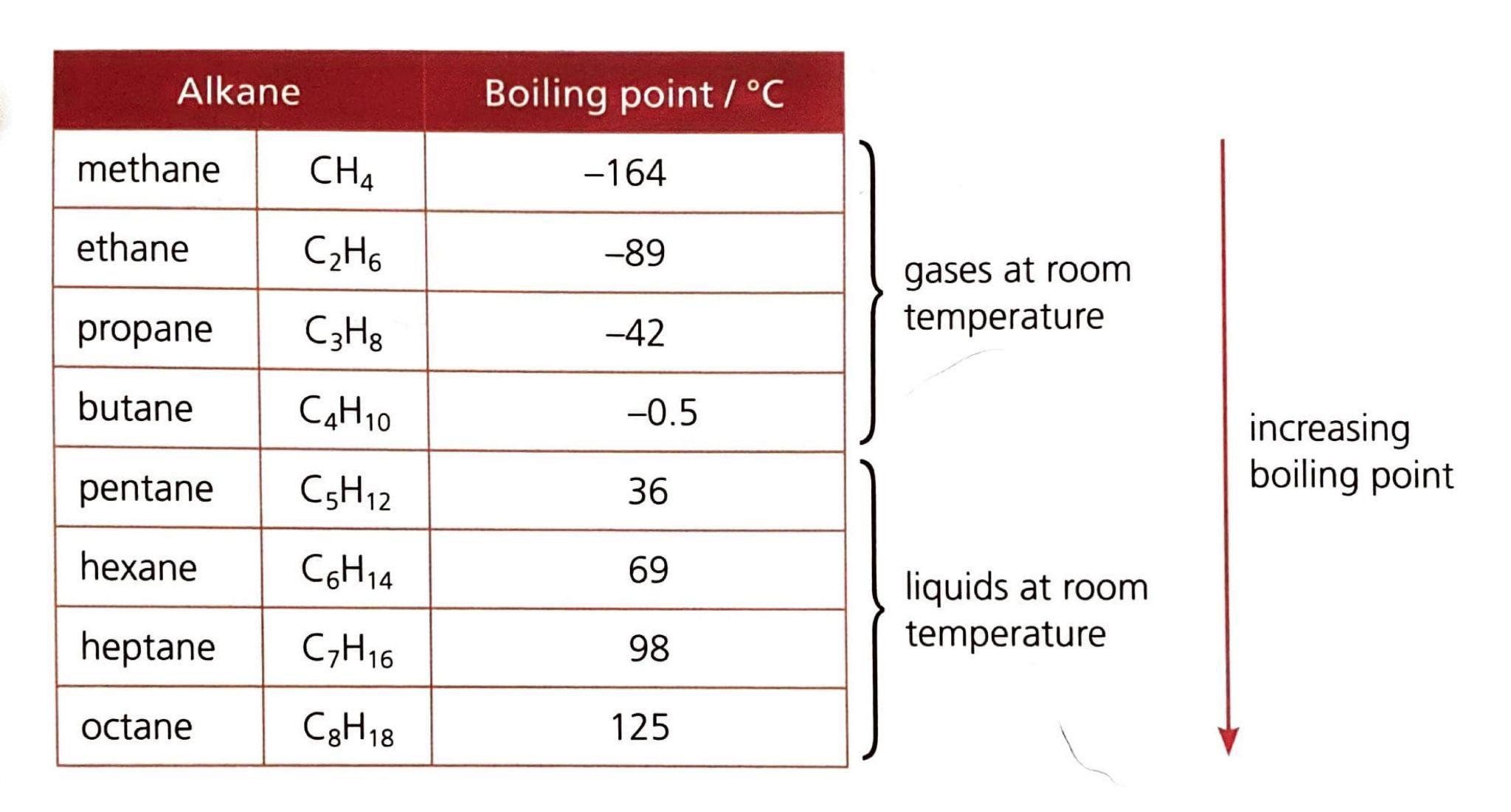
Fossil fuels, such as coal, crude oil, and natural gas, release energy through combustion reactions. The balanced chemical equations for the complete combustion of representative components of these fuels, along with their standard enthalpy changes (ΔH), are as follows:
- For coal, primarily carbon:
C(s) + O2(g) → CO2(g) ΔH = −393.5 kJ
- For crude oil, represented by octane: C8H18(l) + 12.5O2(g) → 8CO2(g) + 9H2O(l) ΔH = −5470 kJ
- For natural gas, primarily methane: CH4(g) + 2O2(g) → CO2(g) + 2H2O(l) ΔH = −890.3 kJ
The efficiency of these combustion processes, which is a measure of how much useful energy is obtained from the total input energy, can be calculated using the formula: Efficiency = (useful output energy / total input energy) × 100. The typical efficiency values for power generation using these fossil fuels are summarized in the table below.
| Fossil Fuel |
Typical Efficiency Values |
| Gas |
40-50% |
| Oil |
35-45% |
| Coal |
35-40% |
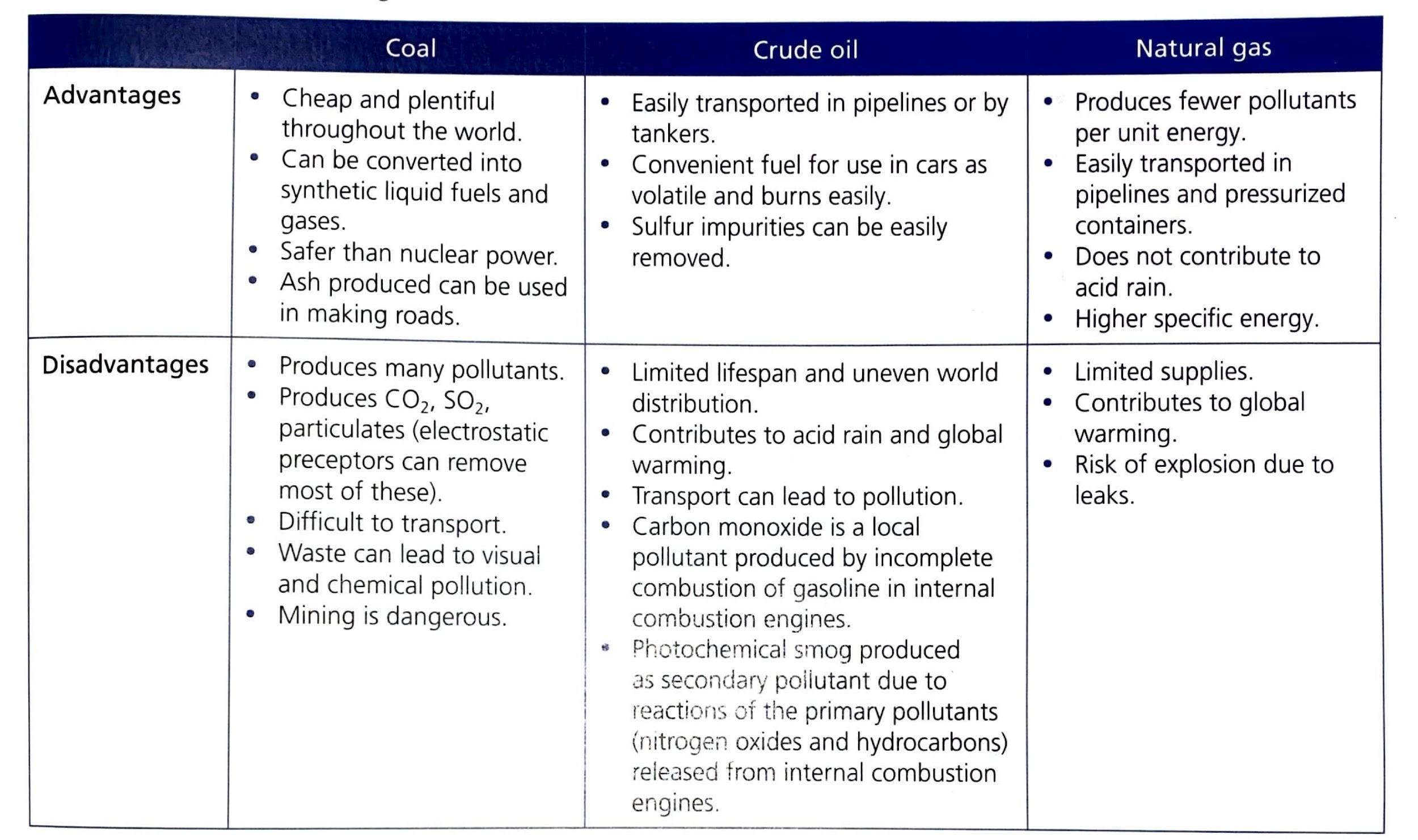
The Paradox of Coal Utilization
Despite coal producing the least amount of energy per unit mass and exhibiting the lowest efficiency among the fossil fuels, it is still utilized very frequently. This widespread use can be attributed to its abundance and relatively low extraction costs, making it an economically attractive option for many countries, particularly those with large domestic reserves.
Understanding Incomplete Combustion and Its Consequences
Incomplete combustion is a significant issue, particularly with fuels like coal, which often contain impurities. Longer hydrocarbon chains, characteristic of heavier fuels, require more oxygen for complete combustion, making them more susceptible to incomplete combustion if oxygen supply is limited. The consequences of incomplete combustion are multifaceted and detrimental:
- Less energy is released per mole of fuel compared to complete combustion, reducing the overall efficiency of energy production.
- Harmful by-products are released, including carbon monoxide (CO), a toxic gas, and carbon particulates (soot), which contribute to air pollution and respiratory problems.
- Unreacted fuel can pose a fire hazard, especially in industrial settings.
Considering these factors, natural gas (methane) is the least likely fossil fuel to undergo incomplete combustion because it has the shortest hydrocarbon chain, requiring less oxygen for complete oxidation compared to the longer chains found in crude oil and coal.
Illustrating Different Degrees of Combustion
The combustion of various carbon-containing substances can result in different products depending on the availability of oxygen. For instance, the complete combustion of carbon, propane, or ethanol yields carbon dioxide and water when oxygen is plentiful. However, with limited oxygen, incomplete combustion occurs, leading to the formation of carbon monoxide and/or carbon particulates (soot), in addition to carbon dioxide and water. When oxygen is extremely limited, the production of carbon monoxide and soot becomes more pronounced.